
Introduction
Most cleanroom projects don't fail during construction — they fail during planning. A missed requirement in week two becomes a costly redesign in month six, and by then, delays and budget overruns are almost inevitable.
Cleanroom project planning is the process of defining requirements, designing systems, and coordinating construction to create a controlled environment that meets specific cleanliness, regulatory, and operational standards. For project managers and facility planners in pharmaceutical, biotech, medical device, aerospace, and electronics industries, getting this process right is the difference between a compliant facility delivered on time and one that drags on with rework.
The numbers reflect how often the planning phase is underestimated: industry estimates suggest 70–80% of cleanroom builds experience schedule delays and 65–75% incur cost overruns, with late-stage design changes and coordination gaps as the leading causes. This guide walks through each phase of cleanroom project planning — what decisions need to be made, what factors drive success, and where projects most commonly go wrong.
TL;DR
- Cleanroom project planning covers needs assessment, design, regulatory strategy, procurement, and construction — each a distinct specialized phase
- Critical early decisions include ISO classification requirements, construction methodology (modular vs. stick-built), and realistic budget allocation with 15–20% contingency
- Early regulatory engagement and stakeholder alignment are the two factors most likely to determine whether a project stays on schedule
- Most project delays trace back to rushed requirements definition or commissioning timelines underestimated by 50% or more
- Pre-construction planning spans 3–6 months; modular builds can cut total project timelines by 40–60% vs. traditional construction
What Is Cleanroom Project Planning?
Cleanroom project planning is the systematic process of scoping, designing, and coordinating all the moving parts needed to deliver a compliant controlled environment. The result is a working project roadmap that covers:
- Design specifications and ISO classification targets
- Regulatory strategy (FDA, Health Canada, EMA, and similar bodies)
- Budget allocation and cost controls
- Timeline milestones and phasing
- Vendor and contractor selection
What sets cleanroom project planning apart from standard construction is what you're actually controlling. A conventional building manages load, weather, and code. A cleanroom manages invisible threats: airborne particles, microorganisms, temperature swings, humidity fluctuations, and pressure differentials between zones.
That requires specialized knowledge of HVAC design, filtration systems, material compatibility, and operational workflows — skills most general contractors simply don't have.
Why Proper Planning Is Critical for Cleanroom Projects
Poor planning carries a steep price in cleanroom projects. Industry analysis indicates that approximately 70–80% of cleanroom projects suffer from timeline delays, and 65–75% face cost overruns — primarily driven by unplanned design changes and coordination gaps between specialized trades. Common consequences include:
- Project delays averaging 6–12 months
- Budget overruns of 30–50%
- Regulatory compliance failures requiring expensive retrofits
- Operational inefficiencies that persist throughout the facility's lifecycle
Proper planning addresses critical success factors:
- Alignment between process requirements and facility design
- Regulatory readiness with documented contamination control strategies
- Realistic expectations for budget and timeline
- Risk mitigation through systematic identification and management
- Coordination among multiple specialized vendors (HVAC, controls, architectural, equipment suppliers)
Cleanroom project planning is a regulatory expectation, not just professional good practice. FDA regulations (21 CFR Part 211) and EU GMP Annex 1 explicitly require documented planning processes, design rationale, and risk assessments as part of facility qualification documentation.
Regulatory bodies also expect written procedures for sanitation, cleaning schedules, and maintenance to be established during the planning phase — before a single wall panel goes up.
Pre-Design Planning: Defining Requirements
Stakeholder Engagement and Needs Assessment
Skipping a structured needs assessment is the fastest way to design a facility your team outgrows before it's operational. Gather input from all stakeholders early to capture:
- Current and future processes
- Production volumes and growth projections
- Product characteristics and sensitivity
- Operational workflows and personnel movement
Classification Requirements Definition
Determine ISO cleanliness class based on product sensitivity and regulatory requirements. ISO 14644-1 defines classes based on particle concentration limits—for example, ISO 5 allows 3,520 particles/m³ at ≥0.5 µm.
Key decisions include:
- Critical zones vs. support areas — ISO 5 for aseptic filling, ISO 7-8 for background zones
- Environmental parameters — Temperature (18°C–24°C), humidity (30–60%), pressure cascades
- Airflow patterns — Unidirectional (laminar) for ISO 5, turbulent for ISO 7-8
- Documentation rationale — Written justification for each classification decision
Space Programming and Layout Planning
Calculate required square footage based on:
- Equipment footprint and process adjacencies
- Personnel flow and material flow patterns
- Future expansion capacity (plan for 20–30% growth)
- Utility access and infrastructure requirements
Regulatory Strategy Development
Identify applicable regulations and establish compliance pathways:
- FDA 21 CFR Part 211 for pharmaceutical manufacturing (US)
- Health Canada GMP (GUI-0029) for Canadian pharmaceutical facilities
- EU GMP Annex 1 for sterile product manufacture
- USP 797/800 for compounding pharmacies (US) or applicable provincial standards
- Timeline for regulatory submissions or notifications
Budget Framework Establishment
Develop preliminary cost estimates covering:
- Construction costs: $75–$1,500 USD per square foot depending on classification (US market reference; Canadian figures vary)
- Soft costs: Validation (1–5%), A/E fees (7.5–14.5%), CM fees (5–7.5%)
- Contingency allocation: 15–20% for unforeseen requirements
- Capital planning alignment: Synchronize cleanroom investment with existing budget approval cycles and organizational timelines
How the Cleanroom Project Planning Process Works
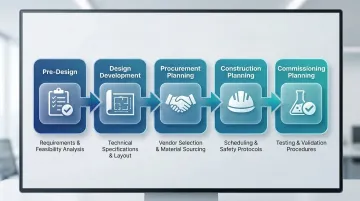
Cleanroom project planning follows five distinct phases. Each builds on the last, moving from high-level needs assessment through design, procurement, construction, and final commissioning:
- Pre-Design — needs assessment and requirements definition
- Design Development — architectural and engineering design
- Procurement Planning — vendor selection and equipment specification
- Construction Planning — methodology selection and scheduling
- Commissioning Planning — testing and qualification strategy
Process inputs:
- Business objectives and product requirements
- Regulatory mandates and budget constraints
- Timeline requirements and existing facility limitations
- Organizational capabilities and decision-making authority
Process controls:
- Stage-gate reviews where stakeholders approve each phase before proceeding
- Documented decision-making processes
- Risk registers that track and mitigate project risks
- Regular cross-functional team meetings
Process outputs: Initial conceptual requirements transform into detailed specifications, preliminary budgets become firm cost estimates, general timelines become detailed project schedules, and vendor options narrow to selected partners.
Here is how each phase unfolds in practice.
Phase 1: Requirements Definition and Feasibility (Weeks 1–4)
This phase involves:
- Conducting stakeholder interviews and documenting process requirements
- Establishing preliminary classification needs
- Assessing site constraints and utility availability
- Developing conceptual layouts
- Creating feasibility-level cost estimates (±30–40% accuracy)
Deliverable: Requirements specification document that serves as the foundation for all subsequent design work.
Phase 2: Design Development and Engineering (Weeks 5–16)
This phase translates requirements into detailed designs:
- Architectural layouts showing room configurations and traffic patterns
- Mechanical/HVAC designs specifying air handling systems and filtration
- Electrical designs for power distribution and controls
- Plumbing and process utilities for water, gases, and waste systems
- Construction methodology selection (modular vs. stick-built) based on timeline, budget, and flexibility requirements
Cost estimate accuracy improves to ±15% during detailed design.
Phase 3: Procurement and Vendor Selection (Weeks 12–20, overlaps with design)
The procurement phase includes:
- Issuing RFPs for specialized systems (HVAC, controls, architectural panels)
- Evaluating proposals based on technical capability, regulatory experience, and cost
- Conducting vendor audits for critical suppliers
- Negotiating contracts with clear scope definitions and performance guarantees
Phase 4: Construction Planning and Execution (Weeks 20–40+, project-dependent)
Construction phase planning involves:
- Finalizing construction sequencing and logistics
- Coordinating utility shutdowns and tie-ins
- Establishing quality control checkpoints
- Planning for ongoing operations (if building in occupied facility)
- Scheduling regulatory inspections
Modular cleanroom approaches can reduce this phase by 40–60% compared to traditional stick-built construction. ACH Engineering's turnkey cleanroom approach handles design, manufacturing, and on-site assembly as a single coordinated process — typically completing installation in weeks rather than the months required by conventional construction.
Key Factors That Affect Cleanroom Project Planning
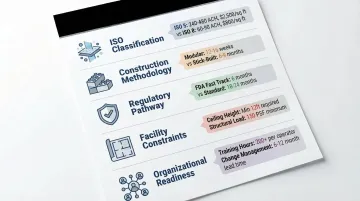
Five factors consistently shape cleanroom project scope, cost, and timeline. Understanding each one early prevents costly surprises during design and construction.
ISO classification: Your target cleanliness class drives everything from HVAC sizing to filter coverage. ISO Class 5 requires 300–480 air changes per hour with 60–70% ceiling filter coverage, versus just 30–60 ACH and 7–15% coverage for ISO Class 7. The cost gap is equally wide — ISO 5 builds can exceed $1,000 USD per square foot, while ISO 8 typically runs $75–$150 USD.
Construction methodology: Modular cleanrooms reach completion in weeks through parallel factory fabrication, while traditional stick-built projects run sequential trades over several months. Modular systems are easier to reconfigure later, but require thorough upfront planning for utility connections and structural support.
Regulatory pathway: Projects requiring pre-approval inspections need 3–6 months of regulatory review built into the schedule. Others can proceed straight to post-construction validation — a distinction that meaningfully shifts your project timeline.
Existing facility constraints: Ceiling heights, floor loading capacity (cleanroom HVAC systems can impose 21–30 lbs per square foot), and available utility infrastructure often dictate design decisions. Infrastructure gaps can trigger upgrade costs that reshape the budget entirely.
Organizational readiness: Internal resource availability, decision-making speed, and prior cleanroom experience all affect how quickly a project clears approval gates and stage reviews. Slow internal approvals are one of the most common — and avoidable — schedule risks.
Common Planning Mistakes and How to Avoid Them
Three mistakes derail cleanroom projects more than any others — and all three are avoidable with early attention.
Rushing requirements definition. Compressing planning to "save time" triggers design changes mid-build, which cost far more than the weeks saved upfront. Allocate 4–6 weeks for structured requirements gathering with all stakeholders. Capture not just current needs, but anticipated changes over the next 3–5 years.
Choosing construction methodology on price alone. Initial cost is one factor, not the deciding one. Evaluate options using a decision matrix that weighs construction speed, reconfiguration capability, accelerated depreciation benefits (where applicable), and full lifecycle costs. Modular systems often win on total cost of ownership even when the sticker price is higher.
Under-budgeting commissioning time. Most teams allocate 2–4 weeks for commissioning. Realistic timelines run 8–12 weeks for comprehensive testing, documentation, and regulatory readiness. Engage your validation team during the design phase — not after construction wraps — so commissioning requirements are built into the schedule from the start.
Frequently Asked Questions
What are cleanroom projects?
Cleanroom projects are construction or renovation initiatives to create controlled environments with specific levels of airborne particle control, typically required in industries like pharmaceuticals, biotechnology, medical devices, electronics, and food production where contamination must be minimized to ensure product quality and safety.
What is the purpose of a cleanroom?
Cleanrooms protect products, processes, or research from contamination by controlling airborne particles, microorganisms, temperature, humidity, and pressure. They're essential for manufacturing sterile products, handling sensitive materials, and conducting research where environmental variables must be tightly controlled.
How long does it take to plan and build a cleanroom?
Planning typically requires 3–6 months for requirements definition, design development, and procurement. Construction timelines vary from 3–4 months for modular systems to 8–12+ months for traditional stick-built cleanrooms, depending on size, complexity, and classification requirements.
What is the difference between modular and stick-built cleanrooms?
Modular cleanrooms use prefabricated panels assembled on-site, offering faster installation, easier reconfiguration, and lower total costs. Stick-built cleanrooms are constructed from scratch, providing more customization for complex layouts at the cost of longer build timelines.
What are the main phases of a cleanroom project?
The five main phases are:
- Requirements definition and feasibility
- Design development and engineering
- Procurement and vendor selection
- Construction and installation
- Commissioning and qualification (testing and validation)
How much does a cleanroom project cost?
Costs vary by size, classification, and complexity. Construction runs $150–$400+ per square foot, with ISO 5 facilities potentially exceeding $1,000 per square foot. Factor in validation (1–5% of construction cost) and A/E fees (7.5–14.5%) when building your budget.


