
Introduction: The Critical Challenge of Particle Contamination in Controlled Environments
Particle contamination remains one of the most significant threats to product integrity across pharmaceuticals, biotechnology, electronics, and healthcare. Even microscopic particles measuring 0.1–5 microns can compromise sterile injectable products, damage sensitive semiconductor circuits, or invalidate critical research results. For manufacturers, the stakes are high: biopharmaceutical facilities experience batch failures attributed to contamination at a rate of 2.3%, with single batch losses often exceeding $1–2 million.
People are the biggest contamination risk in any controlled environment — personnel generate between 50,000 and 180,000 particles per minute (≥0.5 µm) just through normal activity. Traditional cleanroom construction methods, requiring 6–18 months to complete and substantial capital investment, compound the problem by delaying time-to-market and straining budgets.
Modular cleanroom solutions offer a strategic alternative: flexible, cost-effective contamination control that meets ISO 14644 standards while cutting deployment timelines by 50–70%. This guide covers how modular systems work, what particle classification means for your application, and the key design factors that determine contamination performance.
TLDR: Key Takeaways on Modular Cleanroom Solutions for Particle Contamination Management
- Modular cleanrooms install in 4-12 weeks versus 6-18 months for traditional construction, accelerating time-to-market
- HEPA filtration removes 99.97% of particles ≥0.3 microns, enabling ISO Class 5-8 compliance for regulated industries
- Initial investment runs 15-30% lower than stick-built cleanrooms, with further savings from accelerated tax depreciation schedules
- Prefabricated panels enable easy expansion, relocation, or reconfiguration without major facility disruption
- Integrated environmental controls maintain precise temperature, humidity, and pressure differentials for contamination prevention
Understanding Particle Contamination: Sources, Risks, and Classification Standards
Particle Contamination Sources in Manufacturing Environments
Contamination in controlled environments originates from four primary sources: personnel, processes, facilities, and the external environment. Personnel represent the dominant threat, with research quantifying human particle emission rates at approximately 50,000 to 180,000 particles per minute (≥0.5 µm) per person. Physical activity dramatically escalates this rate—walking or vigorous movement can push concentrations to the upper end of that range compared to sitting still.
Beyond personnel, process-related contamination stems from raw materials, equipment wear, and manufacturing activities. Facility infrastructure—building materials, HVAC systems, and structural components—sheds particles when poorly designed, while outdoor air infiltration adds a fourth vector.
Containment at the source matters: proper gowning protocols can reduce particle emissions by approximately 50%, with sterile garments made of synthetic materials achieving 10 to 30 times lower emission rates during movement compared to non-sterile alternatives.
Industry-Specific Particle Size Concerns
Different industries focus on different particle size ranges based on their product sensitivities:
Pharmaceutical Manufacturing: Focuses on 0.5-5 micron particles that can harbor microorganisms or cause injectable product failures. Even a single hair fiber can contaminate multiple batches—as evidenced by a 2025 FDA Warning Letter citing "extrinsic mammalian hair" contamination across nearly 40 batches, resulting in costly recalls.
Semiconductor/Electronics Manufacturing: Requires control of sub-0.1 micron particles that can cause circuit failures or yield losses. Modern chip features measure in nanometers, making even the smallest particles catastrophic to production.
Medical Device Assembly: Particle sensitivity varies by device type, with implantable devices requiring the most stringent controls to prevent patient harm and regulatory action.
ISO 14644 Classification Framework
ISO 14644-1:2015 defines cleanroom classifications based on maximum allowable particle concentrations per cubic meter of air. The system ranges from ISO Class 1 (the cleanest) to ISO Class 9 (the least stringent).
ISO 14644-1:2015 Maximum Allowable Particle Concentrations (particles/m³)
| ISO Class | ≥ 0.5 µm | ≥ 1.0 µm | ≥ 5.0 µm |
|---|---|---|---|
| ISO 5 | 3,520 | 832 | - |
| ISO 6 | 35,200 | 8,320 | 293 |
| ISO 7 | 352,000 | 83,200 | 2,930 |
| ISO 8 | 3,520,000 | 832,000 | 29,300 |
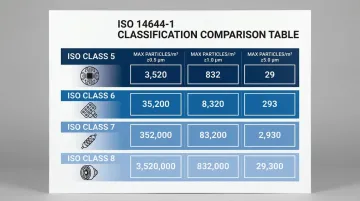
Selecting the right classification for your process is the first design decision—it determines airflow rates, filtration specifications, and overall construction cost. Typical industry targets include:
- Pharmaceutical sterile manufacturing: ISO Class 5-7 (Grade A/B/C environments)
- Medical device assembly: ISO Class 7-8 for most applications
- Semiconductor fabrication: ISO Class 3-5 for critical lithography processes
- Compounding pharmacies: ISO Class 5-7 per USP 797 requirements
The Hidden Costs of Inadequate Contamination Control
Financial exposure from contamination extends far beyond the immediate batch loss. For facilities with capacities over 1,000 liters, contamination is the leading cause of batch failures (2.3%), with direct costs frequently exceeding USD $1-2 million per incident.
Cost Components Include:
- Raw material losses and disposal costs
- Production downtime and schedule delays
- Regulatory penalties and inspection responses
- Product recalls and market withdrawals
- Damaged customer relationships and lost revenue
A facility losing USD $10,000 per day in production due to contamination-related downtime accumulates USD $3.65 million in annual opportunity cost. Accelerating facility deployment by six months through modular construction can recover USD $1.8 million of that lost value—making the case for purpose-built controlled environments a financial argument, not just a compliance one.
Modular Cleanroom Solutions: Architecture and Contamination Control Mechanisms
What Makes a Cleanroom "Modular"
Modular cleanrooms are prefabricated, self-supporting structures constructed from interlocking panels with integrated HVAC systems and reconfigurable layouts. Unlike stick-built cleanrooms requiring permanent construction, extensive HVAC retrofitting, and limited flexibility, modular systems arrive as prefabricated components manufactured offsite for rapid onsite assembly.
Three Common Modular Types:
- Softwall cleanrooms: Use vinyl curtains with aluminum frames, offering flexibility and cost-effectiveness but limited temperature/humidity precision
- Hardwall cleanrooms: Feature solid prefabricated panels (HPL, powder-coated steel, u-PVC) providing superior environmental control for strict classification requirements
- Hybrid (RigidWall) cleanrooms: Combine aluminum frames with clear acrylic panels, balancing structural integrity with visibility
ACH Engineering specializes in all three configurations, designing modular solutions that meet ISO Class 1-9 requirements while maintaining the flexibility to expand, relocate, or reconfigure as operational needs evolve.
HEPA and ULPA Filtration Systems
Filtration is what separates a cleanroom from a standard controlled space. HEPA (High-Efficiency Particulate Air) filters capture 99.97% of particles ≥0.3 µm, while ULPA (Ultra-Low Penetration Air) filters achieve 99.999% capture efficiency for particles ≥0.12 µm — tested to ISO 29463 and EN 1822 standards.
Filter Placement Strategies:
- Ceiling-mounted fan filter units (FFUs): Create unidirectional flow for ISO Class 5 environments
- Ducted systems: Provide mixed flow for less critical ISO Class 6-8 areas
- Proper sealing: Eliminates bypass leakage that compromises performance
Air Change Rates by Classification:
- ISO Class 8: Minimum 20 air changes per hour (ACH)
- ISO Class 7: Typically 40-60 ACH
- ISO Class 5: Requires 240-480 ACH through unidirectional airflow
ACH Engineering integrates HEPA and ULPA filtration across its modular cleanroom configurations, with FFU selection and placement sized to meet each project's specific ISO classification target.
Airflow Pattern Design for Particle Control
The right airflow strategy depends on ISO classification — and getting it wrong means particles accumulate rather than clear.
Unidirectional (laminar) airflow creates vertical or horizontal air curtains that sweep particles away from critical zones. ISO Class 5 environments require this approach, with air moving in parallel streamlines at 0.36-0.54 m/s. For less critical ISO Class 6-8 areas, turbulent (mixed) airflow is sufficient — air enters through ceiling diffusers and exits via low-wall returns, diluting rather than eliminating contamination.
Beyond the room itself, pressure cascading prevents cross-zone contamination. Maintaining positive pressure differentials of at least 10 Pascals between adjacent zones stops lower-grade air from migrating into higher-grade areas. Continuous monitoring with alarm systems catches pressure failures before they become compliance events.
Environmental Control Integration
Filtration and airflow remove particles — but environmental control keeps them from accumulating in the first place.
Humidity Control (30-50% RH):
- Prevents static buildup that attracts particles to surfaces
- Inhibits microbial growth below 60% RH
- Reduces electrostatic discharge in electronics manufacturing
Temperature Control (18-22°C/64-72°F):
- Ensures process consistency
- Maintains operator comfort, reducing perspiration and movement-related particle shedding
- Supports stable chemical reactions in manufacturing
Building Management Systems (BMS) tie these parameters together — monitoring and adjusting humidity, temperature, and pressure in real time while logging data for regulatory audits and operational troubleshooting.
Key Benefits of Modular Cleanrooms for Particle Contamination Management
Rapid Deployment and Reduced Construction Time
Modular cleanrooms install in 4-12 weeks compared to 6-18 months for traditional construction, cutting deployment time by 50-70%. Prefabrication occurs offsite while site preparation happens simultaneously, shortening overall project timelines.
This speed advantage translates directly to revenue. A pharmaceutical manufacturer needing to launch a new product can reach market months earlier, capturing sales that would otherwise go to competitors. For a facility losing $10,000 daily in potential production, a six-month acceleration represents $1.8 million in recovered opportunity cost.
Prefabrication also reduces on-site construction disruption, dust generation, and contamination risks during installation— benefits that matter especially to facilities maintaining active operations. Those same cost dynamics extend into the build itself.
Cost-Effectiveness and ROI Advantages
Modular cleanrooms typically cost 15-30% less in initial capital investment than equivalent stick-built facilities. But the financial advantages extend beyond upfront costs.
Operational Cost Savings:
- HVAC systems account for 50-70% of cleanroom operating costs
- Energy-efficient FFUs with variable frequency drives reduce consumption by 20-40% annually
- Reduced maintenance requirements due to modular component design
- Lower utility consumption through optimized airflow patterns

Tax Benefits: Modular cleanrooms often qualify as tangible personal property (equipment), allowing accelerated depreciation over 7 years versus 39 years for traditional construction. This improves near-term cash flow and net present value.
ROI Scenario: A $500,000 modular cleanroom investment avoiding a $2 million contamination incident achieves 4:1 ROI in a single event, not accounting for ongoing operational savings.
Flexibility and Scalability for Future Needs
Pharmaceutical product lines evolve, regulatory requirements change, and production capacity needs fluctuate. Modular panels and components can be reconfigured, expanded, or relocated without major facility disruption — an advantage permanent construction simply can't match.
Two scenarios illustrate the practical value:
- Scaling up: A biotech company experiencing 40% demand growth can add modular panels in under two weeks, maintaining production continuity. Traditional construction would require months of planning, permitting, and building.
- Relocating: Companies leasing space or consolidating operations can dismantle and move entire modular cleanrooms, preserving their investment rather than writing off permanent infrastructure.
ACH Engineering's Turnkey Approach
ACH Engineering delivers comprehensive modular cleanroom solutions that address particle contamination from initial design through final commissioning. Each project covers fast installation, reduced capital costs, and designs built to ISO 14644 and cGMP standards.
Operating across North America from Ontario to Alberta, ACH provides end-to-end project management ensuring proper airflow validation, particle count testing, and regulatory documentation. Their modular systems support:
- ISO Class 1-9 cleanroom classifications
- FDA, cGMP, USP 797/800, and NAPRA compliance
- Industries including pharmaceuticals, biotechnology, medical devices, semiconductors, and compounding pharmacies
Superior Material Selection and Surface Integrity
Modular panels use non-porous, smooth materials that don't shed particles. ACH Engineering offers three primary panel types:
| Panel Type | Key Properties | Best For |
|---|---|---|
| Powder-Coated Steel | Galvanized core, corrosion-resistant coating | High-durability applications |
| High Pressure Laminate (HPL) | Chemical-resistant, withstands repeated cleaning | Pharmaceutical and biotech environments |
| u-PVC | Affordable, customizable, lightweight | General ISO-classified cleanrooms |
All materials feature seamless panel joints and coved corners that eliminate particle traps and facilitate thorough cleaning. These design elements comply with cGMP requirements mandating smooth, continuous surfaces in pharmaceutical manufacturing.
Critical Design Elements for Effective Particle Contamination Control
Gowning and Personnel Flow Management
Proper gowning removes particles before cleanroom entry, dramatically reducing contamination risk.
Gowning Room Design:
- Airlocks with interlocked doors prevent simultaneous opening, maintaining pressure cascades
- Sticky mats capture particles from shoe soles
- Air showers use high-velocity HEPA-filtered air to dislodge particles from garments
- Unidirectional flow (clean-to-dirty progression) prevents cross-contamination
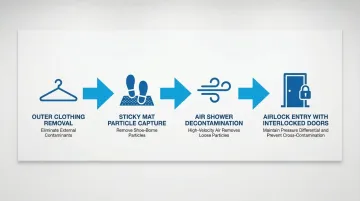
For high-grade areas (Grade A/B), interlocking door systems are required to prevent both doors from being open simultaneously. Outdoor clothing should not enter changing rooms leading to Grade B and C areas.
Material and Equipment Transfer Protocols
Pass-through chambers with interlocked doors and HEPA filtration enable material introduction without contamination. Advanced "active" pass-throughs use HEPA filtration purges to clean chamber air, reaching ISO Class 7 conditions in roughly 45 seconds before the receiving door opens.
Before transfer, personnel should follow consistent surface decontamination steps:
- Wipe all materials with appropriate disinfectants
- Remove outer packaging in ante-rooms before entry
- Stage equipment in decontamination areas prior to cleanroom introduction
Monitoring and Validation Systems
Continuous particle counting systems in critical zones (ISO 5/Grade A) alert operators when counts exceed classification limits, enabling immediate corrective action before contamination impacts products.
Certification Testing Requirements:
- ISO Class 5 (Grade A/B): Requalification every 6 months
- ISO Class 7/8 (Grade C/D): Requalification annually
- Tests include filter integrity, airflow visualization, particle count verification, and pressure differential confirmation
ACH Engineering handles validation as part of its turnkey delivery process — commissioning completed projects and confirming certification compliance before handover.
Industry Applications: Tailoring Modular Solutions to Specific Contamination Challenges
Contamination requirements vary significantly by industry—what qualifies as a controlled environment in one sector may fall short in another. The following breakdown covers three primary application areas where modular cleanrooms solve distinct regulatory and operational challenges.
Pharmaceutical and Biotechnology Manufacturing
Pharmaceutical manufacturing demands the most stringent contamination controls, driven directly by patient safety requirements.
Classification Requirements:
- Critical Zones (Grade A/ISO 5): Aseptic filling and open product handling require 3,520 particles/m³ (≥0.5 µm) maximum
- Background Areas (Grade B/ISO 5-7): Surround Grade A zones during aseptic operations, typically maintained at ISO 5 at-rest and ISO 7 in-operation
- Support Areas (Grade C/D): ISO Class 7-8 for solution preparation and less critical stages
FDA Guidance for Industry (2004) and EU GMP Annex 1 (2022) establish these requirements. ACH Engineering's modular cleanrooms support cGMP compliance and FDA inspection readiness through proper design, material selection, and validation protocols.
Medical Device and Electronics Assembly
These two industries share a cleanroom environment but have distinct classification needs:
- Medical devices: ISO Class 7-8 for most assembly and packaging; stricter for sterile or implantable devices. Manufacturers must comply with ISO 13485 and FDA 21 CFR Part 820.
- Semiconductor fabrication: ISO Class 3-5 for critical lithography and wafer etching, with tight controls over electrostatic discharge (ESD) and airborne molecular contamination (AMC) per SEMI standards.
For semiconductor and electronics clients, ACH Engineering designs ESD-safe cleanrooms with specialized materials and environmental controls that prevent static buildup throughout the facility.
Healthcare Facilities and Compounding Pharmacies
Hospital compounding pharmacies must comply with USP 797 (sterile compounding) and USP 800 (hazardous drug handling) requirements. These standards mandate specific ISO classifications for different compounding activities.
Modular cleanrooms provide compliant environments in existing hospital or pharmacy spaces without extensive renovation. Solutions meeting USP 797/800 and NAPRA standards incorporate pass-through chambers, specialized ventilation for hazardous drugs, and proper material flow design — all designed by ACH Engineering to fit within existing facility footprints.
The modular approach enables healthcare facilities to implement compounding pharmacy cleanrooms quickly, without disrupting active patient care.
Implementation Considerations: From Planning to Commissioning
Initial Assessment and ISO Classification Selection
Successful cleanroom implementation begins with a contamination risk assessment based on product sensitivity, process requirements, and regulatory standards.
Assessment Questions:
- What particle sizes pose the greatest risk to your products?
- What regulatory standards apply to your industry and products?
- What processes will occur in the cleanroom (open product handling, closed processes)?
- What is your anticipated production volume and growth trajectory?
Working with cleanroom specialists like ACH Engineering ensures appropriate ISO class selection and airflow configuration based on these factors.
Installation and Integration with Existing Facilities
Modular systems can integrate with existing utilities and minimize facility downtime — but the site must meet baseline requirements before installation begins:
Site Preparation Requirements:
- Adequate ceiling height for HEPA filter installations
- Sufficient HVAC capacity or provision for supplemental units
- Appropriate electrical infrastructure for environmental controls
- Floor loading capacity for equipment and personnel
ACH Engineering's design services assess these requirements during the planning phase, ensuring proper site preparation before component delivery. Their prefabricated approach allows much of the manufacturing to occur offsite while site preparation proceeds, compressing overall timelines.
Validation, Testing, and Ongoing Maintenance
ISO 14644-3 commissioning protocols verify that cleanrooms meet both design specifications and regulatory requirements:
Key Commissioning Tests:
- Filter integrity testing confirms HEPA/ULPA filters are properly sealed without leaks
- Airflow visualization (smoke studies) demonstrates proper particle sweep patterns
- Particle count verification confirms ISO classification compliance in as-built, at-rest, and operational states
- Recovery testing measures time to return to specification after contamination events (typically 15-20 minutes)

Once commissioned, maintaining classification requires consistent monitoring and scheduled upkeep.
Ongoing Maintenance:
- Regular filter monitoring and replacement based on performance data (pressure drop, leak testing) rather than fixed schedules
- Surface cleaning protocols following validated procedures
- Continuous monitoring of pressure differentials, temperature, and humidity
- Periodic recertification every 6-12 months depending on classification
Validation documentation — covering each commissioning test, baseline readings, and corrective actions — forms the compliance record that regulatory bodies expect during audits.
Frequently Asked Questions
What ISO cleanroom class do I need for pharmaceutical manufacturing?
Sterile injectable manufacturing typically requires ISO Class 5-7 (Grade A/B/C), while oral solid dose manufacturing may only need ISO Class 7-8. Requirements vary by product type — FDA Guidance for Industry and EU GMP Annex 1 are the primary references, and an experienced cleanroom engineer can help translate those standards into a compliant facility design.
How much does a modular cleanroom cost compared to traditional construction?
Modular cleanrooms typically cost 15-30% less in initial investment, with additional savings in installation time and energy efficiency. Final costs depend on size, ISO class, and features. Faster capital cost recovery on modular builds also improves overall ROI compared to conventional construction.
How long does it take to install a modular cleanroom?
Typical installation ranges from 4-12 weeks depending on size and complexity, compared to 6-18 months for traditional construction. Prefabrication occurs offsite while site preparation proceeds, compressing overall timelines by 50-70%.
Can modular cleanrooms be expanded or reconfigured later?
Yes. Modular panels and components are designed for easy reconfiguration, expansion, or relocation without major facility disruption. This makes them well-suited to evolving production volumes, regulatory updates, or site changes.
What maintenance is required to maintain particle contamination control?
Three core tasks keep contamination control intact: filter replacement (typically every 2-5 years based on performance monitoring), surface cleaning following validated protocols, and certification testing every 6-12 months. Frequency varies by ISO classification.
Do modular cleanrooms meet FDA and ISO regulatory requirements?
Yes. Properly designed and validated modular cleanrooms fully comply with FDA cGMP, ISO 14644, and USP standards when built to specification. Compliance depends on design quality, materials, and validation — all areas where working with a qualified cleanroom engineer makes the difference.


