
Introduction: The Critical Importance of Cleanroom Contamination Control
Contamination control failures carry devastating consequences in regulated industries. Between 2012 and 2023, 93% of sterile drug recalls were traced to sterility failures, with a single contaminated batch costing manufacturers US$1–2 million or more. Regulatory scrutiny continues to intensify—the FDA issued 561 Form 483s to drug facilities in FY2024, with environmental monitoring and contamination control deficiencies among the most frequently cited violations.
Those stakes make contamination control far more than a housekeeping exercise. Across pharmaceutical, biotech, medical device, and electronics manufacturing, it directly determines product integrity, patient safety, and regulatory standing.
Understanding ISO Cleanroom Classification Standards
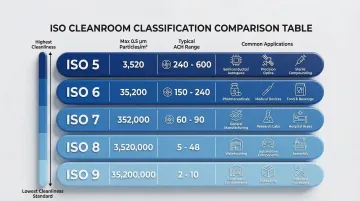
ISO 14644-1:2015 provides the foundational framework for classifying cleanroom air cleanliness by particle concentration. The classification system defines maximum allowable particle counts at specific size ranges, with lower ISO numbers indicating stricter contamination control requirements.
Particle Count Limits and Air Change Requirements
| ISO Class | 0.5 µm Particles/m³ | Typical ACH Range | Common Applications |
|---|---|---|---|
| ISO 5 | 3,520 | 300-480 | Aseptic filling, sterile compounding, microelectronics |
| ISO 6 | 35,200 | 180 | Medical device assembly, lower-risk sterile zones |
| ISO 7 | 352,000 | 60 | Pharmaceutical preparation, compounding background zones |
| ISO 8 | 3,520,000 | 20 | General clean manufacturing, equipment cleaning |
| ISO 9 | 35,200,000 | 10-15 | Support zones, material staging areas |
Your target ISO class drives every downstream design decision — from HVAC sizing and air change rates to gowning protocols and environmental monitoring frequency. ISO 5 environments require unidirectional airflow and continuous particle monitoring, while ISO 8 facilities operate with far less intensive controls. Getting the classification right at the outset prevents costly retrofits and compliance gaps later.
Primary Sources of Cleanroom Contamination
The Human Factor: Personnel as the Largest Contamination Source
Humans represent the single largest contamination source in controlled environments, responsible for 75–80% of cleanroom contamination. The human body continuously sheds skin flakes carrying viable microorganisms, with each person shedding approximately 500 million skin cells daily.
Particle generation varies dramatically by activity level:
- Sitting motionless: ~100,000 particles (≥0.3 µm) per minute
- Standing still: ~500,000 particles per minute
- Walking: ~1,000,000 particles per minute
Beyond skin shedding, personnel introduce contamination through cosmetics, hair, jewelry, and improper gowning techniques. Even properly gowned operators generate significant particle counts through movement, making personnel training and behavior protocols the most important factors to control.
Airborne Contamination and Environmental Factors
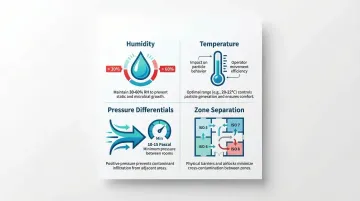
Room air naturally contains millions of particles that pose constant contamination risks. HEPA filtration systems remove 99.97% of particles at 0.3 µm, but effectiveness depends on proper air change rates and pressure differentials. Four environmental variables directly determine how well those systems perform:
- Humidity below 30% increases static electricity and particle attraction; above 60%, it promotes microbial growth
- Temperature fluctuations affect particle behavior and operator comfort, indirectly driving more movement and more shedding
- Pressure differentials of at least 10–15 Pascals between adjacent rooms block contamination ingress from lower-grade areas
- Inadequate zone separation allows particle migration through doorways, pass-throughs, and shared HVAC systems
Materials, Equipment, and Process-Related Contamination
Manufacturing equipment and materials introduce contamination through multiple pathways. Machinery generates metal particles through friction, hydraulic systems can leak oils and lubricants, and vibrating equipment disperses settled particles back into the air.
Common material and process contamination sources:
- Raw materials and packaging containing embedded particles or microbial contamination
- Cleaning solutions and chemicals that leave residues or introduce new contaminants
- Static electricity attracting and holding particles on surfaces and equipment
- Tools and fixtures that shed particles through wear and handling
Static control is often underestimated here — a single ungrounded surface can accumulate enough charge to pull particles back onto freshly cleaned equipment.
Facility Infrastructure and Design Flaws
Even well-maintained cleanrooms hide contamination sources within their infrastructure. Aging facilities develop leaks, peeling paint, and degraded seals that continuously introduce particles. Improperly designed airflow patterns create dead zones where contamination accumulates.
Infrastructure-related contamination sources include:
- HEPA filter leaks allowing unfiltered air bypass
- Deteriorating wall and ceiling panels shedding particles
- Malfunctioning HVAC systems failing to maintain proper pressure and air changes
- Poor cleanroom layout creating turbulent airflow and particle recirculation
Most of these problems are design failures, not maintenance failures — which is why cleanroom layout and material selection decisions made at the planning stage have consequences that last the life of the facility.
Personnel-Related Contamination Control Strategies
Gowning Protocols and PPE Requirements
Proper gowning serves as the primary barrier between personnel and the cleanroom environment. IEST-RP-CC003 provides comprehensive guidance on garment selection, emphasizing non-shedding materials and particle-containing construction features.
Critical gowning protocol elements:
- Don garments in sequence from cleanest to dirtiest areas to minimize particle generation
- Use low-linting fabrics with tunnelized elastic and bound seams to contain particles
- Ensure integrated hoods, boot covers, and face masks provide full-body coverage
- Size garments correctly — gaps allow particles to escape
- Replace garments based on classification requirements and visible contamination
Sterile vs. non-sterile garments: EU GMP Annex 1 mandates sterilized protective garments for Grade A/B zones (equivalent to ISO 5), including sterile gloves with sleeves tucked inside. Lower classifications permit clean-processed garments that minimize particles without requiring sterilization.
Personal Hygiene and Behaviour Protocols
People are one of the highest sources of contamination in any cleanroom. Strict hygiene and behaviour standards limit particle generation and microbial risk — particularly in higher-grade environments where cosmetics, jewellery, and nail polish are prohibited.
Key hygiene and behaviour controls:
- Restrict cosmetics, perfumes, and lotions that generate particles
- Prohibit jewellery, watches, and accessories that trap contamination
- Require handwashing before gloving and after breaks
- Implement cuff sealing techniques to prevent particle escape at wrist and ankle openings
- Minimize movement through slow, deliberate actions
- Prohibit touching face, adjusting garments, or other particle-generating behaviours
Training and Competency Programs
Research shows critical procedural skill performance can decline as early as 3 months after training, with half of initial gains lost after roughly 6.5 months. Without structured reinforcement, even well-trained personnel drift back toward non-compliant habits.
An effective training program includes:
- Comprehensive initial qualification covering contamination sources, gowning, and cleanroom behaviour
- Annual (or more frequent) refresher training to counteract skill decay
- Regular competency assessments to verify proper technique
- Maintained training records for regulatory compliance and trend analysis
- Hands-on practice and visual demonstrations rather than passive lectures
Environmental and Facility Control Measures
Air Filtration and HVAC System Management
HEPA and ULPA filters form the foundation of airborne contamination control. HEPA filters capture ≥99.97% of particles at 0.3 µm, while ULPA filters achieve ≥99.9997% efficiency at the same size.
Filter specifications and maintenance:
- Test filter integrity using IEST-RP-CC034 leak testing procedures after installation and periodically during operation
- Replace filters based on pressure drop measurements and integrity test results, typically every 6-12 months for critical grades
- Calculate required CFM using the formula: (Room Volume × ACH) ÷ 60 minutes
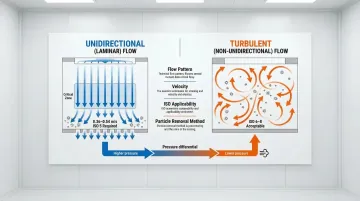
Airflow patterns and pressure control:
- Unidirectional (laminar) flow: Required for ISO 5 zones, sweeps particles away from critical areas with parallel airstreams at 0.36-0.54 m/s
- Turbulent (non-unidirectional) flow: Acceptable for ISO 6-8, mixes supply air with room air through induction
- Pressure differentials: Maintain minimum 10-15 Pascals between adjacent rooms, with continuous monitoring and alarms for deviations
Surface Cleaning and Disinfection Protocols
Effective cleaning, disinfection, and sterilization serve distinct functions — and confusing them leads to protocol gaps. Each process targets a different threat level and requires separate validation.
Cleaning vs. disinfecting vs. sterilizing:
- Cleaning: Removes visible soil, product residues, and disinfectant buildup
- Disinfecting: Kills vegetative bacteria and fungi but may not eliminate spores
- Sterilizing: Destroys all microorganisms including bacterial and fungal spores
Disinfectant selection and rotation:
- Select agents validated against target organisms on specific surface materials
- Rotate disinfectants including a sporicidal agent to control spore-forming organisms and prevent resistant flora development
- Use sterile disinfectants and detergents in Grade A/B areas
- Document contact times, concentrations, and application methods
Cleaning technique and frequency:
- Clean from cleanest to dirtiest areas and from top to bottom
- Use cleanroom-grade, low-linting wipes and mops
- Establish frequencies based on classification, with daily cleaning for ISO 5-6 and less frequent schedules for lower grades
- Validate cleaning effectiveness through surface sampling and visual inspection
Environmental Monitoring and Real-Time Systems
Continuous monitoring provides immediate contamination event detection and trend analysis capabilities. EU GMP Annex 1 mandates continuous viable air monitoring for Grade A zones during critical processing.
Monitoring system components:
- Particle counters: Measure non-viable particle concentrations at 0.5 µm and 5.0 µm in real-time
- Microbial samplers: Collect viable organisms through volumetric air sampling and settle plates
- Environmental sensors: Track temperature, humidity, and pressure differentials continuously
- Data management systems: Aggregate monitoring data, generate alerts, and support trend analysis
Monitoring frequency and locations: ISO 14644-2 requires monitoring plans based on risk assessment. Higher classifications demand more frequent monitoring, with ISO 5 zones requiring continuous or multiple daily measurements at locations near critical operations.
Material and Equipment Decontamination
Every item entering a cleanroom is a potential contamination vector. Pass-through chambers provide controlled transfer points where materials are decontaminated before crossing into classified zones.
Decontamination methods:
- Surface wiping: Remove particles from packaging using sterile disinfectants
- ETO gas sterilization: ISO 11135:2014 specifies requirements for low-temperature sterilization of heat-sensitive materials
- Gamma irradiation: ISO 11137-1 defines radiation sterilization processes achieving SAL of 10^-6
- Cleanroom-grade packaging: Use materials that don't shed particles or generate static
Material transfer procedures:
- Inspect all items before transfer for visible contamination
- Remove outer packaging in lower-grade areas before pass-through
- Use anti-static protocols to prevent particle attraction during transfer
Static Control Measures
Static electricity attracts and holds particles on surfaces — and the same transfer process that brings materials into a cleanroom can generate significant charge. Discharge events then damage sensitive products and dislodge settled contamination. A layered approach addresses both generation and accumulation.
Static control methods:
- Ionizing equipment: Install ionizing bars and anti-static guns to neutralize charged surfaces
- Antistatic garments: Use cleanroom apparel certified to EN 1149-5 standards for electrostatic dissipative properties
- Humidity control: Maintain relative humidity above 30% to reduce static generation
- Anti-static materials: Use conductive or dissipative packaging, flooring, and work surfaces
- Grounding: Ensure proper grounding of equipment, personnel, and work surfaces
Equipment and Material Management Best Practices
Every item brought into a cleanroom is a potential contamination source. Selecting the right equipment and managing materials carefully are foundational steps that prevent particle generation before it starts.
Equipment Selection
Choose cleanroom-compatible equipment that minimizes particle generation and outgassing. Avoid materials with paper cores, adhesives that leave residues, or components that shed particles through wear. Key selection criteria include:
- Low particle generation during operation
- Minimal outgassing of volatile organic compounds
- Chemical compatibility with cleaning and disinfection agents
- Smooth, easy-to-clean surfaces without crevices that trap contamination
Maintenance and Calibration
Even well-selected equipment introduces contamination risk if it isn't properly maintained. A structured schedule keeps equipment performing reliably and prevents gradual particle buildup:
- Establish preventive maintenance schedules based on manufacturer recommendations
- Clean equipment thoroughly before and after maintenance activities
- Calibrate monitoring instruments regularly to ensure accurate measurements
- Document maintenance activities, calibration results, and equipment performance trends
Material Labeling and Identification
Standard labels and tapes are among the most overlooked contamination sources in a cleanroom. Switching to compatible alternatives removes an unnecessary risk:
- Use cleanroom-compatible labels and tapes without paper cores
- Select adhesives that don't leave residue or generate particles
- Implement clear identification systems to prevent mix-ups and contamination events
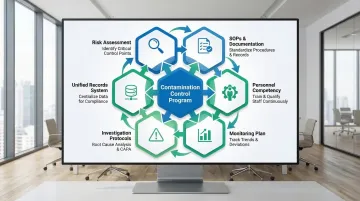
Implementing a Comprehensive Contamination Control Program
Effective contamination control requires integrating multiple strategies into a cohesive program with clear accountability and continuous improvement mechanisms.
Program components:
- Assess contamination sources, evaluate their impact, and prioritize control measures by risk level
- Document gowning protocols, cleaning schedules, environmental monitoring plans, and material transfer procedures in formal SOPs
- Define competency standards and retraining frequencies for all personnel who access the controlled environment
- Establish sampling locations, frequencies, and alert/action limits within a structured monitoring plan
- Set investigation and response protocols for contamination events before they occur — not after
- Maintain cleaning logs, environmental monitoring records, training records, and deviation investigations in a unified documentation system
Coordinating all of these elements — from facility infrastructure and HVAC design to monitoring protocols and personnel training — is where many programs fall short. Each component affects the others, and gaps in one area can undermine even well-executed work elsewhere.
ACH Engineering's turnkey cleanroom solutions approach addresses this by managing the full scope of cleanroom projects from initial design through operational validation. Their team understands how contamination control requirements shift across ISO classifications, which means the facility infrastructure, environmental controls, and cleanroom validation services are designed to work as a system — not as separate checkboxes.
Frequently Asked Questions
What should a cleanroom contamination control SOP include?
A complete contamination control SOP should address all critical operational touchpoints:
- Gowning procedures with step-by-step donning sequences
- Cleaning protocols specifying agents and frequencies
- Environmental monitoring schedules with sampling locations
- Material transfer procedures through pass-throughs
- Personnel training requirements and competency assessments
- Documentation practices including cleaning logs and deviation investigations
What are the rules of cleanroom contamination control?
Six core rules govern contamination control in cleanrooms:
- Minimize contamination sources through proper facility design
- Control ingress via gowning protocols and material decontamination
- Maintain HEPA filtration and correct pressure differentials
- Follow strict hygiene protocols at all times
- Conduct regular environmental monitoring with documented results
- Keep comprehensive records of all activities and deviations
What types of contamination must be prevented from leaving a cleanroom?
Controlled materials, hazardous substances, and potentially contaminated items — including product waste, used gowning, and equipment — must be contained and decontaminated before leaving the cleanroom. This prevents particles and microorganisms from spreading to adjacent areas.
What are the FDA guidelines for cleanroom contamination control?
FDA 21 CFR 211 establishes cGMP requirements mandating smooth surfaces, temperature/humidity controls, HEPA filtration, and validated cleaning procedures for aseptic processing. EU GMP Annex 1 covers sterile manufacturing in detail, requiring continuous monitoring and a Sterility Assurance Level (SAL) of 10⁻⁶ for sterile products.
How often should cleanroom environmental monitoring be performed?
Frequency depends on ISO classification. ISO 5 zones require continuous particle monitoring and multiple daily microbial samples; ISO 6–7 zones typically use several checks per shift; ISO 8 zones can be monitored daily or weekly. ISO 14644-2 provides risk-based guidance for setting schedules.
What is the difference between clean-processed and sterile cleanroom garments?
Clean-processed garments are manufactured in controlled conditions to minimize particle generation but are not sterile, making them suitable for ISO 6-8 environments. Sterile garments undergo additional sterilization through gamma irradiation or ETO gas to achieve specific Sterility Assurance Levels required for aseptic environments like ISO 5 Grade A/B zones where sterility is critical.


