What is a Clean Room?

A clean room (or cleanroom) is an enclosed control environmental space within a research or manufacturing facility which is designed to control contamination levels, temperature, pressure, and humidity to meet certain environmental conditions.
Cleanrooms are a critical part of a wide range of industries and research facilities, including pharmaceuticals, food & packaging, biotechnology, microelectronics, nutraceuticals, and medical cannabis and many more.
The aim of a cleanroom is to ensure quality product, research integrity, and the safety of workers.
Why Do You Need a Clean Room?
Cleanrooms are used in industries where air particles can adversely impact the outcome of finished physical products. A clean room is required when you need to:
- Identify specific threats that can affect the purity of products.
- Ensure worker safety (some raw materials used for product manufacture can release hazardous contaminants).
- Protect end-users like patients and healthcare providers from biological contamination.
- Access information on the performance of the heating, ventilation, and air conditioning systems.
- Meet ISO class standards for product or process certification.

What is a Modular Clean Room?
A modular cleanroom is a prefabricated, freestanding structure used in new and rapidly growing industries. Solid walls are used in the construction of modular cleanrooms to make them robust, yet simple. The idea behind this type of design is to make it possible for the cleanroom to be extended or relocated to fit any change in demand.
From a few square meters to hundreds of square meters, modular cleanrooms are built off-site and installed on-site, allowing for an efficient construction process that complies with ISO standards.
Modular cleanrooms should include the following components:
- Prefabricated Modular walls.
- Suspended – Walkable ceiling.
- Cleanroom Doors
- Cleanroom Windows / Vision panels
- Coving
- Lab casework / lab furniture
How Are Clean Room Classifications Determined?
The classification of a cleanroom depends on several factors, including air quality inside the clean space. The air quality can be determined from the particulate test, and amount of air required within the cleanroom – air change per hour (ACH).
A cleanroom must be constructed, designed, and operated according to the ISO 14644-1 classification of cleanliness. The more vulnerable a product is to contamination, the higher the level of cleanliness required.
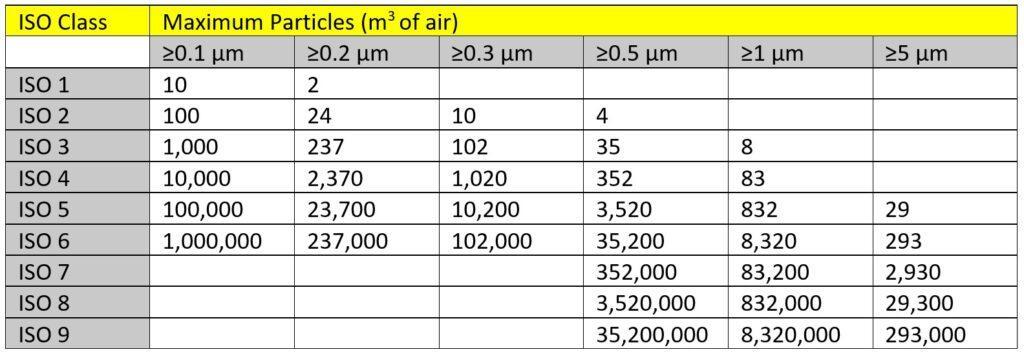
There are nine ISO cleanroom classes: ISO 1, ISO2, ISO 3, ISO 4, ISO 5, ISO 6, ISO 7, ISO 8, and ISO 9. ISO 1 indicates the highest standard of cleanliness, while ISO 9 represents the “dirtiest” class.
As an example, an ISO 7 cleanroom is rated at 352,000 parts per cubic meter, which means that there must be no more than 352,000 particles greater than 0.5µm in the cleanroom. ISO 7 cleanrooms use HEPA filter systems to ensure air cleanliness levels do not exceed 10,000 particles (≥0.5µm) per cubic foot.
Typically, a GMP (Good Manufacturing Practice) cleanroom in the ISO class 7 or higher should have an anteroom for gowning; this is where garbing procedures, personnel hand hygiene, and other activities that could produce large amounts of particles are performed. The anteroom keeps street dirt away from the cleanroom.
How Can You Keep Your Clean Room Free of Contaminants?
Here are five ways you can minimize the contaminants in a cleanroom:
1. Practice Good Hygiene
One of the biggest threats to cleanrooms is personnel-generated contamination. Every work environment should encourage standard cleanroom methods and protocols like:
Use of Proper Gowning Garments
Gravity pulls particulates downwards and garments are influenced by the pull of gravity. As a result, it is important that you do protective garments that will prevent contaminants from reaching clean parts of the garment.
Cleanroom garments are designed to provide coverage and ensure control of microbial contamination. They also protect you, the work environment, and manufactured products from contamination. Cleanroom garments may include hooded barrier coveralls designed to protect personnel from hazardous solutions and barrier gowns used for the administration of cytotoxic medication.
Cleanroom smocks and pocket coveralls can be used in less critical clean room areas.
Move Slowly
Rapid movement in the cleanroom can lead to the spread of particles and contaminants. Cleanroom personnel should move slowly and deliberately around the workstation when entering or exiting the cleanroom. Slow movement can also prevent slips, falls, and injuries.
2. Temperature and Humidity
Most cleanrooms have humidity specifications ranging between 30% and 40% RH and temperature specifications of 21 °C (or 69.8°F) + 2°C. This humidity and temperature level creates an ideal cleanroom environment that prevents bacterial growth. It also creates an ideal environment for employees, ensuring their comfort in work inside this clean space for longer period.
3. Use the Right Supplies
Office supplies are one of the most overlooked contamination sources in a cleanroom. Purchase cleanroom-compliant versions of office supplies like mouse pads, paper, notebooks, and pens.
It is important that you keep these supplies in the cleanroom; you can contaminate the cleanroom environment by simply moving a pen from an office to a cleanroom. Also avoid posting anything on the walls like sticky notes, as these can release particles.
4. Avoid Unnecessary Conversations
Every spoken word produces and expels saliva particles into the air. Although cleanroom personnel often wear facemasks, they are not always 100% effective. While speaking is necessary to perform certain tasks in the cleanroom, avoiding unnecessary talk can reduce the emission of saliva particles, which ultimately reduces the risk of contamination.
5. Use the Right Cleaning Materials
Cleaning supplies like swabs, wipes, mops, and brooms used to keep cleanroom flooring, coving and other parts of the cleanroom should be rated for your ISO class before use in the cleanroom.
Using just any cleaning material can compromise the sterility of a cleanroom. It is also important that you ensure that cleaning materials for the cleanroom are only used in the cleanroom.
How Does a Clean Room Control Contamination Levels?
The following elements work together to create a cleanroom environment that keeps contamination away:
High-efficiency particulate air (HEPA) filters
HEPA filters are critical for contamination control in the cleanroom. They filter particles are tiny as 0.3 microns with about 99.9% minimum particle-collection efficiency. The air within the cleanroom space constantly passes through HEPA filters to eliminate airborne contaminants and allow the circulation of fresh air.
Ventilation System
Ventilation is simply air exchange – the replacement of total air with fresh air. Cleanroom ventilation is determined by what is required to ensure the maintenance of indoor air quality, maintenance of building pressurization, and replacement of process exhaust.
Pressurization
A cleanroom should be maintained at static pressures greater than the surrounding air to prevent infiltration from outside. This is known as differential pressure.
Negative differential pressure is only used when dealing with specific hazardous materials.
Conclusion
Clean room technologies play a vital role in a range of industries by ensuring the purity and integrity of products and processes. When properly constructed and designed, cleanrooms are high-performance machines that are essential for maintaining the safety of products, end-users, and workplace personnel.
GET IN TOUCH
Complete the form below to get in touch with our team.
