
Introduction
In April 2025, the FDA issued a warning letter to a compounding pharmacy for failing to perform adequate environmental monitoring in ISO 5 aseptic zones—a stark reminder that even established facilities can fall short on contamination control. Separately, scientists analyzing NASA's Phoenix Mars Lander cleanroom discovered 26 previously unknown bacterial species that survived rigorous decontamination protocols, demonstrating that contamination challenges persist even in the most controlled environments.
Despite decades of technological advancement and stricter protocols, cleanroom contamination remains a persistent challenge across pharmaceutical, biotech, semiconductor, and aerospace industries. Human personnel shed millions of particles per minute, nanoscale contaminants threaten semiconductor yields at sub-3nm nodes, and extremophile microorganisms evolve resistance to standard cleaning agents.
This article covers:
- Recent contamination incidents and industry news from 2024–2025
- Top contamination sources and what makes them difficult to control
- Emerging challenges, including cross-contamination in multi-product facilities
- Best practices for prevention, monitoring, and contamination-resistant cleanroom design
TLDR: Key Takeaways on Cleanroom Contamination
- NASA identified 26 extremophile bacteria species in spacecraft cleanrooms that resist standard decontamination — a direct threat to pharma and biotech facilities
- Personnel contribute 75-80% of cleanroom contamination through particle shedding, with walking generating 10x more particles than sitting
- Material receiving and pre-cleanroom preparation are now recognized as critical contamination control points
- Semiconductor manufacturing faces new threats from nanoscale particles (3-4nm) and molecular contaminants that traditional counters miss
- Effective contamination control spans facility design, real-time monitoring, automation, and ongoing personnel training
Recent Cleanroom Contamination Incidents and Industry News (2024-2025)
NASA Extremophile Discovery Challenges Standard Protocols
In 2025, researchers published findings in Microbiome revealing 26 previously unknown bacterial species in NASA's Phoenix Mars Lander cleanroom. These extremotolerant organisms possess genes for DNA repair, spore formation, and biofilm production — allowing them to survive hydrogen peroxide vapor and UV radiation cycles.
The implications extend well beyond aerospace. If extremophiles can persist in one of the world's most rigorously controlled environments, pharmaceutical and biotech facilities face comparable risks from resistant microorganisms that standard cleaning validation may not detect.
FDA Enforcement Intensifies for Environmental Monitoring Failures
The FDA issued multiple warning letters in 2024-2025 targeting contamination control deficiencies:
- Empower Clinic Services (April 2025): Cited for inadequate environmental monitoring in ISO 5 zones, specifically failing to sample within the critical aseptic processing area during certification
- Optikem International (June 2024): Flagged for facility design failures where aseptic lines were open to room environment without barrier protection, and action levels permitted microbial growth in ISO 5 areas
- Catalent Indiana (November 2025): Received citation for mammalian hair contamination in sterile products, with inadequate root cause investigation beyond supplier assessment
These actions reflect a broader regulatory shift: the FDA has aligned its enforcement expectations with Contamination Control Strategy (CCS) principles established in the revised EU GMP Annex 1, which now function as a global benchmark across sterile manufacturing.
Semiconductor Industry Confronts Sub-3nm Contamination Threats
As chip geometries shrink below 3nm, particles as small as 3-4nm are now considered "killer" defects. Traditional laser particle counters — which detect down to 20nm — can no longer keep pace.
The industry now faces two distinct detection challenges:
- Nanoscale solid particles below the threshold of conventional optical systems
- Airborne molecular contaminants (AMCs) at parts-per-trillion concentrations, causing corrosion, haze, and lithography defects
Advanced Monitoring Technologies Emerge
New contamination detection systems introduced in 2024-2025 include:
- Real-time AMC monitors: TOFWERK's Semicon AMC Monitor uses time-of-flight mass spectrometry to detect molecular acids, bases, and organics with single-digit ppt sensitivity
- AI-driven analytics: Machine learning systems correlate facility data with contamination events, enabling predictive maintenance and faster root cause analysis
- Sub-10nm particle detection: Condensation particle counters and Scanning Mobility Particle Sizers detect nucleation events missed by traditional optical systems
Top Sources of Cleanroom Contamination
Table 1: Contamination Source Breakdown by Industry Application
| Contamination Source | Contribution Rate | Primary Control | Regulatory Reference |
|---|---|---|---|
| Personnel (skin, hair, garments) | 75-80% | Gowning protocols, training | IEST-RP-CC003, EU GMP Annex 1 |
| Equipment and materials | 10-15% | Qualification, cleaning validation | ISO 14644-5 |
| HVAC / facility infrastructure | 5-10% | Preventive maintenance, pressure cascades | ISO 14644-2 |
| Process activities | 5-10% | Enclosure, automation, RABS | FDA 2004 Aseptic Guidance |
Personnel as Primary Contaminant Source
Humans are responsible for 75-80% of cleanroom contamination, making personnel the single largest contamination vector. A motionless, fully gowned person generates approximately 100,000 particles per minute (≥0.3 µm), escalating to 1,000,000 particles per minute when walking.
Activity-based particle generation:
- Sitting still: baseline 100,000 particles/minute
- Walking: 10x increase to 1,000,000 particles/minute
- Active work: complex movements spike generation to 5,000,000 particles/minute
Contamination pathways include:
- Skin cells shed at over 1 billion cells per 24 hours
- Hair, follicles, and respiratory droplets from breathing and speaking
- Cosmetics, perfumes, and personal care products
- Improper gowning that leaves gaps where particles escape

The "bellows effect" pumps particles out through garment collars and pant legs with each movement, creating a personal contamination cloud that follows operators throughout the cleanroom. ### Equipment and Tools
Even with perfect gowning compliance, the equipment itself becomes a contamination source. Manufacturing equipment introduces particles and chemical contaminants through:
- Friction from moving parts generating wear particles
- Hydraulic systems and lubricants that aerosolize or leak
- Plastics, adhesives, and coatings off-gassing volatile organic compounds (VOCs)
- Cleaning agents or inadequate drying leaving residual contaminants on surfaces
Maintenance tools brought in without proper cleaning validation add external particles, oils, and microorganisms on top of these equipment-generated risks.
Materials and Components
Raw materials, packaging, and incoming components are significant contamination vectors, particularly during pre-cleanroom phases:
- Surface particles, bioburden, and packaging debris arrive with incoming materials before any cleanroom controls apply
- Improper storage conditions allow microbial growth or particle accumulation before materials ever enter the controlled space
- Unpacking, staging, and transfer activities shed particles during the transition into the cleanroom
Pre-cleanroom handling controls — incoming inspection protocols, controlled staging areas, and validated transfer procedures — directly determine how much contamination load enters the facility to begin with.
Environmental and Facility Factors
Facility design and environmental systems contribute to contamination when:
- HVAC failures — inadequate filtration, insufficient air change rates, or filter breaches — allow uncontrolled particles in
- Loss of positive pressure differentials permits unfiltered air infiltration from adjacent spaces
- Humidity swings promote microbial growth at high levels and increase electrostatic particle attraction at low levels
- Design flaws — poor material flow paths, inadequate airlocks, and air stagnation dead zones — create persistent contamination pockets
Process and Procedural Failures
Human error and procedural deviations cause contamination through:
- Inadequate cleaning validation
- Improper documentation or failure to follow SOPs
- Insufficient training on gowning and behavioral protocols
- Skipped environmental monitoring or delayed corrective actions
Microbial Contamination
Bacterial and fungal contamination presents distinct detection and removal challenges:
- Living (viable) microorganisms require fundamentally different detection methods and control strategies than inert particles
- Some organisms carry DNA repair genes and spore-forming capabilities that resist standard decontamination — a finding highlighted in NASA cleanroom research
- Biofilms form protective matrices on surfaces that resist routine cleaning and disinfection
- Water systems, personnel, incoming materials, and environmental surfaces all serve as microbial reservoirs
Emerging Contamination Challenges in Modern Cleanrooms
Cross-Contamination in Multi-Product Facilities
Pharmaceutical and biotech facilities producing multiple drug products face heightened cross-contamination risks. The FDA's 2022 warning letter to Nephron Pharmaceuticals cited cross-contamination between products that the firm failed to identify promptly. EU GMP Annex 1 now mandates appropriate technical and operational separation measures when handling distinct product types concurrently.
Complex Manufacturing Processes
Beyond multi-product separation, the therapies themselves create novel contamination profiles that standard protocols weren't designed to handle:
- Protein-based biologics are highly vulnerable to particulate and microbial contamination
- Living cells in cell therapies introduce biological contamination risks that require ultra-clean environments
- Viral vectors used in gene therapy demand stringent biocontainment to prevent cross-contamination and environmental release

Nanoscale and Molecular Contamination
The contamination challenge extends beyond biology. In semiconductor and advanced electronics manufacturing, traditional particle counters can't detect threats operating at the nanoscale:
- Particles below 20nm and molecular contaminants at parts-per-trillion (ppt) levels cause measurable yield loss
- Standard optical counters are blind to sub-20nm particles and gaseous molecular contaminants entirely
- Time-of-flight mass spectrometry, cavity ring-down spectroscopy, and condensation particle counters are now required to close that detection gap
Best Practices for Contamination Prevention and Control
Comprehensive Personnel Training
Effective training programs include:
- Step-by-step gowning protocols with documented verification checkpoints
- Behavioral controls — no face-touching, slow deliberate movement, and active awareness of particle generation
- Scheduled refresher training paired with competency assessments to confirm retention
- Access restrictions that limit entry to trained, qualified personnel only
Cleaning and Disinfection Protocols
Choosing the right agents — and rotating them — prevents microbial resistance from taking hold:
- Use multiple disinfectant types on a rotation schedule
- Add periodic sporicidal agents to address spore-forming organisms
- Validate cleaning effectiveness through surface sampling before and after
Frequency depends on classification level:
- ISO Class 5 (Grade A/B): Daily or per-shift disinfection
- ISO Class 7/8: Daily to weekly, scaled to activity level
- Gowning rooms: More frequent than the cleanroom itself — high foot traffic means higher particle load
Environmental Monitoring Programs
Strategic monitoring includes:
- Continuous particle counting in ISO Class 5 zones, with periodic sampling in lower-classification areas
- Active air sampling to detect and quantify airborne microorganisms
- Swabs and contact plates for both routine surface checks and deviation investigations
- Trending analysis across all data streams to spot patterns before they become excursions
Material and Equipment Qualification
Every item entering a cleanroom represents a contamination risk. Incoming inspection protocols manage that risk at the source:
- Visually inspect all materials for damage or visible contamination
- Clean or sterilize before cleanroom entry using double-door autoclaves or disinfection chambers
- Document the full chain of custody for traceability
Reusable equipment adds another layer of responsibility:
- Establish and validate cleaning procedures specific to each piece of equipment
- Verify that residual contamination levels meet defined acceptance criteria
- Keep cleaning logs current and schedule periodic revalidation
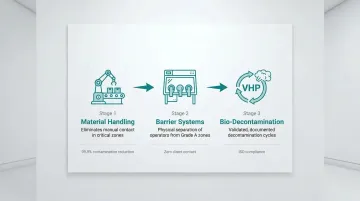
Automation to Reduce Human Contact
Human presence is the single largest source of viable contamination in most cleanrooms. Automated systems address this directly:
- Conveyors, robotic arms, and automated guided vehicles move materials through critical zones without manual handling
- RABS and isolators place a physical barrier between operators and critical zones, cutting viable contamination to near-zero in Grade A environments
- Vaporized hydrogen peroxide systems deliver consistent, validated bio-decontamination cycles with full cycle documentation
Designing Contamination-Resistant Cleanrooms
Proper cleanroom design from the conceptual phase minimizes contamination risks through strategic planning and component selection.
Critical Design Elements
Unidirectional Workflow
- Design material and personnel flow to prevent cross-contamination
- Separate raw material entry from finished product exit
- Implement cascading pressure differentials (highest in critical zones)
HVAC System Design
- ISO Class 5: Unidirectional airflow at 0.36–0.54 m/s (effectively hundreds of air changes per hour)
- ISO Class 7/8: 20–600 air changes per hour depending on classification
- Filtration: HEPA filters (H13/H14) retain ≥99.95% of 0.3 µm particles; ULPA filters (U15–U17) retain ≥99.999% of 0.12 µm particles for ISO Class 3 and stricter
Surface Material Selection
- Non-shedding, non-porous surfaces (stainless steel, epoxy-coated walls)
- Seamless coving at wall-ceiling and wall-floor junctions to eliminate particle traps
- Easy-to-clean, chemically resistant finishes
Elimination of Dead Spaces
- Avoid corners, crevices, and recessed areas where air stagnates
- Design accessible surfaces for thorough cleaning
- Minimize horizontal surfaces where particles settle
Modular Cleanroom Advantages
Modular cleanroom systems offer flexibility to adapt to changing contamination control requirements. ACH Engineering builds these principles into every turnkey cleanroom project, with modular components designed specifically to minimize contamination at every stage:
- Prefabricated modular components reduce on-site construction contamination
- Walkable ceilings eliminate overhead access contamination risks
- Flush-design doors maintain pressure differentials during material and personnel flow

The modular approach also supports future expansion, reconfiguration, and adaptation to evolving regulatory requirements, and ACH Engineering's advanced cleanroom solutions are built to accommodate these changes without major reconstruction.
Regulatory Updates and Compliance Requirements
Staying current with regulatory requirements is one of the most demanding aspects of cleanroom operations. Standards have tightened across every major sector — here's where the key frameworks stand today.
ISO 14644 Series
ISO 14644-1 (2015): Defines cleanroom classification by particle concentration for ISO Class 1–9. Does not cover biological or chemical contamination.
ISO 14644-2 (2015): Mandates risk-based monitoring plans to prove continued compliance, moving beyond simple periodic testing.
ISO 14644-17 (2021): Addresses particle deposition rates, critical for understanding surface cleanliness.
FDA and EU GMP Annex 1
The revised EU GMP Annex 1 (2022) is now the dominant standard for sterile manufacturing:
- Contamination Control Strategy (CCS) — mandatory living document defining all critical control points
- Continuous monitoring — required for Grade A (ISO 5) zones to detect excursions in real time
- RABS/Isolators — explicitly recommended; alternative approaches require documented justification
- Multiple disinfectants — required to prevent microbial resistance, with periodic sporicidal agents

Beyond the cross-industry frameworks, several sectors operate under their own standards that layer additional requirements on top of ISO classification.
Industry-Specific Standards
Semiconductor (SEMI standards):
- SEMI F21: Classification of airborne molecular contaminant levels
- SEMI F104: Test method for particle contribution from ultrapure water and chemical systems
Aerospace (NASA standards):
- NASA-STD-8719.27: Defines biologically controlled cleanrooms for planetary protection
- Recommends ISO Class 5–8 environments for flight hardware with bioburden controls
Medical Device (ISO 13485):
- Specifies requirements for personnel health, cleanliness, and clothing when contact could compromise product safety
- Links environmental control directly to risk management processes
Frequently Asked Questions
What is the most common source of contamination in a cleanroom?
Personnel are the #1 contamination source, accounting for 75–80% of particulates. A walking person generates 1,000,000 particles per minute. Equipment, materials, and process failures contribute the remainder.
How often should a cleanroom be cleaned?
Cleaning frequency depends on ISO classification and activity level. ISO Class 5 areas typically require daily or per-shift disinfection. ISO Class 7/8 areas may be cleaned daily to weekly based on risk assessment and monitoring results.
Can you wear deodorant in a cleanroom?
Most cleanroom protocols prohibit scented products including deodorant, perfume, and cologne. These contain volatile organic compounds that contribute to chemical contamination and can generate particles, compromising the controlled environment.
What are the different cleanroom classifications and their contamination limits?
ISO 14644-1 defines ISO Class 1–9 based on particle counts. ISO Class 5 (pharmaceutical Grade A/B) allows maximum 3,520 particles/m³ at ≥0.5 µm. ISO Class 7 (Grade C) allows 352,000 particles/m³ at ≥0.5 µm.
How do you detect contamination in a cleanroom?
Standard detection methods include:
- Real-time particle counters for airborne particulates
- Viable air samplers for microbial contamination
- Surface swabs and contact plates for surface monitoring
- Settle plates for passive air sampling
- Mass spectrometry for molecular contaminants
What happens if a cleanroom fails contamination testing?
Standard protocol involves:
- Immediate investigation, documentation, and potential production hold
- Root cause analysis to identify the contamination source
- Corrective and preventive actions (CAPA) implementation
- Retesting to verify the fix before resuming operations


