
Introduction
Cleanroom construction projects are among the most technically demanding and regulated building projects in modern industry. A single oversight in design specifications, airflow calculations, or material selection can cascade into certification failures costing $50,000-$200,000 and delays of 3-6 months—compromising product quality and regulatory compliance.
This guide addresses facility managers, pharmaceutical and biotech companies, semiconductor manufacturers, and anyone planning a cleanroom project from initial concept through final certification.
Whether you're building a sterile compounding pharmacy, a semiconductor fabrication facility, or a biotech research lab, the stakes are high. Regulatory agencies like Health Canada and the FDA expect precise environmental controls, and certification bodies require rigorous documentation proving your facility meets ISO 14644 standards.
We'll walk through the complete lifecycle—from initial requirements gathering and design, through construction and testing, to final certification—providing actionable insights to help you deliver a compliant, efficient cleanroom on schedule and budget.
TLDR
- Four critical phases: Design & Planning, Construction Execution, Testing & Validation, and Certification & Compliance
- ISO classification, construction method (modular vs. traditional), and regulatory requirements must be determined upfront
- Modular construction reduces project timelines by approximately 65% compared to traditional builds
- Budget CAD $200-$1,000+ per square foot based on ISO class and construction method
- Comprehensive documentation and validation throughout the process ensures certification success
Understanding Cleanroom Construction Requirements
Cleanroom construction creates a controlled environment where airborne particles, temperature, humidity, and pressure are regulated to specific standards.
Unlike conventional construction, every design decision—from wall materials to HVAC capacity—directly impacts your ability to achieve and maintain certification.
The ISO Classification System
The ISO 14644-1 standard defines air cleanliness by particle concentration limits. Your classification drives every subsequent design requirement:
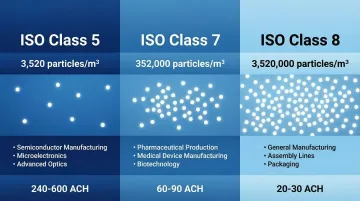
ISO Classification Comparison:
| ISO Class | Particles/m³ (≥0.5 µm) | Typical Application |
|---|---|---|
| ISO 5 | 3,520 | Aseptic filling, critical sterile operations |
| ISO 7 | 352,000 | Buffer zones, compounding rooms |
| ISO 8 | 3,520,000 | Non-sterile support areas, gowning rooms |
These classifications represent dramatic differences. ISO Class 5 environments allow only 3,520 particles per cubic meter, while ISO Class 8 permits up to 3,520,000—a thousand-fold difference that directly changes HVAC requirements, construction costs, and operational complexity.
Industry-Specific Regulatory Frameworks
Understanding your classification is the first step. Your industry then determines which regulatory framework governs your cleanroom design:
- Pharmaceutical Manufacturing (FDA cGMP): 21 CFR 211.42 requires separate or defined areas for aseptic processing with smooth, hard, easily cleanable surfaces
- Compounding Pharmacies (USP 797/800): ISO Class 5 primary engineering controls within ISO Class 7 buffer rooms; hazardous drugs require negative pressure containment
- European Operations (EU GMP Annex 1): Defines four grades (A-D), with Grade A critical zones equivalent to ISO 5 during operations
- Semiconductor/Electronics: Often requires ISO Class 4 or cleaner for advanced manufacturing nodes
How End-Use Application Drives Design
Pharmaceutical manufacturing demands different features than semiconductor production. A sterile filling line requires unidirectional airflow and personnel airlocks, while a battery dry room prioritizes ultra-low humidity control.
Early clarity on your application prevents costly redesigns later. Establish clear project goals upfront:
- Production capacity and throughput requirements
- Future expansion needs (plan for 5-10 year growth)
- Contamination control strategy specific to your product
- Budget constraints and timeline expectations
Phase 1: Design and Planning
Design decisions made in this phase determine 70-80% of your total project cost and timeline. Invest the time to get it right.
Conducting a Comprehensive Needs Assessment
Start with a thorough requirements definition:
- Cleanroom classification (ISO class based on product sensitivity)
- Size and layout (square footage, ceiling height requirements)
- Workflow patterns (material flow, personnel movement)
- Utility requirements (electrical capacity, process gases, waste handling)
Space Planning Principles
Effective cleanroom layouts follow contamination control logic:
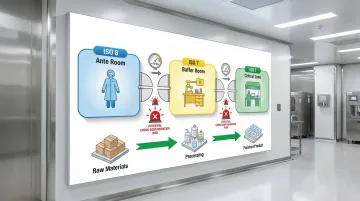
- Material Flow: Design unidirectional flow from raw materials through processing to finished product. Avoid cross-contamination by separating incoming and outgoing materials.
- Personnel Flow: Implement graded gowning sequences. Personnel should move from lower to higher classifications (ISO 8 ante room → ISO 7 buffer room → ISO 5 critical zone) through airlocks with interlocking doors.
- Equipment Placement: Position large equipment to maintain proper airflow patterns. Avoid creating dead zones where particles accumulate.
- Zone Segregation: Physically separate clean and dirty operations. Use pressure cascades to prevent contamination migration.
HVAC System Design: The Cleanroom Backbone
Your HVAC system is the single most critical—and expensive—component, typically accounting for 25-40% of total project cost.
Key design parameters include:
- Air Changes Per Hour (ACH): While ISO 14644 doesn't mandate specific ACH rates, FDA guidance suggests minimum 20 ACH for ISO 8 areas, with significantly higher rates (240-600+ ACH) for ISO 5 critical zones to maintain unidirectional airflow.
- Filtration Requirements: HEPA filters (99.99% efficient at 0.3 µm) are standard for ISO 5-8 environments. ULPA filters (99.9999% efficiency) may be required for semiconductor applications with smaller particle concerns.
- Pressure Cascades: Maintain positive pressure differentials of 10-15 Pascals (Pa) between adjacent rooms of different classifications to prevent contamination from entering. Continuous monitoring with documented alarms is essential.
- Temperature and Humidity Control: Specifications vary by industry. Pharmaceutical cleanrooms typically maintain 20-22°C and 30-50% RH, while battery dry rooms require <1% RH.

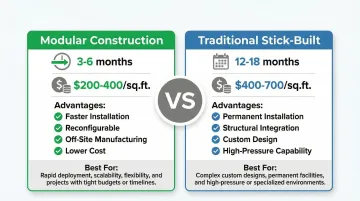
Modular vs. Traditional Construction Methods
This decision fundamentally impacts your timeline and budget:
Modular Cleanrooms:
- Timeline: 3-6 months total project duration
- Cost: $200-$400 CAD per sq. ft. for ISO 5-7 classifications (North American pricing varies by region)
- Advantages:
- Faster installation with reconfigurable layouts
- Lower upfront cost
- Off-site manufacturing reduces contamination risk
- Best for: Projects requiring speed, future flexibility, or phased expansion
Traditional Stick-Built:
- Timeline: 12-18 months total project duration
- Cost: $400-$700 CAD per sq. ft. for ISO 5-7 classifications (North American pricing varies by region)
- Advantages:
- Permanent installation with structural integration
- Better for high-pressure requirements
- Fully custom designs
- Best for: Long-term permanent facilities with stable requirements
Working with specialized cleanroom engineers during the design phase can identify cost-saving opportunities early. For example, ACH Engineering's design services help clients right-size HVAC systems and optimize room adjacencies before construction begins, avoiding expensive mid-project changes. Their modular approach allows for phased expansion as production scales.
Utility Infrastructure Planning
Integrate utilities early in the design process:
- Electrical Systems: Calculate load requirements for HVAC, process equipment, and lighting
- Process Piping: Plan routes for purified water, WFI, clean steam, and process gases
- Compressed Gases: Size distribution systems for nitrogen, compressed air, and specialty gases
- Waste Handling: Design separate systems for hazardous and non-hazardous waste
Phase 2: Construction Methods and Execution
Construction Timeline Overview
Typical phases and durations:
- Preconstruction Planning: 2-4 weeks (permits, site preparation, final drawings)
- Site Preparation: 1-2 weeks (demolition, base building modifications)
- Installation: 4-12 weeks (modular) or 24-40 weeks (traditional)
- Commissioning: 2-4 weeks (system startup and initial testing)
Modular Cleanroom Construction
Modular systems use prefabricated wall panels, ceiling grids, and integrated components manufactured off-site in controlled conditions. This approach shifts much of the construction work to factory settings, where quality control is easier to maintain.
Key advantages:
- Off-site manufacturing reduces on-site labor by 40-60%
- Controlled factory environment minimizes contamination during fabrication
- Parallel manufacturing and site preparation compress timelines
- Factory Acceptance Testing (FAT) catches issues before delivery
ACH Engineering's modular cleanroom solutions demonstrate this approach, offering flexibility and adaptability for future expansion while meeting stringent ISO standards.
The systems include customizable wall panel materials (PVC, FRP, stainless steel) suited to different industry requirements—from pharmaceutical facilities requiring chemical resistance to food processing operations needing washdown capability.
Traditional Cleanroom Construction
While modular construction offers speed and flexibility, some projects require traditional stick-built methods. These cleanrooms use drywall, masonry, or other conventional materials assembled entirely on-site.
Traditional construction makes sense in specific scenarios:
- Permanent installations with no anticipated changes
- High differential pressure requirements (>50 Pa)
- Complex architectural integration with existing structures
- Projects where initial cost is less critical than long-term durability
Traditional construction requires sequential trade coordination—framing, then electrical, then HVAC, then finishes. This extends project duration to 6-12 months for construction alone.
Critical Construction Considerations
Maintaining Cleanliness During Installation
Implement clean build protocols per ISO 14644-4. This includes controlling personnel access, protecting surfaces during construction, and cleaning thoroughly before final testing.
Trade Sequencing
Coordinate trades to prevent contamination:
- Install HVAC ductwork before wall finishes
- Protect installed filters until final startup
- Schedule final cleaning after all penetrations are sealed
Quality Control Checkpoints
Establish hold points for inspection:
- After framing: verify dimensions and penetrations
- Following HVAC rough-in: verify duct cleanliness and routing
- Before ceiling installation: inspect plenum for debris
- Upon completion: verify seal integrity before testing
Phase 3: Testing and Validation
Testing verifies that your constructed cleanroom meets design specifications and regulatory requirements before operational use. This phase involves systematic performance validation and documentation that proves compliance. Without proper testing, you risk operational failures and regulatory non-compliance.
Key Performance Tests
Critical tests include:
- Particle Count Testing: Using calibrated discrete particle counters, measure airborne particle concentrations at designated sampling locations determined by ISO 14644-1 based on room area
- Airflow Velocity and Visualization: FDA and EU Annex 1 require smoke studies to demonstrate unidirectional airflow and proper sweeping action over products in critical zones, with video documentation
- HEPA Filter Integrity Testing: Conduct aerosol challenge testing (introducing test particles to verify filtration) using PAO or DOP at installation to detect seal or media breaches
- Pressure Differential Verification: Measure and document pressure cascades between adjacent rooms, ensuring all differentials meet the minimum 10-15 Pa requirement with doors closed
- Temperature and Humidity Validation: Verify control systems maintain specified ranges under various load conditions over extended periods (typically 24-72 hours)
The Commissioning Process
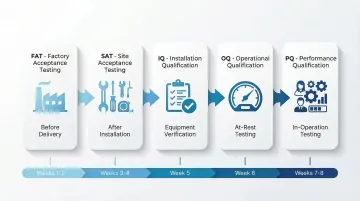
Once testing validates individual systems, commissioning integrates them into a complete operational environment. This follows a structured qualification sequence:
Factory Acceptance Testing (FAT): For modular systems, verifies airflow, filtration, and pressure performance before delivery, reducing on-site risk and accelerating installation
Site Acceptance Testing (SAT): Confirms installed systems meet specifications after installation but before operational use
Installation Qualification (IQ): Documents that equipment and systems are installed per design specifications, including verification of materials, dimensions, and utilities
Operational Qualification (OQ): Tests systems under "at-rest" conditions (no personnel or production) to verify airflow velocity, particle counts, pressure differentials, and filter integrity
Performance Qualification (PQ): Verifies compliance under "in-operation" conditions with personnel present and simulated production activities—the worst-case contamination scenario
Phase 4: Certification and Compliance
The Certification Process
After completing qualification testing, certification validates that your cleanroom meets applicable standards and confirms operational readiness.
Start by selecting an accredited third-party certification body with experience in your industry. These firms review documentation, witness testing, and issue formal certification reports.
Prepare your documentation package, which includes:
- Design specifications and as-built drawings
- Equipment specifications and cut sheets
- Validation protocols (IQ/OQ/PQ) with completed test results
- Standard Operating Procedures (SOPs) for operation and maintenance
- Training records for operations personnel
- Environmental monitoring plans
Schedule certification body visits only after successful OQ/PQ completion. Allow 2-4 weeks for report generation and any follow-up testing.
Industry-Specific Certification Requirements
Certification requirements vary by industry and regulatory jurisdiction. Understanding what inspectors will assess helps ensure first-time approval.
FDA Inspections (Pharmaceutical)
FDA inspectors assess physical facilities under 21 CFR 211.42, examining surface finishes, pressure monitoring systems, and documented environmental control data.
Have these records ready:
- Pressure differential logs
- Particle count trends
- Maintenance records
ISO 14644 Certification Audits
Independent auditors verify that particle concentrations meet your classification limits. ISO 14644-2 requires routine monitoring and periodic requalification—every 6 months for ISO 5 cleanrooms, annually for ISO 6-8.
State Board Inspections (Compounding Pharmacies)
State pharmacy boards verify USP 797/800 compliance, including proper pressure relationships, personnel gowning procedures, and cleaning validation.
Common Challenges and How to Avoid Them
Design Mistakes
Common design mistakes that derail cleanroom projects:
- Underestimating air changes: Insufficient ACH leads to particle count failures during testing. Work with HVAC engineers experienced in ISO requirements versus actual operational needs.
- Skipping expansion planning: Include 20-30% growth capacity in utility systems from the start. Modular designs simplify future additions, but traditional builds need oversized infrastructure.
- Ignoring workflow paths: Material and personnel flows that intersect create cross-contamination risks. Design one-way traffic patterns with physical separation between clean and dirty zones.

Construction Pitfalls
Avoid these construction phase mistakes:
- Rushing without complete plans: Starting construction with incomplete drawings generates change orders and delays. Invest 2-4 months in design development before breaking ground.
- Weak quality control: Contamination from construction can persist for months after completion. Enforce clean build protocols with controlled access, surface protection, and continuous cleaning schedules.
- Allowing debris accumulation: Construction dust in ductwork or plenums causes test failures. Seal all openings during installation and deep-clean systems before mounting final filters.
Certification Failures
Common reasons certification delays occur:
- Missing documentation: Gaps in test reports, unsigned protocols, or incomplete training records halt approval. Organize and maintain all documentation from project start.
- Premature system testing: HVAC systems require 24-72 hours of continuous operation to stabilize. Testing too early causes avoidable failures and expensive retests.
- Undertrained staff: Personnel need hands-on training in gowning, cleaning protocols, and monitoring procedures before certification audits. Schedule comprehensive training at least two weeks before testing.
Conclusion
Successful cleanroom construction from design to certification requires a systematic approach through four distinct phases, each with specific deliverables and quality checkpoints.
The journey from initial concept to operational facility typically spans 6-18 months, with timelines heavily influenced by your choice of construction method and the rigor of your planning process.
Early investment in proper design and planning pays dividends throughout the project lifecycle. Decisions about ISO classification, construction method, and HVAC design made in the first 60 days determine 70-80% of your total cost and schedule. Rigorous testing protocols and comprehensive documentation ensure faster certification and long-term operational efficiency.
Partnering with experienced cleanroom specialists who understand both the technical requirements and regulatory landscape specific to your industry is essential. Providers like ACH Engineering offer end-to-end project management and engineering services across North America, combining modular flexibility with turnkey execution to streamline the path from concept to certification. The right partner transforms complexity into clarity, ensuring your cleanroom meets both regulatory standards and operational needs from day one.
Frequently Asked Questions
What is a clean room in construction?
A cleanroom is a specially designed space where environmental parameters—airborne particles, temperature, humidity, and pressure—are controlled to ISO 14644 standards. These facilities use specialized materials and systems to minimize contamination introduction, generation, and retention.
What is a clean room contractor?
A cleanroom contractor specializes in designing, building, and validating controlled environments with expertise in ISO 14644 compliance, specialized HVAC systems, and contamination control. Unlike general contractors, they understand pressure cascade management and the IQ/OQ/PQ validation processes required for certification.
How much does it cost per square foot to build a cleanroom?
Costs range from $200-$1,000+ per square foot depending on ISO classification. ISO Class 5 runs $400-$800+/sq. ft., while ISO Class 8 costs $200-$500/sq. ft. Modular construction reduces costs by 15-30% compared to traditional methods.
How long does it take to design and build a cleanroom?
Total project duration ranges from 6-18 months: 2-4 months for design, 3-6 months for modular construction (6-12 months for traditional), plus 1-2 months for certification. Modular approaches can achieve operational status in as little as 6 months.
What are the most important factors to consider when selecting a cleanroom classification?
Consider process requirements (particle sensitivity), regulatory mandates, and operational costs (higher classifications require more air changes and energy). Avoid over-classifying—an ISO 7 room costs significantly less than ISO 5 to build and operate.
Can an existing building be converted into a cleanroom facility?
Yes, existing buildings can be converted using modular self-supporting systems. Key requirements include 10-12 feet ceiling height for HVAC plenums, structural capacity for rooftop equipment, and utility infrastructure supporting 20+ air changes per hour.


