
Introduction
ISO cleanroom classification tables define particle concentration limits for controlled environments across pharmaceutical, biotech, medical device, semiconductor, and other critical manufacturing industries. According to the FDA's Guidance for Industry: Sterile Drug Products Produced by Aseptic Processing, ISO 14644-1 designations provide uniform particle concentration values for cleanrooms in multiple industries, making them essential for regulatory compliance worldwide.
Understanding how to read and interpret ISO 14644-1 classification tables is fundamental for facility planning, regulatory compliance, and ensuring product quality.
Many professionals struggle with selecting appropriate classifications, interpreting particle count limits, and understanding the practical implications of different ISO classes on design and operational costs.
This guide will help you confidently read ISO classification tables, select the right cleanroom class for your application, and understand how your choice impacts facility design and operational costs.
TL;DR
- Nine cleanroom classes (ISO 1-9) defined by maximum particle counts per cubic meter
- Particle limits range from 0.1µm to 5µm with stricter counts for lower classes
- ISO 5, 6, 7, and 8 replace legacy Federal Standard 209E classes (Class 100, 1,000, 10,000, 100,000)
- Lower ISO classes require higher air change rates and significantly increase HVAC costs
- Proper interpretation requires understanding "considered particle sizes" and occupancy states (at rest vs. operational)
Understanding ISO 14644-1 Classification Tables
ISO 14644-1 is the international standard that replaced Federal Standard 209E in 1999. The 2015 revision (Edition 2) introduced significant improvements to statistical accuracy and sampling methods, including a hypergeometric sampling model.
This model allows each sampling location to be treated independently with 95% confidence that at least 90% of the cleanroom area complies with limits.
The classification table (Table 1 in ISO 14644-1:2015) specifies maximum particle concentrations allowed per cubic meter of air for particles of specified sizes.
Cleanroom classification is based on actual particle concentration measurements, not design features, and must be verified through testing at specified sampling locations using light scattering airborne particle counters (LSAPC) compliant with ISO 21501-4.
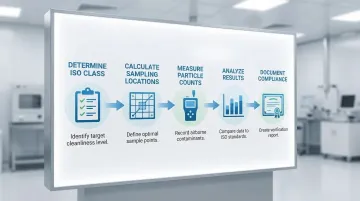
Considered Particle Sizes
Not all particle sizes are measured for every ISO class. The standard uses "considered particle sizes" - measuring only those particle sizes where concentrations are meaningful.
For ultra-clean environments like ISO 5, measuring particles ≥5µm isn't practical because concentrations are too low. Conversely, for ISO 7 environments, measuring particles ≥0.1µm isn't meaningful because concentrations are too high to measure accurately.
Occupancy States
Classification must be reported in one of three occupancy states:
- As-built: Complete facility with services functioning but no equipment, furniture, or personnel
- At rest: Fully equipped with systems operating but no personnel present
- Operational: Normal production with equipment running and specified personnel present
The difference between "at rest" and "operational" is critical. Operational conditions typically show higher particle counts due to personnel movement and process activities, which is why many pharmaceutical facilities must meet classification limits in both states.
ISO Cleanroom Classes: ISO 1 Through ISO 9
The ISO classification system ranks cleanroom cleanliness from ISO 1 (most stringent particle control) to ISO 9 (basic contamination control). Each classification defines maximum allowable particle concentrations at specific particle sizes, determining which environments suit different manufacturing processes.
ISO Classes 1-4: Ultra-Clean Environments
These ultra-clean classifications are typically used in semiconductor manufacturing and nanotechnology. Photolithography and deposition areas often require ISO Class 3 or 4, where even sub-micron particles can cause defects in microchip circuits and precision components.
ISO Class 5: Critical Pharmaceutical Manufacturing
Pharmaceutical sterile manufacturing transitions to ISO Class 5 (equivalent to Fed Std Class 100), where regulatory agencies like the FDA mandate strict particle control for critical aseptic zones. ISO Class 5 environments require:
- Unidirectional airflow with 35-70% ceiling coverage
- 240-360 air changes per hour
- Maximum 3,520 particles ≥0.5µm per cubic meter
- Maximum 832 particles ≥1.0µm per cubic meter
ISO Classes 6-7: Common Industrial Classifications
ISO Class 6 (Class 1,000) and ISO Class 7 (Class 10,000) represent the most common pharmaceutical and medical device manufacturing classifications.
Manufacturing facilities use ISO 7 for non-sterile compounding, device assembly, and supporting areas adjacent to ISO 5 critical zones.
Particle Limits:
| ISO Class | ≥0.5µm | ≥1.0µm | ≥5.0µm |
|---|---|---|---|
| ISO 6 | 35,200 | 8,320 | 293 |
| ISO 7 | 352,000 | 83,200 | 2,930 |
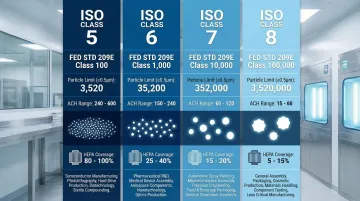
ISO Class 8: Less Critical Operations
ISO Class 8 (Class 100,000) suits less critical operations like packaging areas, ante-rooms, and secondary manufacturing zones. This classification requires only 10-25 air changes per hour with 5-15% ceiling coverage.
The reduced infrastructure requirements make ISO 8 significantly more economical than higher classifications, while still maintaining controlled contamination levels for non-sterile processes.
ISO Class 9: Minimal Control
ISO Class 9 provides minimal cleanroom control for storage areas, transition spaces, and material staging zones. Facilities use this classification when basic particulate control suffices—cleaner than conventional buildings but less demanding than production environments.
This entry-level classification offers the most cost-effective solution for supporting spaces that don't require stringent contamination control.
How to Read and Interpret Classification Tables
Classification tables organize data with rows representing ISO classes (1-9) and columns showing particle size thresholds (≥0.1µm, ≥0.2µm, ≥0.3µm, ≥0.5µm, ≥1µm, ≥5µm).
Understanding how to read these tables is essential for proper cleanroom verification and compliance.
Reading Specific Cells
Each cell value represents the maximum particle count per cubic meter. For ISO 7 at ≥0.5µm, the value 352,000 means no more than 352,000 particles of 0.5 microns or larger are permitted.
These limits are cumulative—the 0.5µm threshold includes all particles 0.5µm and larger, not just those at exactly 0.5µm.
Understanding Blank Cells
Some cells are intentionally blank for practical measurement reasons:
| Scenario | Example | Reason |
|---|---|---|
| Concentrations too high | ISO 7 at ≥0.1µm | Particle counts exceed reliable measurement range |
| Concentrations too low | ISO 5 at ≥5µm | Counts fall below accurate detection thresholds |
| Sampling losses | Large particles in clean classes | Particles >1µm may be lost in sampling systems |
The M Descriptor for Macroparticles
The 2015 revision introduced the "M descriptor" for macro-particles (≥5.0µm), expressed as "ISO M (concentration; particle size); measurement method." This is particularly relevant for pharmaceutical Grade A environments where 5µm particles were removed from the main ISO 5 classification but still need specification.
Example: ISO M (20; ≥5.0µm); LSAPC (Liquid-borne Particle Counter)
Step-by-Step Verification Example
To verify an ISO 7 cleanroom:
- Measure particles at ≥0.5µm (must be ≤352,000/m³)
- Measure particles at ≥1µm (must be ≤83,200/m³)
- Measure particles at ≥5µm (must be ≤2,930/m³)
- Sample at locations determined by room area per ISO 14644-1
- Verify 95% confidence level that 90% of area meets limits

Particle Size Categories and Their Significance
Particle sizes are measured in micrometres (µm or microns), where 1 micron equals 0.001 millimetres. For reference, human hair diameter is about 50-100 microns.
Different industries prioritize different particle size ranges based on their manufacturing requirements and contamination risks.
| Particle Size Range | Primary Industries | Critical Impact |
|---|---|---|
| 0.1-0.5µm | Semiconductors, Electronics | Circuit defects, precision component failures |
| 0.5-5µm | Pharmaceuticals, Medical Devices | Microbial contamination, sterility compromise |
| 5-10µm | Food & Beverage, Cosmetics | Visible product defects, quality issues |
Semiconductor and Electronics Manufacturing
Ultra-fine particles (0.1-0.5µm) pose the greatest risk in semiconductor fabrication because:
- Single particle can bridge microchip circuits and cause complete device failure
- Contamination during photolithography reduces production yields by 15-30%
- Precision components require monitoring down to 0.1µm to maintain tolerances
- Defects discovered late in production multiply costs exponentially
Pharmaceutical and Medical Device Manufacturing
Larger particles (0.5-5µm) are the primary concern because they can:
- Carry microorganisms and compromise sterility
- Cause product contamination
- Create visible defects in finished products
This is why pharmaceutical cleanrooms focus on the 0.5-5µm range rather than ultra-fine particles.
Converting Between ISO and Federal Standard 209E
Many specifications and facility documents still reference Federal Standard 209E classifications, even though the standard was officially canceled in November 2001. Understanding the conversion to ISO 14644-1 is essential when working with older documentation or clients using legacy terminology.
Conversion Reference
| Fed Std 209E | ISO Class | Particle Limit (≥0.5µm) |
|---|---|---|
| Class 1 | ISO 3 | 35/m³ |
| Class 10 | ISO 4 | 352/m³ |
| Class 100 | ISO 5 | 3,520/m³ |
| Class 1,000 | ISO 6 | 35,200/m³ |
| Class 10,000 | ISO 7 | 352,000/m³ |
| Class 100,000 | ISO 8 | 3,520,000/m³ |
Key Measurement Differences
The two standards use fundamentally different measurement approaches:
- Fed Std 209E: Measured particles per cubic foot at 0.5µm only
- ISO 14644-1: Measures particles per cubic meter across multiple size ranges (0.1µm, 0.2µm, 0.3µm, 0.5µm, 1.0µm, 5.0µm)
ISO's multi-size approach provides more comprehensive particle characterisation, but you'll need conversion factors when comparing legacy Fed Std specifications to current ISO requirements.

How ISO Classifications Impact Cleanroom Design
Classification selection directly impacts construction and operational costs, with HVAC systems representing the largest expense factor. Higher ISO classes require exponentially more air changes and filtration coverage, driving up both initial investment and ongoing energy consumption.
HVAC System Requirements
Higher classifications require more sophisticated systems:
- ISO 5: Unidirectional airflow, 240-360 air changes per hour, 35-70% HEPA ceiling coverage
- ISO 7: Mixed airflow, 30-60 air changes per hour, 15-25% ceiling coverage
- ISO 8: Mixed airflow, 10-25 air changes per hour, 5-15% ceiling coverage
According to a Lawrence Berkeley National Laboratory study, pharmaceutical cleanrooms can consume up to 15 times more energy than commercial buildings, with over 50% of electricity consumed by HVAC systems.
Cascade Approach
These intensive HVAC requirements make contamination control critical. Achieving ISO 5 typically requires passing through progressively cleaner anterooms to prevent contamination ingress.
A common pharmaceutical design uses:
- ISO 8 anteroom → ISO 7 buffer area → ISO 6 gowning → ISO 5 critical zone
FDA guidance recommends pressure differentials of at least 10-15 Pascals between adjacent rooms of differing classification.
Cost Implications
Moving from ISO 8 to ISO 5 increases construction and operational costs by approximately 3-5x due to significant increases in required air change rates and filtration coverage.
Cost multipliers by classification change:
- ISO 8 → ISO 7: 1.5-2x increase
- ISO 7 → ISO 6: 2-2.5x increase
- ISO 6 → ISO 5: 2.5-3x increase
Companies like ACH Engineering address this through modular construction approaches that reduce costs while maintaining regulatory compliance.
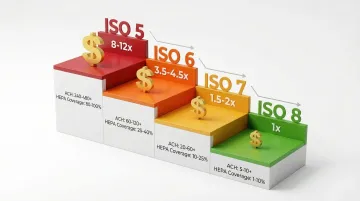
Common Mistakes When Selecting Classifications
Over-Specifying Classifications
Specifying ISO 5 when ISO 7 would suffice unnecessarily increases construction costs by 3-5x and operational energy consumption by similar multiples without improving product quality.
Proper risk assessment should determine the minimum classification that ensures product quality and regulatory compliance. Common over-specification scenarios include:
- Pharmaceutical compounding facilities selecting ISO 5 for non-sterile preparation areas
- Medical device manufacturers applying cleanroom standards to packaging areas that don't contact product
- Research labs specifying uniform ISO 6 throughout when only critical processes require that level
Another widespread issue stems from misinterpreting the classification values themselves.
Treating Limits as Targets
The values in classification tables are maximum limits, not target values. Cleanrooms should be designed with sufficient margin to consistently stay well below classification limits during normal operation.
Best practices for maintaining operational margin:
- Design for 30-50% below classification limits to account for equipment aging
- Build in buffer capacity for process variability and increased production volumes
- Plan for filter degradation between change-out cycles
- Allow headroom for temporary personnel increases during validation or training periods
For example, an ISO 7 cleanroom (≤352,000 particles/m³ at ≥0.5 µm) should target operational levels around 200,000-250,000 particles/m³ under normal conditions.
Conclusion
ISO 14644-1 classification tables provide the standardized framework for specifying and verifying cleanroom air cleanliness across all industries requiring controlled environments.
Understanding these tables goes beyond memorizing particle count limits. You need to grasp considered particle sizes, occupancy states, measurement methods, and the practical design implications of each classification level.
Proper classification selection balances regulatory requirements, product protection needs, and cost considerations. Moving from ISO 8 to ISO 5 can increase costs by 3-5x, making it essential to specify only the cleanliness level your processes actually require.
Implementing the right ISO classification requires expertise across design, construction, and validation. ACH Engineering provides turnkey controlled environment solutions across North America, from initial classification selection through commissioning and ongoing support.
Our modular cleanroom approach achieves required ISO classifications while managing costs through:
- Prefabricated components that reduce construction time
- Rapid installation that minimizes operational disruption
- Reconfigurable designs that adapt as your requirements evolve
Frequently Asked Questions
What are the cleanroom classifications?
ISO 14644-1 defines nine cleanroom classes (ISO 1 through ISO 9) based on maximum allowable airborne particle concentrations per cubic meter. ISO 1 is the cleanest with strictest limits, while ISO 9 is least stringent, verified by measuring particle counts at specified sizes.
Is ISO Class 5 or Class 7 better for cleanrooms?
"Better" depends entirely on your application requirements. ISO 5 is cleaner and required for sterile pharmaceutical manufacturing and critical semiconductor processes, but costs 3-5x more to build and operate than ISO 7. ISO 7 is sufficient for many medical device assembly operations and non-sterile pharmaceutical manufacturing at significantly lower cost.
How do legacy class numbers (Class 100, Class 10000) map to ISO cleanroom classifications?
Federal Standard 209E Class 100 equals ISO 5, Class 1,000 equals ISO 6, Class 10,000 equals ISO 7, and Class 100,000 equals ISO 8. The legacy standard measured particles per cubic foot at 0.5µm only, while ISO measures per cubic meter across multiple particle sizes.
What is the difference between "at rest" and "operational" cleanroom classifications?
"At rest" means the cleanroom is fully constructed with HVAC running but no personnel or production activity. "Operational" means normal production with personnel and equipment active. Operational conditions typically show higher particle counts due to human activity.
How do you determine which ISO class you need for your application?
Consult industry-specific regulations (FDA guidance for pharmaceuticals, ISO 13485 for medical devices), consider your product's sensitivity to contamination, and conduct risk assessments. The goal is to determine the minimum classification that ensures product quality and regulatory compliance without over-specifying and unnecessarily increasing costs.
What particle sizes are measured for each ISO classification?
Particle sizes depend on the ISO class. Cleaner classes (ISO 1-5) measure smaller particles (0.1-0.5µm), while less stringent classes (ISO 7-9) focus on larger particles (0.5-5µm). The ISO 14644-1 table specifies considered particle sizes for each class.


