
Introduction
Contamination failures cost manufacturers millions annually. Between 2020 and 2024, nearly 4,000 medical devices were recalled, while pharmaceutical recalls driven by contamination continue to rise. A single recall can cost between $10 million and $100 million, with catastrophic failures reaching $600 million in total impact.
For manufacturers in pharmaceuticals, semiconductors, medical devices, and other precision industries, cleanroom technology provides the contamination control that prevents product failures and regulatory violations. The challenge isn't knowing that cleanrooms matter — it's determining the right classification, design approach, and implementation strategy for your specific process.
This guide covers cleanroom fundamentals, ISO classification systems, design and construction approaches, and the practical factors that affect cost and compliance — so you can move forward with clarity.
TLDR
- Cleanroom technology controls airborne particulates through integrated filtration, airflow design, and operational protocols
- ISO 14644-1 defines nine cleanliness levels from ISO 1 (ultra-clean) to ISO 9 (standard air) based on particle counts
- Key industries include pharma sterile manufacturing (ISO 5), medical device assembly (ISO 7–8), and semiconductor fab (ISO 1–4)
- Implementation ranges from full facilities to localized mini-environments depending on contamination sensitivity
- Matching cleanroom class to product needs, HVAC design, and operational discipline determine project success
What Is Cleanroom Technology?
Cleanroom technology is an integrated system of engineering controls, specialized equipment, and operational protocols designed to maintain defined air cleanliness by controlling particulate contamination. Unlike standard HVAC systems or general "clean manufacturing" practices, cleanroom technology creates controlled, quantifiable, and validated contamination levels.
The system encompasses five core components:
- Physical cleanroom structure - Sealed walls, ceilings, and floors using non-shedding materials
- HVAC and filtration systems - HEPA or ULPA filters removing 99.97-99.9995% of particles
- Specialized materials - Smooth, sealed surfaces that prevent particle generation and accumulation
- Personnel protocols - Gowning procedures, movement restrictions, and training programs
- Monitoring systems - Continuous tracking of particle counts, pressure differentials, and environmental conditions
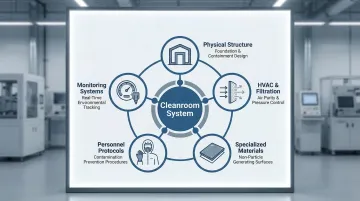
These components can scale to fit any operation. Manufacturers don't need a full-facility implementation — localized solutions such as mini-environments, laminar flow hoods, or modular cleanroom zones can deliver the same validated control at a fraction of the footprint.
What sets cleanroom technology apart is that contamination control is validated and measurable at every stage of the manufacturing process — not an assumption, but a documented standard.
Why Manufacturers Use Cleanroom Technology
Manufacturers implement cleanroom technology to meet three critical requirements: regulatory compliance, product quality protection, and contamination prevention.
Regulatory Compliance Requirements
Both FDA regulations (21 CFR 210/211) and Health Canada's Good Manufacturing Practices mandate equipment controls over air pressure, microorganisms, dust, humidity, and temperature for drug manufacture. The 2022 revision of EU GMP Annex 1 introduced even stricter particle limits for sterile manufacturing, capping Grade A environments at just 20 particles per cubic meter for sizes ≥ 5.0 µm — a benchmark that now influences North American facility design for exporters.
ISO 14644-1:2015 serves as the global baseline for particle classification across all industries requiring contamination control.
Product Quality Protection
Regulatory standards exist because contamination directly destroys product value. The failures are measurable:
- Semiconductor manufacturing - Single particles can destroy microcircuit functionality
- Medical device production - Contamination creates safety hazards and device malfunctions
- Pharmaceutical manufacturing - Particulates compromise sterility and drug efficacy
The pharmaceutical industry alone spends nearly $1 billion annually on energy for cleanroom operations — a cost manufacturers accept because a single contamination event can mean product recalls, regulatory action, or patient harm.
Three Types of Contamination Control
Cleanroom technology prevents distinct contamination categories:
- Particulate contamination - Dust, fibers, skin cells, and other solid particles
- Chemical contamination (AMC) - Airborne molecular contaminants including vapors and chemical substances
- Biological contamination - Bacteria, fungi, viruses, and other viable microorganisms
Understanding which contamination type poses the greatest risk to your process is the starting point for determining cleanroom classification, airflow design, and filtration requirements.
How Cleanroom Technology Works
Cleanroom technology operates through continuous air filtration, pressure control, and contamination source management to create and maintain controlled environments.
Fundamental Operating Principle
Outside air enters through pre-filters for temperature and humidity conditioning, then passes through HEPA (High-Efficiency Particulate Air) or ULPA (Ultra-Low Particulate Air) filters that remove 99.97–99.9995% of particles.
The filtered air circulates through the cleanroom, diluting and removing contaminants before returning through floor or wall grilles for reconditioning and re-filtration.
Airflow Design and Circulation
Two airflow strategies are used depending on the classification level required:
Laminar (unidirectional) flow moves filtered air in parallel streamlines at constant velocity (typically 0.36–0.54 m/s), sweeping particles downward or horizontally toward return filters. This approach prevents particle accumulation in critical zones and is essential for ISO 5/Grade A environments used in pharmaceutical sterile manufacturing.
Turbulent (non-unidirectional) flow uses high air change rates (20–60+ per hour) to dilute and remove contaminants through continuous circulation. It's the standard for ISO 6–8 environments where less stringent contamination control is acceptable.
Air Change Rates by Classification
| Cleanroom Class | Air Changes Per Hour | Typical Application |
|---|---|---|
| ISO 5 (Grade A) | 200-480+ ACH | Pharmaceutical sterile filling |
| ISO 6 | 70-160 ACH | Aseptic preparation areas |
| ISO 7 | 30-70 ACH | Medical device assembly |
| ISO 8 | 10-20 ACH | Component handling areas |
Pressure Control and Contamination Prevention
Pressure management determines whether a cleanroom protects its product, its personnel, or both.
| Pressure Type | How It Works | Primary Applications |
|---|---|---|
| Positive pressure | Higher internal pressure pushes air outward through gaps, blocking contaminant entry | Semiconductor, electronics manufacturing |
| Negative pressure | Lower internal pressure contains air inside, preventing hazardous material escape | Pharmaceutical compounding, biotech |
Beyond pressure, physical barriers reinforce contamination control at entry and exit points:
- Air locks and gowning rooms create procedural checkpoints before personnel enter critical zones
- Sticky mats capture particles from footwear before they can be tracked inside
- Pressure differentials of 10–15 Pa between adjacent cleanroom classes prevent contamination migration across zones
Where Cleanroom Technology Is Applied in Manufacturing
Cleanroom technology supports critical manufacturing operations across diverse industries where microscopic contamination threatens product quality, safety, or functionality.
Primary Manufacturing Applications
| Industry | Typical ISO Class | Key Requirement |
|---|---|---|
| Pharmaceutical Production | ISO 5–7 (Grade A–C) | Sterile injectables, IV solutions, compounding zones |
| Semiconductor Fabrication | ISO 1–4 | Wafer processing and photolithography; single particles destroy microcircuits |
| Medical Device Assembly | ISO 7–8 | Implantables, catheters, surgical instruments |
| Biotechnology | ISO 5–7 | Cell culture, gene therapy, biopharmaceutical production |
| Aerospace Components | ISO 7–8 | Contamination-free assembly for extreme operating conditions |
| Precision Optics | ISO 6–8 | Lens manufacturing and optical coatings; surface defects compromise performance |
Process-Specific Implementation
Manufacturers rarely need a cleanroom for an entire facility. Final assembly, sterile packaging, and quality testing require controlled environments — preparatory steps typically do not.
This targeted approach keeps costs in check while maintaining contamination control where it matters most. Modular cleanroom systems make this practical by enclosing only the specific process areas that demand it, without committing to full-facility construction.
Cleanroom Classification and Standards for Manufacturers
ISO 14644-1:2015 establishes the global standard for cleanroom classification, defining nine classes based on maximum allowable particle concentrations at specified particle sizes.
ISO 14644-1 Classification System
| ISO Class | Particles ≥0.5 µm per m³ | Particles ≥5.0 µm per m³ | Typical Application |
|---|---|---|---|
| ISO 1 | - | - | Advanced semiconductor lithography |
| ISO 2 | 4 | - | Semiconductor wafer processing |
| ISO 3 | 35 | - | High-precision semiconductor work |
| ISO 4 | 352 | - | Semiconductor fabrication |
| ISO 5 | 3,520 | 29 | Pharmaceutical sterile manufacturing |
| ISO 6 | 35,200 | 293 | Medical device assembly |
| ISO 7 | 352,000 | 2,930 | Medical device packaging |
| ISO 8 | 3,520,000 | 29,300 | Component preparation areas |
| ISO 9 | 35,200,000 | 293,000 | Standard room air (reference) |

EU GMP Grades for Pharmaceutical Manufacturing
The 2022 revision of EU GMP Annex 1 defines grades that map to ISO classes but include operational states and microbial limits:
| EU GMP Grade | Operational State | Particles ≥0.5 µm per m³ | ISO Equivalent | Typical Use |
|---|---|---|---|---|
| Grade A | At Rest / In Operation | 3,520 | ISO 5 | Critical zones (filling, aseptic connections) |
| Grade B | At Rest | 3,520 | ISO 5 | Background for Grade A |
| Grade B | In Operation | 352,000 | ISO 7 | Background for Grade A during production |
| Grade C | At Rest | 352,000 | ISO 7 | Less critical manufacturing stages |
| Grade D | At Rest | 3,520,000 | ISO 8 | Component handling after washing |
Matching Classification to Requirements
These standards don't exist in isolation — your facility must be built to the class your process and regulators actually require. Three factors drive that decision:
- Product contamination sensitivity - What particle size and concentration threatens product quality?
- Regulatory requirements - What standards apply to your industry and products?
- Contamination risk assessment - What are the actual contamination sources and pathways?
Over-specifying cleanroom requirements (choosing a cleaner class than necessary) dramatically increases capital and operational costs without improving product quality. Under-specifying creates compliance risks and product failures.
Each step up in ISO class can increase construction and operating costs substantially — getting the classification right from the start is one of the most consequential decisions in any cleanroom project.
Key Factors That Affect Cleanroom Technology Performance
Four factors determine cleanroom performance — and a weakness in any one of them can compromise the entire controlled environment.
Personnel Behavior and Gowning Protocols
Humans are the largest contamination source in cleanrooms. A person sitting motionless generates 100,000 particles (≥0.3 µm) per minute. Walking generates 10,000,000 particles per minute. Humans also shed skin scales carrying bacteria, and studies show that up to 99% of microorganisms in cleanrooms originate from personnel.
Controlling personnel contamination requires:
- Strict gowning procedures with full-body coveralls, hoods, gloves, and face masks
- Movement restrictions to minimize particle generation
- Continuous training on contamination prevention behaviors
- Regular monitoring of personnel compliance

HVAC System Design and Maintenance
Filter efficiency, air change rates, and pressure differentials directly impact cleanliness levels. System performance degrades without proper maintenance.
Key maintenance practices include:
- HEPA filter leak testing at installation and every 6-12 months
- Pressure drop monitoring across filters to detect loading
- Regular replacement of pre-filters to extend HEPA filter life
- Continuous monitoring of pressure differentials between zones (10-15 Pa minimum)
Material Selection and Surface Design
Every surface in a cleanroom is a potential contamination source — material selection directly affects how well the environment can be cleaned and maintained.
- Seamless wall-ceiling and wall-floor connections (coving)
- Non-porous, easily cleanable surfaces throughout
- Equipment with smooth profiles and minimal horizontal ledges where particles settle
- Materials that don't shed fibers or particles during use
Process Equipment and Activities
Machinery vibration, material handling, and production activities all introduce particles — equipment placement and enclosure design determine how well these sources are managed.
- Equipment enclosures to contain localized particle generation
- Positioning relative to airflow patterns to avoid disrupting clean zones
- Regular cleaning and maintenance schedules
- Process validation to verify contamination levels during actual production
Common Misconceptions About Cleanroom Technology
Cleanroom projects often stall — or get over-budgeted — because of assumptions that don't hold up under scrutiny. Here are four of the most common misconceptions, and what's actually true.
"Cleanroom Means Sterile"
Cleanrooms control particulate contamination but aren't necessarily free of microorganisms. ISO 14644-1 focuses exclusively on inert particles, not viable organisms. Sterile environments require additional controls including bioburden monitoring, sanitization protocols, and aseptic processing techniques.
Only cleanrooms specifically designed and validated as aseptic processing environments can claim sterility.
"Higher Cleanroom Class Is Always Better"
Choosing ISO 4 instead of ISO 7 doesn't improve product quality if contamination risks don't justify it. Cleanrooms are 30-50 times more energy-intensive than typical commercial buildings, with costs scaling exponentially as cleanliness requirements increase.
Over-specification wastes capital on construction, increases operational costs through higher energy consumption, and provides no measurable benefit to product quality or regulatory compliance.
"Cleanroom Technology Alone Guarantees Product Quality"
Physical infrastructure means nothing without operational discipline. Improper gowning, inadequate training, poor material handling, and lax process controls undermine even the most sophisticated cleanroom design.
The most common contamination failures traced back in post-incident reviews aren't equipment breakdowns — they're human process gaps.
"Cleanrooms Are Only for High-Tech Industries"
Cleanroom adoption has spread well beyond semiconductors and pharma. Industries now routinely applying controlled environment principles include:
- Food processing — aseptic packaging extends shelf life without relying on preservatives
- Cosmetics — preservative-free formulations require strict particulate and biological controls
- Agricultural cultivation — cannabis and indoor farming operations use cleanrooms to meet health regulations
- Medical devices — even non-implantable devices often require ISO 7 or ISO 8 environments

Any product sensitive to particulate, chemical, or biological contamination benefits from a controlled environment.
When Alternative Solutions May Be More Appropriate
Full cleanroom facilities aren't always the optimal solution. Here are three scenarios where alternatives make more sense:
Localized Contamination Control
When only specific process steps require contamination control, mini-environments, laminar flow workbenches, or isolators provide targeted protection at lower cost than room-scale cleanrooms. These localized solutions create ISO 5 conditions in small work zones while surrounding areas remain at ISO 7-8.
Best for:
- Small-scale production or research operations
- Processes where only final assembly requires clean conditions
- Facilities with limited space or budget constraints
Process Redesign or Improved Housekeeping
Some contamination issues stem from inadequate cleaning procedures, poor material handling, or process design flaws rather than ambient air quality. Enhanced housekeeping, improved workflows, or equipment modifications may address contamination without cleanroom investment.
Consider when:
- Contamination sources are identifiable and controllable
- Current practices fall short of basic GMP requirements
- Process changes can eliminate exposure to contaminants
Modular or Portable Cleanroom Solutions
Manufacturers with changing production needs, temporary requirements, or uncertain long-term demand benefit from flexible solutions. Unlike fixed construction, modular cleanrooms can be expanded, relocated, or reconfigured as production demands shift — a practical hedge against uncertain growth timelines.
This flexibility makes them well-suited for:
- Companies anticipating facility changes or expansion
- Contract manufacturers serving multiple clients
- Research organizations with evolving project requirements
The right choice depends on your contamination sources, production volume, and facility timeline. A formal risk assessment is the starting point — ACH Engineering works with manufacturers across North America to evaluate these factors and design the appropriate cleanroom solution, whether that's a full facility, a localized mini-environment, or a modular hybrid.
Frequently Asked Questions
What is cleanroom technology?
Cleanroom technology is a system of engineering controls and protocols that maintains specific air cleanliness levels by filtering airborne particles and controlling contamination sources. It integrates HVAC systems, specialized materials, gowning procedures, and monitoring systems to create validated, measurable contamination control.
What does a cleanroom technician do?
Cleanroom technicians maintain cleanroom operations through environmental monitoring, surface cleaning, equipment maintenance, proper gowning procedures, and condition documentation to support regulatory compliance.
What is the difference between a cleanroom and a laboratory?
Laboratories are workspaces for research and testing that may or may not require cleanroom conditions. Cleanrooms are specifically designed controlled environments with validated cleanliness levels based on ISO classifications. Some laboratories require cleanroom conditions for sensitive work, while others operate as standard lab spaces.
What ISO cleanroom class do I need for my manufacturing process?
Required cleanroom class depends on product contamination sensitivity and regulatory requirements. Pharmaceutical sterile manufacturing typically requires ISO 5, medical device assembly uses ISO 7-8, and semiconductor fabrication demands ISO 1-4.
How much does cleanroom technology cost to implement and maintain?
Costs range from tens of thousands for localized solutions like laminar flow hoods to several million for full facilities. ISO 8 cleanrooms cost considerably less than ISO 5 due to lower filtration and air change requirements. Ongoing costs — particularly energy, which runs 30-50x higher than standard buildings — should factor into any budget assessment.
Can I retrofit my existing facility with cleanroom technology?
Most existing facilities can accommodate cleanroom retrofits through modular wall systems, HVAC upgrades, and controlled zones. Feasibility depends on ceiling height, structural capacity, utility availability, and spatial configuration — with ceiling height and HVAC capacity typically being the most limiting factors.


