
Introduction
Regulatory agencies increasingly scrutinize wall surface conditions during facility inspections. FDA 483 observations have cited facilities for peeling paint, residues, and damaged wall surfaces that compromise cleanability and contamination control.
PVC wall panels have become the surface of choice in pharmaceutical facilities, biotech labs, hospital laboratories, and R&D cleanrooms. Their non-porous, seamless surfaces resist microbial growth and simplify cleaning protocols.
Yet lab-grade cleaning requires validated protocols, approved agents, and detailed documentation to satisfy FDA inspections, ISO 14644 classifications, and GMP audits. Surface maintenance isn't just about appearance—it's a regulatory requirement.
This guide covers:
- Step-by-step cleaning procedures for PVC panels in controlled environments
- Compliance considerations for FDA, ISO 14644, and GMP audits
- Cleaning frequency based on ISO classification levels
- Common pitfalls that trigger regulatory observations
TLDR
- Use 70% isopropyl alcohol or pH-neutral detergents with lint-free tools to maintain compliance and contamination control
- Clean daily for ISO 5-6 cleanrooms, twice weekly for ISO 7-8, and weekly for general labs
- Follow proper protocol: inspect first, clean top-to-bottom, allow 2-3 minute dwell time, rinse with deionized water, dry completely
- Avoid bleach >10%, acetone, MEK, abrasive scrubbers, and undocumented cleaning activities
- Proper cleaning maintains contamination control, extends panel lifespan 15+ years, and ensures audit readiness
How to Clean PVC Wall Panels in Lab Environments (Step-by-Step Protocol)
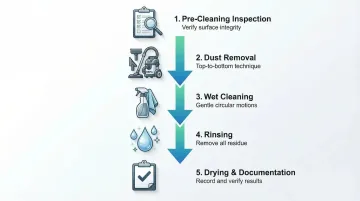
Step 1: Pre-Cleaning Preparation and Inspection
Before touching a single panel, conduct a thorough visual inspection for cracks, delamination, or damaged seams that could harbor contaminants. These defects compromise the entire controlled environment and must be documented and repaired before cleaning proceeds.
Clear the wall vicinity completely—move equipment, furniture, and materials to enable unobstructed access to all panel surfaces including corners and seams where contamination accumulates.
Verify cleaning agent approval:
- Cross-reference your lab's SOPs to confirm agents are approved
- Check MSDS sheets for PVC compatibility
- Ensure disinfectants are EPA-registered and validated for your facility
Don appropriate PPE as required by your lab's safety protocols: nitrile gloves, eye protection, and lab coats are standard minimum requirements.
Step 2: Dust and Debris Removal
Start with a HEPA-filtered vacuum with soft brush attachment or electrostatic dust cloth to remove loose particulates. Work systematically from top to bottom—gravity is your ally, pulling dislodged particles downward onto uncleaned surfaces rather than recontaminating clean areas.
Critical focus areas:
- Panel seams and joints
- Corner trim and transitions
- Wall-ceiling interfaces
- Areas behind relocated equipment
For ISO Class 5-7 cleanrooms, standard dusters are unacceptable. Apply only lint-free, cleanroom-grade polyester wipes that won't shed fibers and compromise particle counts.
Step 3: Wet Cleaning Application
Prepare your cleaning solution strictly according to lab SOP specifications. 70% isopropyl alcohol is the most common lab disinfectant, though pH-neutral detergents (pH 6-8) diluted per manufacturer specifications work for routine cleaning.
Application technique matters:
Follow this protocol to prevent contamination:
- Never spray directly onto panels—overspray penetrates seams and creates moisture problems
- Dampen lint-free microfiber cloth or cleanroom wipe with solution
- Clean in systematic overlapping strokes, top to bottom, left to right
- Maintain unidirectional wiping to prevent recontamination
Critical: Observe required contact time. Disinfectants require 2-3 minutes of wet contact time to achieve validated microbial kill. Surfaces must remain visibly wet for the entire duration. If the solution evaporates prematurely due to low humidity or high airflow, reapply immediately.
Step 4: Rinsing and Residue Removal
Rinse panels thoroughly with clean water and fresh lint-free cloth to remove all cleaning agent residue. The 2022 EU GMP Annex 1 explicitly mandates residue management—cleaning agents themselves become contaminants if not completely removed.
For critical environments (ISO 5-6): Use sterile or deionized water for final rinse. Tap water contains dissolved minerals (calcium, magnesium, silica) that leave deposits, harbor microbial growth, and contribute to particle counts.
Inspect for streaks, residue, or missed areas. Re-clean immediately rather than documenting a deficient cleaning cycle.
Step 5: Drying and Post-Cleaning Verification
Immediately dry panels with clean, dry lint-free cloth. Water spots create microenvironments for microbial growth and compromise cleanroom classifications.
Conduct post-cleaning visual inspection verifying panels are clean, dry, and free from damage, looking specifically for:
- Streaking or residue
- New cracks or surface damage
- Seal integrity at joints
- Discoloration indicating chemical incompatibility
Document everything. Record in your lab logbook: date, time, personnel, cleaning agents used (including lot numbers), concentrations, contact times, and any observations. This documentation is non-negotiable for GMP compliance and will be reviewed during audits.
When Should You Clean PVC Wall Panels in Lab Environments?
Cleaning frequency isn't arbitrary—it must be risk-based and aligned with your lab classification, processes conducted, and regulatory requirements.
Frequency by Cleanroom Classification
ISO 14644 classifications directly drive cleaning intensity:
| ISO Class | Cleaning Frequency | Rationale |
|---|---|---|
| ISO 5-6 (Grade A/B) | Daily or per shift | Critical aseptic areas require frequent cleaning to maintain sterility |
| ISO 7-8 (Grade C/D) | 2-3 times weekly | Lower risk areas; frequency depends on traffic and activity levels |
| General Labs | Weekly to bi-weekly | Non-classified spaces still require regular contamination control |

Event-Driven Cleaning Triggers
Scheduled cleaning is baseline—certain events demand immediate response:
- Spills or contamination incidents: Address within minutes and document the incident
- High-particulate processes: Clean after completion of activities generating unusual dust or debris
- Before critical operations: Sanitize surfaces before sterile compounding, aseptic processing, or sensitive analytical work
- After maintenance or construction: Terminal cleaning required after any activity breaching the cleanroom envelope
Operational Considerations
High-traffic areas and splash zones around sinks, autoclaves, or chemical handling stations require more frequent attention than low-touch walls.
Pharmaceutical and biotech facilities operating under FDA oversight must follow validated cleaning schedules with documented rationale for frequency decisions.
What You Need Before Cleaning PVC Wall Panels in Labs
Maintaining lab compliance and panel integrity requires approved materials and documented procedures. Starting with the right supplies prevents contamination, material damage, and regulatory issues.
Approved Cleaning Agents and Solutions
Lab-approved disinfectants:
- 70% isopropyl alcohol (IPA) or sterile 70% ethanol—industry standard for routine disinfection
- EPA-registered disinfectants specifically validated for PVC compatibility (quaternary ammonium compounds, hydrogen peroxide blends)
- pH-neutral detergents (pH 6-8) approved for cleanroom or lab use
Critical compatibility note: PVC is resistant to alcohols and hydrogen peroxide but vulnerable to ketones and aromatic solvents.
Never use acetone, MEK, xylene, or toluene—these cause immediate softening, swelling, and structural failure.
Bleach restrictions: Diluted bleach (≤10% sodium hypochlorite) can be used if approved in SOPs, but requires thorough rinsing.
Higher concentrations or prolonged contact yellow and degrade PVC surfaces.
Proper Cleaning Tools and Materials
For ISO Class 5-7 environments:
- Lint-free microfiber cloths or cleanroom-grade polyester wipes (never cotton or paper towels)
- Soft-bristle brushes (nylon or polyester) for textured panels or seams—never wire or abrasive materials
- HEPA-filtered vacuum with soft brush attachment
- Labelled spray bottles dedicated to specific cleaning solutions
Tool validation matters: Cleaning materials must be low-linting and non-shedding.
This prevents particle generation that defeats the purpose of cleaning.

Safety, Compliance, and Documentation Requirements
- Current lab SOPs and cleaning validation protocols accessible during cleaning
- Appropriate PPE per lab safety requirements and cleaning agent MSDS
- Cleaning logbook or electronic documentation system
- For GMP environments: training records confirming personnel are qualified to perform cleaning procedures
Key Parameters That Affect Cleaning Results in Lab Environments
Several critical variables impact cleaning effectiveness. Control these to achieve consistent, compliant results.
Cleaning Agent Concentration and Contact Time
Incorrect dilution reduces disinfection efficacy, while excessive concentration damages PVC surfaces. 70% IPA requires 30-60 second contact time for bactericidal/virucidal activity, but sporicidal activity demands 3 minutes.
Insufficient dwell time fails to eliminate contaminants, creating a false sense of security while contamination persists. Surfaces must remain visibly wet for the entire contact time.
Critical considerations:
- Low humidity and high airflow accelerate evaporation, potentially causing surfaces to dry before disinfection completes
- Monitor environmental conditions throughout cleaning
- Reapply solution as needed to maintain visible wetness for full contact time
Water Quality and Rinsing Technique
Tap water contains minerals that leave deposits, while inadequate rinsing leaves chemical residue that attracts particulates. EU GMP Annex 1 and WHO guidelines emphasize that final rinse water must be pharmaceutical grade—typically Purified Water (PW) or Water for Injection (WFI).
Proper rinsing technique delivers three critical benefits:
- Prevents streaking and maintains particle counts in ISO 5-7 areas
- Eliminates residues that could interfere with lab processes
- Removes chemical traces that harbor microbial growth
Always use deionized or sterile water for final rinse in classified cleanroom zones. Tap water introduces the very contaminants you're working to remove.
Cleaning Tool Selection and Technique
Abrasive materials scratch PVC, creating micro-crevices that harbor bacteria. Improper technique spreads contaminants rather than removing them.
Tool and technique requirements:
- Use lint-free, non-abrasive materials validated for cleanroom use
- Apply unidirectional wiping in overlapping strokes to prevent recontamination
- Clean from clean areas toward dirty areas, top to bottom
- Never reverse direction or use circular motions
Circular wiping patterns redistribute contaminants onto surfaces you've already cleaned, undermining the entire cleaning process.
Environmental Conditions During Cleaning
Temperature and humidity directly affect cleaning solution performance and drying time. Clean at controlled room temperature (68-72°F/20-22°C) for optimal results.
Environmental factors to monitor:
- High humidity extends drying time, increasing microbial growth risk
- Low humidity causes rapid evaporation that compromises disinfectant contact time
- Temperature fluctuations alter chemical reaction rates in cleaning agents
Documentation and Validation Compliance
Regulatory agencies require documented evidence that cleaning procedures are followed and effective. Complete cleaning logs immediately after each session—delayed documentation creates compliance gaps.
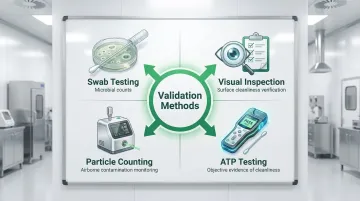
Validation methods include:
- Swab testing for microbial counts
- Visual inspection protocols
- Particle counting in classified areas
- ATP bioluminescence testing
These validation techniques confirm cleaning effectiveness and provide objective evidence for regulatory audits. For cleanroom facilities, ACH Engineering integrates documentation requirements into project specifications, ensuring your controlled environment meets both operational and compliance needs from day one.
Common Mistakes When Cleaning PVC Wall Panels in Labs
Even experienced facility teams make cleaning errors that compromise panel integrity and lab compliance. Avoiding these common mistakes protects your investment and maintains contamination control.
- Using incompatible agents like bleach >10%, acetone, or abrasives that damage surfaces and void warranties
- Failing to observe disinfectant contact time, resulting in inadequate microbial kill
- Inadequate rinsing that leaves residue attracting dust and creating streaks
- Neglecting documentation, creating audit failures and compliance gaps
- Using tap water for final rinse, introducing mineral deposits and contaminants
- Cleaning in circular motions that redistribute rather than remove contaminants
Correcting these habits ensures your PVC panels maintain their protective barrier without compromising your cleanroom classification.
Troubleshooting Common Cleaning Issues in Lab Environments
Even with validated procedures, you'll encounter cleaning challenges that require systematic troubleshooting. The three most common issues—streaking, persistent stains, and elevated particle counts—have identifiable causes and proven solutions.
Streaking or Residue After Cleaning
Likely causes: Cleaning solution too concentrated, inadequate rinsing, water with high mineral content, or dirty cleaning cloths redepositing contaminants.
What to adjust:
- Verify proper dilution ratios against SOP specifications
- Perform additional rinse cycle with deionized water
- Use fresh lint-free cloths for each section—never reuse contaminated cloths
- Check water quality. Switch to pharmaceutical-grade water if necessary
Persistent Stains or Discoloration
Likely causes: Chemical incompatibility causing surface damage, prolonged exposure to staining agents, or improper cleaning technique applying excessive pressure.
What to adjust:
- Identify stain source and discontinue incompatible chemical contact immediately
- Try pH-neutral specialty PVC cleaner with extended contact time and gentle agitation using soft-bristle brush
- For permanent damage, consult panel manufacturer or consider panel replacement. Damaged surfaces cannot be validated as cleanable
Increased Particle Counts Post-Cleaning (Cleanrooms)
Surface cleanliness issues are visible, but cleanroom performance problems require environmental monitoring to detect.
Likely causes: Cleaning tools shedding fibers, inadequate drying allowing microbial growth, contaminated cleaning solutions, or HVAC system malfunction.
What to adjust:
- Switch to certified cleanroom-grade wipes with documented low-linting performance
- Ensure complete drying. Use additional dry cloths if needed
- Prepare fresh cleaning solutions daily—never reuse solutions from previous cleaning cycles
- Verify HVAC system is operating properly during and after cleaning. Differential pressure and air changes per hour must be maintained
Alternatives to Standard Wet Cleaning Methods
Certain lab situations or panel configurations benefit from alternative cleaning approaches.
Steam Cleaning for Deep Sanitization
Best applications:
- Periodic deep cleaning in pharmaceutical manufacturing areas
- Post-contamination event sanitization
- Heavily soiled panels requiring aggressive intervention
Steam delivers superior microbial kill compared to standard wet cleaning. However, it requires specialized equipment, longer drying time, and careful validation.
High-quality PVC panels withstand temperatures up to 140°F (60°C), but steam exceeds 212°F (100°C). Use only if manufacturer validates high-temperature exposure.
Dry Cleaning Methods (HEPA Vacuuming + Dry Wiping)
Best applications:
- Quick maintenance between wet cleaning cycles
- Moisture-sensitive lab environments
- Lightly soiled panels requiring only particulate removal
Dry methods work faster with no drying time. The downside? They don't disinfect. Only loose particulates get removed—microbial contamination remains. Cannot substitute for validated wet cleaning in GMP environments.
Automated Cleaning Systems (Fogging/Misting)
Best applications:
- Large cleanroom facilities requiring whole-room disinfection
- Labs with complex equipment making manual cleaning difficult
- Terminal disinfection after contamination events
Automated systems provide excellent coverage and consistency. The investment is substantial—CAD $15,000-50,000+ for equipment—plus extensive validation requirements. These systems may not address localized heavy soiling effectively.
Vaporized hydrogen peroxide (VHP) is compatible with PVC and leaves minimal residue.
Conclusion
Cleaning PVC wall panels in lab environments requires more than wiping down surfaces. It demands approved agents, documented procedures, and strict compliance with regulatory standards.
Most cleaning failures stem from using incompatible chemicals that damage surfaces, inadequate contact time that fails to disinfect, poor rinsing technique that leaves contaminating residues, or missing documentation that creates audit vulnerabilities.
The right cleaning protocol balances contamination control, panel preservation, regulatory compliance, and operational efficiency. Following validated procedures with approved materials protects both your controlled environment's physical integrity and your facility's regulatory standing.
Facilities planning cleanroom installations can reduce maintenance complexity through proper system design. ACH Engineering provides GMP-compliant modular cleanroom solutions across North America, including seamless wall systems with cleanroom coving that eliminates hard-to-clean joints.
Frequently Asked Questions
How do you clean PVC wall panels?
Clean PVC panels using approved pH-neutral detergents or 70% isopropyl alcohol applied with lint-free cloths in overlapping strokes from top to bottom. Rinse thoroughly with deionized water, dry completely, and document all cleaning activities per lab SOPs.
How often should PVC wall panels be cleaned in different lab classifications?
ISO 5-6 cleanrooms require daily or per-shift cleaning, ISO 7-8 require 2-3 times weekly, and general labs need weekly to bi-weekly cleaning. Always add event-driven cleaning after spills, high-particulate activities, or before critical operations.
What cleaning agents should be avoided on PVC wall panels in labs?
Avoid bleach concentrations above 10%, acetone, MEK, harsh alkaline cleaners (pH >9), aromatic solvents (xylene, toluene), and any non-approved solvents. These agents cause surface degradation, yellowing, and structural failure that compromises panel integrity.
Can you use bleach to disinfect PVC wall panels in pharmaceutical labs?
Diluted bleach (≤10% sodium hypochlorite) can be used if approved in lab SOPs, but requires thorough rinsing with deionized water. Higher concentrations or prolonged contact yellow or degrade surfaces, and residual chlorine corrodes adjacent metal fixtures.
What documentation is required for PVC panel cleaning in GMP-regulated labs?
Document date, time, personnel, cleaning agents (with lot numbers), concentrations, contact times, and deviations in cleaning logs. Periodic validation through visual inspection, environmental monitoring, and residue testing provides objective evidence of effectiveness.
How do you remove stubborn stains from PVC wall panels without damaging the surface?
Try extended contact time (5-10 minutes) with approved pH-neutral cleaner and gentle agitation using a soft-bristle nylon brush. If ineffective, consult the panel manufacturer for specialty PVC cleaners. Avoid abrasive scrubbing, steel wool, or unapproved solvents.


