Introduction: Understanding Controlled Environments and Cleanrooms
Many facility managers face confusion when choosing between controlled environments and cleanrooms—and for good reason. While these terms are often used interchangeably, they represent distinct solutions with different technical requirements, costs, and applications.
A controlled environment regulates temperature, humidity, and airflow for process stability. A cleanroom goes several steps further, adding stringent particle filtration and contamination control to meet ISO classifications.
That distinction carries a real cost. Cleanroom HVAC systems can consume up to 15 times more energy than standard systems, with operational costs that reflect this intensity.
This guide clarifies the definitions, standards, and applications of both solutions, helping you determine which approach your facility needs.
TLDR: Controlled Environment vs Cleanroom Quick Guide
- Controlled environments regulate temperature, humidity, and pressure for stability—not particle elimination
- Cleanrooms add HEPA/ULPA filtration to meet ISO 14644-1 particle count thresholds (ISO 1–9)
- Choose controlled environments for storage and stability; choose cleanrooms when sterile production or strict particle limits apply
- Cleanrooms require 20–480+ air changes per hour versus 2–20 ACH for controlled environments
- ISO 5 cleanrooms typically require recertification every 6 months per ISO 14644-2; controlled environments typically require annual validation
What is a Controlled Environment?
A controlled environment is an enclosed space where specific parameters—temperature, humidity, pressure, and airflow—are regulated within defined limits to maintain process stability. Unlike cleanrooms, the primary goal is environmental consistency rather than particle elimination.
SEMI S14/S2 defines a controlled environment as a "confined area in which the humidity, temperature, particulate matter, and contamination are precisely controlled within specified parameters." This definition emphasizes control over specified ranges rather than achieving ultra-low particle counts.
Key Parameters in Controlled Environments
Controlled environments regulate multiple variables based on application requirements:
- Temperature: 15°C–25°C for general manufacturing and storage; tighter ±2°C tolerances for sensitive processes. Pharmaceutical controlled room temperature (CRT) targets 20°C–25°C; cold rooms run 2°C–8°C.
- Humidity: 30%–60% RH for pharmaceutical storage and general areas. Cold rooms hold 60%–90% RH to prevent moisture loss; battery dry rooms reach dew points below -30°C (<1% RH).
- Pressure: Neutral or slight positive/negative pressure (±5 Pa), without the strict cascade requirements cleanrooms demand.
- Air exchange rates: 2–20 air changes per hour (ACH) — well below cleanroom standards, but sufficient for temperature and humidity control without high-velocity filtration costs.
- Lighting: Spectrum and intensity are adjustable for specialized applications, particularly photoperiod management in agricultural cultivation.

These parameters can be adjusted by the minute, hour, or seasonally depending on process requirements, providing flexibility that cleanrooms—with their rigid classification standards—cannot offer.
Certification and Monitoring Requirements
Controlled environment certification verifies that temperature and humidity stay within specified ranges — not particle count thresholds. Validation typically occurs annually or semi-annually based on industry requirements and internal quality standards.
The process covers three core steps:
- Sensor calibration verification — confirm instruments are reading accurately
- Parameter stability testing — document consistency over representative time periods
- Excursion documentation — record and investigate any out-of-range events
Unlike cleanrooms, controlled environments don't require particle counters or HEPA filter integrity testing.
Common Industries Using Controlled Environments
- Life science laboratories: Stable conditions for experiments without sterile production requirements
- Pharmaceutical storage: Controlled room temperature warehouses maintaining drug stability outside active manufacturing
- Agricultural cultivation: Cannabis facilities target 22°C–28°C and 55%–65% RH to prevent mold and preserve terpene profiles; vertical farms and greenhouses use similar setups to maximize yield
- Battery manufacturing: Lithium-ion dry rooms hold 20°C–25°C with dew points of -40°C to -60°C — moisture control drives the design, not particle counts
- Food and beverage processing: Cold chain storage (4°C–8°C) and processing areas needing sanitary conditions without ISO particle classification
What is a Cleanroom?
A cleanroom is a specialized controlled environment where the concentration of airborne particles is controlled, classified, and continuously managed. ISO 14644-1:2015 defines it as:
"A room in which the concentration of airborne particles is controlled and classified, and which is constructed and used in a manner to minimize the introduction, generation, and retention of particles inside the room."
Cleanrooms maintain separation from outside air through positive pressure and use HEPA or ULPA filtration systems to achieve specific cleanliness levels measured in particles per cubic meter.
Technical Requirements of Cleanrooms
HEPA/ULPA filtration is the foundation of particle control. HEPA filters remove at least 99.97% of particles 0.3 µm in diameter; ULPA filters reach 99.9995% at 0.12 µm. Performance standards are defined by IEST-RP-CC001, EN 1822, and ISO 29463.
Air changes per hour (ACH) requirements are far higher in cleanrooms than in standard controlled environments:
- ISO 8 (Class 100,000): Minimum 20 ACH
- ISO 7 (Class 10,000): 40-60 ACH
- ISO 5 (Class 100): 300-480 ACH or unidirectional airflow velocities of 0.36-0.54 m/s

Positive pressure differentials of at least 10-15 Pascals (Pa) must be maintained between adjacent rooms of differing cleanliness classifications. This prevents contamination ingress and requires continuous monitoring with alarms.
Gowning protocols are the primary control for human-generated contamination:
- ISO 5 / Grade A: Full sterile gowning — masks, hoods, and boots
- ISO 7/8: Hairnets, lab coats or coveralls, and shoe covers
ISO 14644-1 and Classification Standards
ISO 14644-1:2015 is the global standard for classifying air cleanliness by particle concentration. The classification system ranges from ISO 1 (cleanest) to ISO 9, with particle count thresholds defined at multiple size ranges.
Key particle count thresholds (particles per cubic meter):
| ISO Class | ≥0.5 µm | ≥1.0 µm | ≥5.0 µm |
|---|---|---|---|
| ISO 5 | 3,520 | 832 | 29* |
| ISO 6 | 35,200 | 8,320 | 293 |
| ISO 7 | 352,000 | 83,200 | 2,930 |
| ISO 8 | 3,520,000 | 832,000 | 29,300 |
Note: Limits for 5.0 µm particles in ISO 5 were removed in the 2015 revision for classification purposes but are still monitored for sterile manufacturing under EU GMP Annex 1.
Historical FED STD 209E mapping: Although cancelled in 2001, FED STD 209E terminology remains prevalent:
- Class 100 ≈ ISO 5
- Class 10,000 ≈ ISO 7
- Class 100,000 ≈ ISO 8
Industry-specific standards: Specific industries layer additional requirements onto base ISO classifications:
- Aerospace: ASTM E2352 "Standard Practice for Aerospace Cleanrooms and Associated Controlled Environments"
- Semiconductor: SEMI standards (such as SEMI F21) reference ISO 14644-1 but add molecular contamination controls
- Pharmaceutical: EU GMP Annex 1 and FDA Guidance reference ISO classes but impose additional microbiological limits
Certification and Validation Process
Cleanroom certification requires rigorous particle testing using discrete particle counters at defined grid locations throughout the space. Testing occurs in three occupancy states: as-built (empty), at-rest (equipment installed, not operating), and operational (full production conditions). Each state captures a different contamination profile — and all three must pass.
Recertification frequency:
- ISO Class 5 and cleaner: Maximum interval of 6 months
- ISO Class 6-9: Maximum interval of 12 months
Ongoing compliance also requires airflow volume/velocity verification (annually), continuous pressure differential monitoring, and HEPA filter integrity testing (annually or after filter changes).
Common Industries Requiring Cleanrooms
Cleanroom requirements vary by industry, but several sectors depend on them as a non-negotiable part of production:
- Semiconductor manufacturing: Wafer fabrication requires ISO Class 3-5 for critical lithography and processing steps
- Pharmaceutical production: Aseptic filling requires ISO 5 (Grade A) critical zones with ISO 7 (Grade B) backgrounds; terminally sterilized products may use ISO 7 or 8
- Medical device manufacturing: Assembly operations typically require ISO Class 7 or 8, governed by ISO 13485 and FDA 21 CFR 820
- Biotechnology research: GMP laboratories and containment areas operate at ISO 6-8 depending on the process
- Aerospace component assembly: Precision assembly and inspection areas run at ISO 7-8, governed by ASTM E2352 and NASA standards
Key Differences: Controlled Environment vs Cleanroom
The core distinction comes down to what each facility is designed to control: one manages environmental conditions, the other eliminates contamination. Controlled environments prioritize temperature, humidity, and pressure stability — cleanrooms prioritize particle and microbial control above all else.
Core Technical Distinctions
| Parameter | Controlled Environment | Cleanroom |
|---|---|---|
| Filtration | MERV 13-16 (ASHRAE 52.2); captures ~85% of 1-3 µm particles | HEPA (99.97% of 0.3 µm) or ULPA (99.9995% of 0.12 µm) |
| Air Changes (ACH) | 2-20 ACH — sufficient for thermal and humidity control | 20-480+ ACH — required to dilute and remove particulate contamination |
| Pressure Control | Neutral or ±5 Pa differential for comfort or odor control | Strict positive pressure cascade (≥10-15 Pa per step) to block contamination ingress |
| Primary Goal | Process stability, product preservation, occupant comfort | Particulate and microbial contamination elimination |
Certification and Compliance Differences
These technical differences translate directly into how each environment is certified and regulated:
- Controlled environments verify temperature and humidity stability through sensor calibration and periodic data logging — typically validated annually or semi-annually under ASTM and ASHRAE guidelines, with moderate documentation requirements
- Cleanrooms must demonstrate compliance with particle count thresholds at multiple locations and particle sizes, with continuous monitoring, ongoing pressure differential checks, and semi-annual to annual recertification under ISO 14644-1, FDA, GMP, and USP frameworks
- Regulatory burden scales sharply with cleanroom classification — an ISO 5 facility requires far more frequent recertification and validation documentation than an ISO 8 space
Cost and Implementation Considerations
Compliance requirements drive cost — and nowhere is that more visible than in the gap between controlled environment and cleanroom budgets:
- Initial investment: Controlled environments require moderate HVAC capital costs with standard filtration. Cleanroom HVAC alone represents 25-50% of total project cost, driven by HEPA filtration, high ACH requirements, and precision controls.
- Operational costs: Cleanrooms can consume up to 15 times more energy than conventional commercial buildings, with over 50% of that load attributed to HVAC systems — largely due to the higher pressure drop across HEPA filters increasing fan energy demand.
- Maintenance: Controlled environments need quarterly-to-annual filter changes and annual sensor calibration. Cleanrooms require monthly-to-quarterly HEPA replacement, integrity testing, continuous monitoring, and specialized cleaning protocols.
- Certification expenses: Controlled environments carry moderate annual or semi-annual validation costs. Cleanrooms require specialized third-party testing every 6-12 months, with costs that scale by ISO classification.
| Feature | Controlled Environment | Cleanroom |
|---|---|---|
| Initial Investment | Moderate ($50-150/sq ft) | High ($150-1,000+/sq ft depending on ISO class) |
| Operational Costs | Standard energy consumption | 10-15x higher energy consumption |
| Maintenance | Quarterly filter changes, annual calibration | Monthly to quarterly HEPA replacement, continuous monitoring |
| Certification Frequency | Annual or semi-annual | Semi-annual (ISO 5) to annual (ISO 6-9) |

Cleanroom Classifications and Standards
Understanding ISO Cleanroom Classes
The ISO classification system defines cleanliness levels from ISO 1 (ultra-clean) to ISO 9 (basic clean), based on maximum allowable particle concentrations per cubic meter of air.
The four classes most relevant to regulated manufacturing are ISO 5 through ISO 8:
| ISO Class | Legacy Equivalent | Max Particles ≥0.5 µm/m³ | Typical Applications |
|---|---|---|---|
| ISO 5 | Class 100 | 3,520 | Aseptic pharmaceutical filling, semiconductor lithography, optical assembly |
| ISO 6 | — | 35,200 | Pharmaceutical preparation areas, precision medical device assembly |
| ISO 7 | Class 10,000 | 352,000 | General pharmaceutical manufacturing, medical device packaging, aerospace components |
| ISO 8 | Class 100,000 | 3,520,000 | Component handling, equipment washing, less critical manufacturing steps |
Industry-Specific Standards and Requirements
ISO 14644-1 sets the baseline, but most regulated industries layer additional requirements on top of the core particle classification.
- Aerospace (ASTM E2352): Requires molecular contamination controls and specific cleaning protocols for spacecraft and satellite components, beyond standard particle counts.
- Semiconductor facilities (SEMI F21): References ISO 14644-1 but adds airborne molecular contamination (AMC) controls, since trace chemical contaminants — not just particles — can affect chip yields.
- Pharmaceutical (EU GMP Annex 1 / FDA Guidance): Layers microbiological limits onto ISO particle classifications, requiring concurrent particle and microbial monitoring at all critical zones.
The Four Levels of Cleanliness
Pharmaceutical manufacturing takes ISO classes a step further. EU GMP Annex 1 defines four operational grades that incorporate real-world personnel activity — not just static particle counts — into the cleanliness requirements.
| Grade | ISO Equivalent | Conditions | Use Cases |
|---|---|---|---|
| A | ISO 5 | At rest and in operation | Aseptic filling, open ampoules; continuous monitoring required; microbial limit <1 CFU/m³ |
| B | ISO 5 (rest) / ISO 7 (operation) | Both states monitored | Background environment surrounding Grade A zones |
| C | ISO 7 (rest) / ISO 8 (operation) | Both states monitored | Solution preparation, component transfer, less critical manufacturing steps |
| D | ISO 8 (rest) | At rest only | Component handling, equipment washing, material staging |

Industry Applications and Use Cases
Industries Requiring Cleanrooms
- Semiconductor wafer fabrication (ISO 3–5): Lithography, etching, and deposition steps demand ultra-clean air. A single particle can cause chip failure.
- Pharmaceutical sterile manufacturing (ISO 5–7): Aseptic drug-fill operations require ISO 5 critical zones with ISO 7 backgrounds; terminally sterilized products may use ISO 7–8.
- Medical device assembly (ISO 7–8): Implants and surgical instruments need cleanroom assembly to prevent patient infections from particulate or microbial contamination.
- Biotechnology research (ISO 6–8): Cell culture and biopharmaceutical production require contamination-free conditions to protect both product integrity and researcher safety.
- Aerospace precision assembly (ISO 7–8): Guidance systems and optical instruments need particle-free environments — a single contaminant can compromise performance in space.
Industries Using Controlled Environments
- Cannabis cultivation: Temperature (22–28°C) and humidity (55–65%) must stay precise to prevent mold and preserve cannabinoid and terpene yields. Particle control is secondary to environmental stability.
- Food and beverage processing: Cold chain storage (4–8°C) and hygienic processing zones require temperature control and sanitation — not ISO particle classification.
- Cosmetics manufacturing: Formulation and filling areas need stable temperature and humidity for product consistency, but not cleanroom-level air cleanliness.
- Battery dry rooms: Lithium-ion manufacturing demands ultra-low humidity (<1% RH, –40°C dew point) to prevent electrolyte reactions. Moisture is the threat, not particles.
- Compounding pharmacies: USP <795> non-sterile preparations need controlled room temperature; sterile compounding under USP <797> steps up to ISO 7 buffer rooms with ISO 5 hoods.
- R&D laboratories: Testing and validation labs need stable, reproducible conditions — typically controlled environments rather than full ISO-classified cleanrooms.
Facilities Using Both Solutions
Many facilities run cleanrooms and controlled environments side by side, applying each where the process demands it.
| Facility Type | Cleanroom Zones | Controlled Environment Zones |
|---|---|---|
| Pharmaceutical | ISO 5–7 sterile production | Raw material storage, QC labs, stability chambers |
| Electronics | ISO 5–6 semiconductor assembly | Functional testing, burn-in, warehouse storage |
| Medical devices | Sterile assembly and packaging | Component storage, non-sterile manufacturing, QC |
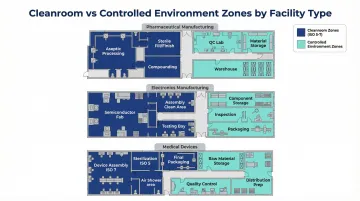
Designing both zones as a single integrated project — rather than treating them separately — reduces construction time and ensures the two environments work together without compliance gaps. ACH Engineering delivers turnkey cleanroom solutions that cover both sides of this equation.
Choosing the Right Solution for Your Facility
Assessment Criteria for Decision-Making
The decision framework centers on one critical question: Does your process involve contamination-sensitive operations that could compromise product quality, patient safety, or device functionality?
Choose a cleanroom if your application involves:
- Sterile pharmaceutical production or aseptic filling
- Medical device assembly or packaging
- Semiconductor wafer processing or microelectronics assembly
- Optical component manufacturing or precision optics
- Aerospace component assembly requiring particle-free conditions
- Biotechnology research with contamination-sensitive cell cultures
Choose a controlled environment if your focus is:
- Environmental stability without strict particle control
- Temperature and humidity regulation for product storage
- Agricultural cultivation requiring climate control
- Battery manufacturing requiring moisture control
- Non-sterile pharmaceutical compounding or quality control
- Food processing requiring sanitary conditions without ISO classification
Key questions to guide your decision:
- What regulatory standards apply to your industry and process?
- What level of particle sensitivity does your product or process have?
- What is your budget for initial capital investment and ongoing operations?
- Do you need ISO certification or just environmental parameter control?
- Will contamination directly impact product quality or safety?
Working with Cleanroom and Controlled Environment Specialists
Consulting with experienced engineering firms who understand both cleanroom and controlled environment design is essential for protecting your facility investment. The wrong choice can result in over-specification (wasting capital) or under-specification (risking product quality and regulatory compliance).
ACH Engineering offers assessment services that evaluate your regulatory obligations, operational constraints, and budget before recommending a solution — ensuring the facility design fits your actual process rather than a generic template. Their turnkey approach covers design, manufacturing, construction, and validation across both controlled environments and cleanrooms.
With project experience spanning pharmaceutical, biotechnology, aerospace, medical devices, semiconductors, and agricultural applications across Canada and the USA, ACH Engineering helps clients determine the right classification level and facility type from the outset.
Planning for Future Needs and Scalability
Proper initial planning can accommodate future expansion or classification upgrades without complete reconstruction, protecting your investment as business needs evolve.
Modular cleanroom and controlled environment solutions offer significant advantages for scalability:
- Components can be easily expanded, relocated, or reconfigured as production volumes change
- Additional features like airlocks, gowning rooms, or biological safety cabinets can be integrated later
- Classification can be upgraded (e.g., from ISO 8 to ISO 7) by increasing filtration coverage and air change rates
- Facilities can be repurposed for different products or processes without major reconstruction
Design considerations for future scalability:
- Oversized HVAC infrastructure to accommodate increased filtration loads
- Structural capacity for additional ceiling-mounted HEPA filters
- Electrical capacity for expanded monitoring and control systems
- Modular wall systems that can be reconfigured without demolition
- Space allocation for future expansion zones
Building scalability into the initial design is far more cost-effective than retrofitting later — and it keeps options open as production volumes, product lines, or regulatory requirements shift.
Frequently Asked Questions
What is the difference between a controlled environment and a cleanroom?
Controlled environments regulate temperature, humidity, and airflow for process stability and product preservation, using standard HVAC filtration (MERV 13-16). Cleanrooms add strict particle filtration through HEPA/ULPA systems to meet ISO 14644-1 classifications, with significantly higher air change rates (20-480+ ACH versus 2-20 ACH) and continuous particle monitoring.
What is the particle count for a clean room?
Particle counts vary by ISO classification. ISO 5 allows maximum 3,520 particles ≥0.5 µm per cubic meter, ISO 7 allows 352,000 particles of the same size, and ISO 8 permits 3,520,000 particles. These thresholds are measured using calibrated particle counters during certification testing.
What are the 4 levels of cleanliness?
The four practical cleanliness levels are:
- Grade A / ISO 5 — ultra-clean zones for aseptic filling
- Grade B / ISO 7 — background environment for sterile operations
- Grade C / ISO 8 — clean areas for solution preparation
- Grade D — basic clean zones for component handling
Each level carries specific particle limits and operational requirements.
What is the clean build protocol in controlled environment?
Clean build protocol covers construction procedures that minimize contamination during facility build-out. Key steps include selecting low-particle-generating materials, staged cleaning before equipment installation, controlled site access, and validation testing to confirm environmental parameters before operations begin.
Can a controlled environment be upgraded to a cleanroom later?
Yes, but the existing structure must support higher HVAC loads, ceiling-mounted HEPA filters, and proper pressure sealing. Modular construction makes this transition significantly more straightforward than traditional build methods.
How often do cleanrooms and controlled environments need recertification?
Controlled environments typically require annual or semi-annual validation of temperature and humidity parameters. Cleanrooms need particle count testing every 6 months for ISO 1–5 (the strictest classifications), or annually for ISO 6–9. Both require continuous monitoring of critical parameters with automated alarm systems.


