
Introduction
When cleanroom environmental controls drift out of spec, the consequences hit fast: scrapped batches, failed audits, unplanned downtime. Temperature, humidity, pressure, airflow, and filtration aren't background systems—they're the foundation your contamination prevention and regulatory compliance stand on.
When these systems fail, the fallout can include:
- Product contamination and batch losses
- Failed ISO or GMP compliance audits
- Costly production shutdowns
- Safety risks to personnel and processes
This guide covers why environmental control maintenance matters, the four maintenance approaches you can implement, how to identify when systems need attention, scheduling guidelines based on cleanroom classification, and best practices for sustaining cleanroom integrity over your facility's lifetime.
TL;DR
- Proactive maintenance prevents contamination, ensures regulatory compliance, and protects product quality
- A layered strategy — routine, corrective, predictive, and overhaul — covers all failure modes
- Monitor parameter drift, airflow issues, filter loading, and alarm frequency as early warning signs
- Schedule maintenance based on ISO classification and operational intensity, from daily checks to annual certifications
- Predictive maintenance reduces costs by 18–25% and extends equipment life by 20–40%
Why Maintenance of Cleanroom Environmental Controls Is Important
Environmental controls directly determine particle counts, microbial contamination levels, and product sterility. In pharmaceutical manufacturing, unplanned downtime costs between $100,000 and $500,000 per hour, while semiconductor facilities can lose over $1 million per hour. These figures make maintenance non-negotiable for quality assurance and business continuity.
Regulatory frameworks also mandate documented maintenance programs:
- ISO 14644-2:2015 requires documented monitoring plans based on risk assessment, with regular calibration of all instrumentation
- EU GMP Annex 1 (2022) mandates requalification every 6 months for Grade A/B areas and 12 months for Grade C/D
- FDA 21 CFR Part 211 (pharmaceutical) and Part 820 (medical devices) require written maintenance procedures and records
The cost case is equally clear. Preventive maintenance runs 3–5 times less than emergency repairs, and predictive maintenance strategies reduce overall maintenance costs by 18–25% while extending equipment lifespan. Well-maintained HVAC systems and HEPA filters can last 20–40% longer than neglected ones — a meaningful return on capital-intensive cleanroom infrastructure.
Proper maintenance also protects process integrity across every cleanroom type:
- Humidity control prevents electrostatic discharge in semiconductor environments, protecting sensitive components
- Temperature stability preserves material integrity in biotech processes, where minor fluctuations can compromise product efficacy
- Pressure differentials block cross-contamination in pharmaceutical manufacturing, keeping sterile zones isolated from lower-grade areas
Types of Maintenance for Cleanroom Environmental Controls
Effective cleanroom maintenance combines multiple approaches—each addresses different failure modes and operational needs. Understanding which strategy fits your facility's ISO classification, production schedule, and risk tolerance is the starting point for building a reliable maintenance program.
Routine / Preventive Maintenance
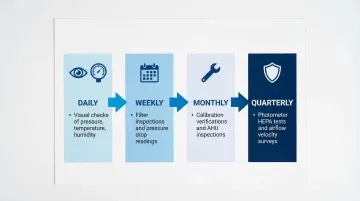
Preventive maintenance (PM) follows scheduled, time-based intervals regardless of system condition. Tasks include:
- Daily visual checks of pressure gauges and temperature/humidity displays
- Weekly filter inspections and pressure drop readings
- Monthly calibration verifications and AHU component inspections
- Quarterly photometer tests for HEPA filter integrity and airflow velocity surveys
PM works best in ISO Class 6–8 cleanrooms with predictable usage patterns, where scheduled intervals align naturally with regulatory documentation requirements.
Cost considerations: PM offers predictable budgeting with scheduled labour and parts. Industry benchmarks suggest allocating approximately 10–15% of initial HVAC system cost annually for preventive maintenance activities.
Corrective / Reactive Maintenance
Corrective maintenance responds to system failures or alarm conditions: temperature excursions, filter blowouts, broken dampers, or failed sensors. While some reactive maintenance is inevitable, over-reliance creates significant risks.
The dangers of reactive-only strategies:
- Unplanned downtime disrupts production schedules and customer commitments
- Emergency repairs cost 3–5 times more than preventive work
- Repeated failures indicate systemic issues requiring root cause analysis
- Regulatory documentation burden increases with each deviation event
Reserve reactive maintenance for genuine exceptions: unexpected failures between scheduled PM cycles, one-off equipment issues, or external disruptions like power outages and extreme weather events.
Predictive / Condition-Based Maintenance
Predictive maintenance (PdM) uses real-time monitoring systems, trend analysis, and equipment health indicators to predict failures before they occur. Instead of fixed intervals, you schedule maintenance based on actual equipment condition.
Key monitoring parameters include:
- Differential pressure trends across filters indicating loading progression
- Motor vibration analysis detecting bearing wear before failure
- Energy consumption patterns revealing system inefficiencies
- Airflow velocity degradation tracked over time
Technologies enabling predictive maintenance include IoT sensors, building management systems (BMS), automated alert systems, and data analytics platforms that track parameter drift and trigger maintenance before critical thresholds.
Predictive maintenance delivers 18–25% cost reductions through optimized scheduling and extended equipment life. The approach requires higher upfront investment in monitoring systems (15–25% above basic controls), but ROI ratios of 10:1 to 30:1 within 12–18 months justify the investment for pharmaceutical, semiconductor, and aerospace operations where unplanned downtime carries regulatory and financial consequences.

Major / Overhaul Maintenance
Overhaul maintenance covers complete filter replacements, AHU rebuilds, control system upgrades, ductwork cleaning, and full recertification — typically reserved for end-of-life components or significant facility changes.
When overhaul maintenance is required:
- After 3–5 years of operation for HEPA filters, even if pressure drop seems acceptable
- When upgrading to stricter ISO classifications
- Following facility expansions that increase system load
- When cumulative minor issues indicate aging infrastructure
Overhauls require cleanroom shutdown (or temporary relocation of operations), third-party recertification, and full documentation updates to validation protocols.
Plan for capital expenditure of 30–50% of the original installation cost. Budget 12–18 months ahead — both to secure financing and to schedule production around the downtime window without disrupting customer commitments.
How to Check If Cleanroom Environmental Controls Need Maintenance
These early warning signs indicate maintenance is overdue or that current schedules need adjustment. Spotting them before they escalate keeps your facility compliant and your production running.
Temperature and Humidity Fluctuations
Acceptable ranges vary by cleanroom classification and process requirements—typically ±2°C for pharmaceutical manufacturing and ±5% RH for medical device assembly. Deviations beyond these documented limits signal underlying HVAC problems worth investigating immediately.
Common causes:
- Failing temperature sensors providing inaccurate readings
- Refrigerant leaks in cooling systems reducing capacity
- Humidifier/dehumidifier malfunctions
- Inadequate AHU capacity for actual heat loads
Warning signs:
- Increasing frequency of alarm events over days or weeks
- Widening temperature/humidity swings throughout the day
- Inability to recover to setpoint after door openings
- Seasonal variations exceeding historical norms
Differential Pressure Problems
Differential pressure between cleanroom zones (typically 10-15 Pascals between adjacent rooms) is critical for contamination control. Loss of pressure differential indicates serious airflow compromise.
Common causes:
- HEPA filter loading (most common—responsible for majority of pressure issues)
- Exhaust system imbalances
- Supply fan failures or reduced capacity
- Door seal degradation allowing air leakage
- Return air pathway blockages
Warning signs:
- Pressure gauge readings falling below minimum thresholds
- Reversed pressure differentials (cleanroom going negative when it should be positive)
- Smoke pattern tests showing air migrating in the wrong direction
Airflow Velocity and Pattern Changes
ISO 14644-3 specifies airflow velocity ranges—typically 0.3-0.5 m/s for unidirectional flow and 20-60 air changes per hour for non-unidirectional flow. Deviations compromise particle removal efficiency.
Common causes:
- Filter loading reducing airflow volume
- Fan belt slippage or wear
- Variable frequency drive (VFD) malfunctions
- Damper positions drifting from calibrated settings
Indicators of problems:
- Visible dust accumulation in previously clean areas
- Increased particle counts during certification tests
- Uneven airflow distribution detected by anemometer surveys
- "Dead zones" where air appears stagnant
Filter Condition and System Alarms
HEPA/ULPA filters have finite lifespans, and monitoring their condition prevents sudden failures. Differential pressure across filters serves as the key indicator.
Track these warning signs:
- Pressure drop across filter banks increasing beyond manufacturer specifications (typically 250 Pa signals replacement)
- Visible filter damage during inspections: tears, gasket failures, frame warping
- Increasing frequency of low airflow alarms
- Filter integrity test failures showing penetration >0.01%
- BMS alerts for fan motor overcurrent (working harder to push air through loaded filters)

Energy Consumption and Equipment Performance
Cleanroom HVAC typically represents 40-60% of facility energy costs. Unusual consumption patterns are often the first sign of inefficiency or impending equipment failure.
Monitor these indicators:
- Monthly energy bills showing upward trends without increased production
- Fan motors running continuously at maximum capacity instead of modulating based on demand
- Cooling/heating systems cycling more frequently than normal
- Longer time to reach setpoint after disturbances
- Inability to maintain conditions during peak occupancy
- Unusual noises from AHUs or fans suggesting bearing wear or imbalance
Cleanroom Environmental Control Maintenance Schedule
No single maintenance schedule works for every cleanroom. Three variables determine the right cadence for your facility:
- ISO classification: Class 5 environments require more frequent checks than Class 8
- Operational intensity: A 24/7 manufacturing floor needs closer attention than an intermittent R&D lab
- Regulatory framework: Pharmaceutical facilities face stricter documentation requirements than electronics manufacturing
| Task | Daily/Per-Shift | Weekly/Monthly | Quarterly/Semi-Annual | Annual |
|---|---|---|---|---|
| Monitoring | Visual inspection of pressure gauges, temperature/humidity displays, alarm panel check | Temperature/humidity data logger review, pressure drop readings | Anemometer airflow velocity surveys, sensor calibration verification | Full ISO certification with particle counting |
| Filters | Airflow indicator verification | HEPA filter pressure drop readings | Filter integrity testing (photometer) | Complete HEPA filter replacement (or as indicated) |
| HVAC Systems | Visual check of AHU operation | Belt and bearing inspection, emergency backup system tests | Control system software updates, deep inspection of components | AHU deep cleaning and component replacement |
| Facility | General cleanroom condition check | Door seal visual inspection | Door seal performance testing | Control system validation, documentation review and update |
Operational variables to consider:
- High-usage facilities running 24/7 manufacturing shifts should increase inspection frequency by roughly 50%
- R&D labs with intermittent operation may extend some intervals, but annual certifications are never optional
- Continuous monitoring systems reduce the need for manual checks — though they require their own calibration schedules
Regulatory requirements set the floor for how often requalification must occur. EU GMP Annex 1 mandates requalification every 6 months for Grade A/B areas (ISO Class 5) and 12 months for Grade C/D areas (ISO Class 7/8). Each requalification event must include particle counting, filter integrity testing, airflow volume/velocity verification, and pressure differential confirmation.
Modular Cleanroom Maintenance Advantages
Modular cleanroom designs offer clear maintenance advantages over traditional stick-built cleanrooms. The prefabricated approach addresses the three most common maintenance pain points:
- HVAC access — removable wall panels let technicians reach mechanical components without tearing into finished surfaces
- Documented protocols — pre-designed systems come with defined maintenance schedules, reducing guesswork during audits
- Reconfigurability — spaces can be expanded or reconfigured without disrupting active environmental controls
ACH Engineering's modular cleanroom solutions feature walkable ceiling systems that allow maintenance personnel to access HEPA filtration systems and environmental controls from above without contaminating the cleanroom environment below. In traditional cleanrooms, ceiling access almost always disrupts the controlled environment — this design removes that risk entirely.

Wall panels, doors, and ceiling systems can be accessed, removed, or replaced without major reconstruction. Standardized components mean replacement parts are readily available — reducing downtime and keeping cleanroom certification current without costly project delays.
ACH Engineering supports clients through the full project lifecycle with cleanroom project management, including maintenance planning documentation that holds up under regulatory audits — whether the facility is in Ontario, Alberta, or elsewhere across North America.
Conclusion
Cleanroom environmental control maintenance is a regulatory requirement and a business necessity — one that directly shapes product quality, compliance status, and operational costs. Unplanned downtime in regulated facilities routinely runs into six figures per hour, and maintenance budgets that aren't spent strategically tend to cost far more in emergency repairs and failed audits.
The most effective programs combine three layers of protection:
- Routine scheduled maintenance that prevents failures before they occur
- Condition monitoring that flags degradation early, before it becomes critical
- Reactive response protocols that contain emergencies when prevention falls short
With that structure in place and documentation to back it up, maintenance stops being a cost center and starts functioning as an asset — one that preserves the significant investment a cleanroom represents.
If you're revisiting your maintenance program or building one from scratch, the starting point is a documented baseline: current equipment condition, calibration status, and filter change history. Everything else — schedules, monitoring thresholds, response plans — follows from that foundation.
Frequently Asked Questions
How to maintain a clean room environment?
Cleanroom maintenance rests on three pillars: environmental control (HVAC, filtration, monitoring systems), personnel and material protocols (gowning, cleaning, traffic patterns), and continuous monitoring. Document corrective actions whenever parameters drift outside specifications to maintain ISO classification and regulatory standing.
What is environmental monitoring in cleanrooms?
Environmental monitoring is the continuous or periodic measurement of critical parameters: particle counts, temperature, humidity, differential pressure, airflow velocity, and microbial levels. The data verifies that the cleanroom holds its ISO classification and provides documented evidence of control for regulatory compliance.
What is an example of environmental control?
One common example is holding a +15 Pascal differential pressure between an ISO Class 7 compounding room and an adjacent ISO Class 8 corridor. Balanced supply and exhaust airflow achieves this, monitored by magnehelic gauges, with alarms set to trigger at ±3 Pascals to prevent contamination migration.
What are cleanrooms and associated controlled environments?
Cleanrooms are ISO 14644-certified environments with documented particle count limits, specific construction standards, and continuous environmental control. Controlled environments monitor parameters like temperature or humidity but don't require ISO certification — cleanrooms carry stricter regulatory requirements for pharmaceutical, medical device, and semiconductor applications.
How often should cleanroom environmental controls be calibrated?
Temperature and humidity sensors typically require calibration every 6-12 months, differential pressure gauges every 12 months, and airflow measurement instruments annually or per manufacturer specifications. All calibration activities must be documented with traceable standards and certificates for regulatory compliance.
What are the most common causes of cleanroom environmental control failures?
The top causes are HEPA filter loading or damage (approximately 40% of failures), HVAC component wear including fan motors, belts, and bearings (30%), control system sensor drift or failure (20%), and inadequate preventive maintenance allowing small issues to cascade into system-wide problems.


