
Introduction
Cleanroom construction costs range from $200 to over $1,000 USD per square foot — and that's before classification requirements push the number higher. Many manufacturers struggle with the intersection of strict regulatory standards, specialized mechanical systems, and the precision execution these projects demand.
According to industry data, HVAC systems alone consume 25-50% of total project budgets, while moving from an ISO 8 to ISO 5 classification can increase costs by over 1,000%. At that scale, a single planning misstep doesn't just delay the project — it risks costly retrofits, failed commissioning, or regulatory rejection.
This guide covers the full cleanroom building process: ISO classifications and regulatory requirements, planning and budgeting, HVAC and material selection, construction protocols, and commissioning. Whether you're building your first controlled environment or expanding existing capacity, you'll leave with a clear framework for making decisions that hold up under scrutiny.
TL;DR
- ISO 14644-1 classifications (ISO 5–8 most common) set particle count limits that drive air change rates, filter coverage, and cost
- Budget CAD $200–$1,000+/sq ft and plan for 6–18 month timelines before design work begins
- HVAC accounts for 25–50% of total project costs — airflow, pressure cascades, and filtration must match your ISO class
- Modular construction installs 30–50% faster than stick-built and supports future reconfiguration
- Commissioning uses IQ/OQ/PQ protocols; requalification runs every 6–12 months post-handover
Understanding Cleanroom Classifications and Requirements
ISO 14644-1 Classification System
The ISO 14644-1:2015 standard replaced the cancelled U.S. Federal Standard 209E in 2001 and now serves as the global framework for air cleanliness classification. Classification is based on maximum allowable particle concentrations per cubic meter of air at specific particle sizes.
Key ISO Classifications:
| ISO Class | Particles/m³ ≥0.5 µm | Equivalent FS 209E | Typical Applications |
|---|---|---|---|
| ISO 5 | 3,520 | Class 100 | Sterile filling, semiconductor fabrication |
| ISO 6 | 35,200 | Class 1,000 | Aseptic processing, critical device assembly |
| ISO 7 | 352,000 | Class 10,000 | Pharmaceutical manufacturing, packaging |
| ISO 8 | 3,520,000 | Class 100,000 | General manufacturing, warehousing |
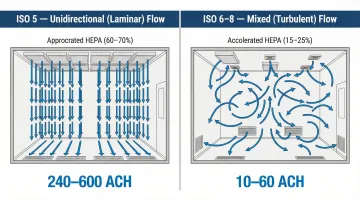
Each classification jump requires exponentially more air changes and filtration. ISO 5 environments may need 240-600 air changes per hour (ACH) compared to just 10-25 ACH for ISO 8, directly impacting both construction and operational costs.

Industry-Specific Regulatory Requirements
Different industries face distinct regulatory frameworks beyond ISO classifications:
Pharmaceutical (FDA cGMP): 21 CFR 211.42 mandates separate or defined areas for aseptic processing with smooth, hard, easily cleanable surfaces. Requirements include HEPA-filtered air under positive pressure and comprehensive environmental monitoring programs.
EU GMP Annex 1: The 2022 revision aligns Grade A environments with ISO Class 5 and explicitly requires surfaces to be "smooth, impervious and unbroken" to minimize particle shedding and accumulation.
Medical Device (ISO 13485): Clause 6.4 requires manufacturers to document work environment requirements when they impact product quality, typically necessitating ISO 7-8 environments for assembly and packaging to minimize bioburden.
How Classification Determines Design Requirements
Each regulatory requirement above maps directly to physical design parameters — and your ISO classification determines the specific targets your facility must hit:
Air Change Rates:
- ISO 8: 10-25 ACH for basic particle dilution
- ISO 7: 30-60 ACH for recovery from personnel activity
- ISO 6: 70-160 ACH for process particle management
- ISO 5: 240-600+ ACH for unidirectional flow
Filter Coverage: ISO 5 environments require 60-70% ceiling coverage with HEPA filters to achieve unidirectional airflow, while ISO 7-8 may need only 15-25% coverage for adequate mixing.
System Redundancy: Higher classifications typically require backup systems for critical components to prevent contamination events during maintenance or failures.
Cost Impact of Classification Choice
According to a Compass International construction cost report, cleanroom construction costs follow a non-linear trajectory as cleanliness requirements increase. Moving from ISO 8 to ISO 5 can increase costs by over 1,000%—from approximately $100–250/sq ft (USD) to over $1,000/sq ft—driven by exponential increases in air handling requirements and filter coverage.
Each ISO class jump typically adds 20-40% to both construction and operational expenses. Getting the classification right at the planning stage — not over-specifying or under-specifying — is where projects win or lose on budget.
Planning Your Cleanroom Project
Establish Comprehensive Project Scope
Begin by documenting intended use, production volume, equipment requirements, and future expansion needs. Incomplete scope definition is the leading cause of budget overruns and costly mid-project redesigns.
A User Requirements Specification (URS) should capture every piece of equipment along with its environmental needs — temperature, humidity, vibration limits — and utility requirements such as electrical, compressed air, and process gases. This document becomes the foundation for all subsequent design decisions.
Develop Realistic Budget Allocations
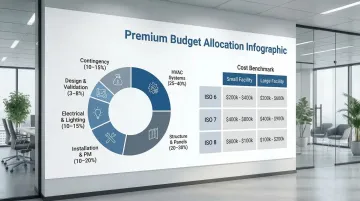
Typical cleanroom budgets should account for:
- HVAC Systems: 25-40% (air handling units, ductwork, filters, controls)
- Structure & Panels: 20-30% (walls, ceiling grid, framework)
- Installation & Project Management: 10-20%
- Electrical & Lighting: 10-15%
- Design & Validation: 3-8%
- Contingency: 10-15% minimum for unforeseen conditions
Cost Benchmarks by Classification (USD):
| Classification | Small (<500 sq ft) | Large (>2,500 sq ft) |
|---|---|---|
| ISO 6 | $300-$550/sq ft | $200-$450/sq ft |
| ISO 7 | $250-$500/sq ft | $150-$400/sq ft |
| ISO 8 | $200-$450/sq ft | $100-$350/sq ft |
Modular construction typically costs 20-30% less than stick-built alternatives while offering faster installation timelines.
Create Realistic Project Timelines
Total project duration typically ranges from 6-18 months:
- Design Phase: 2-4 months for drawings, specifications, and engineering
- Permitting: 1-3 months depending on jurisdiction and project complexity
- Construction: 4-12 months based on size, classification, and site conditions
- Commissioning/Qualification: 1-2 months for testing and validation
Modular cleanroom systems can reduce construction time by 30-50% through parallel off-site fabrication while site preparation occurs.
Assemble Specialized Project Team
Successful cleanroom projects require expertise beyond standard construction:
- Owner's Representative: Protects your interests and coordinates between contractors
- Cleanroom Design Firm: Specialized experience with classification requirements and regulatory compliance
- Mechanical/Electrical Engineers: HVAC and utility system design expertise
- General Contractor: Demonstrated cleanroom construction experience, not just general building
- Commissioning Agent: Independent verification of system performance
Before finalizing your team, ask each firm for references from cleanroom projects in your specific industry and ISO classification range — not just general construction portfolios.
Conduct Thorough Site Evaluation
Assess existing facilities or potential sites across four key areas:
- Structural capacity: Rooftop HVAC equipment can add 50-100 lbs per square foot of load — verify the structure can handle this before finalizing mechanical designs
- Ceiling height: Plan for a minimum 12-14 feet to accommodate plenum space above the cleanroom ceiling; limiting internal height to 10-12 feet reduces total air volume and HVAC capacity requirements
- Utility capacity: Cleanrooms typically require 50-100 watts per square foot of electrical service, plus water supply for humidification and compressed air or process gases — redundancy requirements for critical utilities should be evaluated early
- Environmental factors: Nearby equipment, traffic vibration, electromagnetic interference, and external contamination sources can all affect process quality and makeup air intake design
Cleanroom Design Essentials
HVAC and Air Handling Systems
The HVAC system represents the critical foundation of cleanroom performance and consistently consumes 25-50% of project budgets. System design must address four primary functions:
Makeup Air Units (MAU): Provide conditioned outside air to maintain positive pressure and replace air lost through exhaust systems. MAUs handle the heaviest thermal and humidity loads, demanding industrial-grade dehumidification in humid climates and humidification in dry regions.
Recirculation Air Systems: Continuously filter and recirculate room air to dilute particle concentrations. These systems move the massive air volumes required for high air change rates while maintaining precise temperature control.
Exhaust Systems: Remove contaminated air from process equipment, chemical storage areas, and hazardous material handling zones. Size exhaust capacity to handle worst-case scenarios including equipment purges and emergency ventilation.
Air Distribution: ISO 5 environments require unidirectional (laminar) airflow with HEPA filters covering 60-70% of the ceiling. ISO 6-8 classifications use mixed airflow with strategically placed supply diffusers and return grilles to create proper air patterns without dead zones.
Room Layout and Pressure Cascades
Once HVAC systems are sized, room layout determines how pressure relationships enforce contamination control through airflow direction:
- Highest pressure: Critical processing areas (ISO 5-6)
- Medium pressure: Buffer zones and gowning rooms (ISO 7-8)
- Lower pressure: Corridors and support spaces
- Negative pressure: Hazardous material areas, chemical storage, and exhaust systems
EU GMP Annex 1 recommends minimum pressure differentials of 10 Pascals between adjacent rooms of different grades. ASHRAE guidelines for pharmaceutical applications suggest +0.020 inches w.c. for non-hazardous areas.
Construction Materials and Finishes
Pressure cascades only work when the envelope holds. Material selection focuses on minimizing particle generation and enabling effective cleaning:
Wall and Ceiling Systems: Smooth, non-porous surfaces are mandatory. Options include pharmaceutical-grade vinyl panels, epoxy-coated gypsum, or modular wall systems with flush-mounted joints. All penetrations must be sealed to prevent particle infiltration.
Flooring: Seamless, heat-welded vinyl or epoxy systems prevent particle accumulation in joints. Coved bases (curved wall-to-floor junctions) are required in aseptic areas to eliminate corners where contamination can accumulate and facilitate effective cleaning.
Fixtures and Fittings: Specify flush-mounted light fixtures, outlets, and switches. Minimize horizontal surfaces where particles can settle. Use stainless steel or powder-coated aluminum for durability and cleanability.
Filtration Strategy
Match filter specifications to classification requirements:
HEPA Filters: Standard specification is 99.97% efficiency at 0.3 microns, suitable for ISO 5-8 environments. Filters must be tested for leaks after installation using aerosol challenge methods.
ULPA Filters: Required for ISO 4 or cleaner environments, offering 99.999% efficiency at 0.12 microns. Significantly higher pressure drop and cost than HEPA filters.
Pre-filtration: Install MERV 8-13 pre-filters upstream of HEPA filters to extend filter life and reduce operating costs. Pre-filters capture larger particles before they reach expensive final filters.
Modular vs. Traditional Construction
Modular cleanroom construction offers distinct advantages for many applications:
- Cuts construction time by 30-50% through off-site panel fabrication running in parallel with site prep
- Reduces total project cost by 20-30% versus stick-built alternatives — especially on projects under 5,000 sq ft
- Allows panels to be reconfigured, relocated, or expanded without extensive demolition as processes change
ACH Engineering designs and builds modular cleanrooms — including modular cleanroom devices and components — across ISO 5-8 classifications for pharmaceutical, biotech, and medical device clients throughout Canada. GMP-compliant components — including customizable wall panels, walkable ceilings, flush-design doors, and seamless coving — are manufactured and installed as a turnkey package.
Construction Process and Best Practices
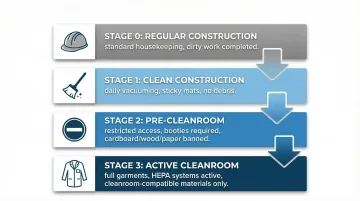
Four-Stage Clean Construction Protocol
Implementing staged cleanliness protocols prevents latent contamination that can compromise final performance:
Stage 0 (Regular Construction): Standard construction practices with enhanced housekeeping. Complete all "dirty work"—drywall installation, concrete cutting, major structural modifications—before sealing the cleanroom envelope.
Stage 1 (Clean Construction): Daily vacuuming and debris removal become mandatory. Introduce sticky mats at entry points to control foot traffic contamination. No drywall dust or construction debris allowed.
Stage 2 (Pre-Cleanroom): Access restricted to essential personnel only. Workers wear booties and protective coverings; cardboard, wood, and paper are banned from the space. Air systems may run without final filters to begin air scrubbing.
Stage 3 (Active Cleanroom): Most restrictive phase. Full garments required—lab coats, hair nets, gloves. HEPA-filtered recirculation systems are active, and only cleanroom-compatible materials and tools are permitted.
Critical Installation Sequences
Follow this sequence to prevent rework and contamination:
- Rough-In Utilities: Install major ductwork, piping, and electrical infrastructure before cleanroom envelope
- Cleanroom Envelope: Install wall panels, ceiling grid, and framework
- HVAC/Mechanical Systems: Install air handling units, ductwork, and controls
- Flooring: Install seamless flooring with coved bases
- Final Filters: Install HEPA/ULPA filters only after Stage 2 cleaning complete
- Equipment Installation: Bring in process equipment and connect utilities
- Final Cleaning: Deep clean all surfaces before commissioning
Quality Control Checkpoints
Implement verification at each stage:
- Envelope Pressure Testing: Verify room can hold specified pressure differentials
- Filter Leak Testing: Aerosol challenge testing of all HEPA filters after installation
- Airflow Verification: Measure air velocities and patterns with smoke studies
- Documentation: Photograph all concealed work before covering for future reference
Those checkpoints are only as reliable as the team executing them — which is why contractor qualifications deserve the same rigour as the technical specs.
Contractor Qualifications and Oversight
Verify cleanroom-specific experience:
- Request references from similar classification projects in your industry
- Require detailed cleaning protocols and material restrictions
- Establish daily inspection routines with documented findings
- Maintain photographic documentation of all installation stages
A turnkey provider like ACH Engineering manages design, construction, and commissioning under one contract — meaning a single point of contact from permit drawings through final performance sign-off, with no handoff gaps between trades.
Commissioning and Validation
Qualification Scope and Protocols
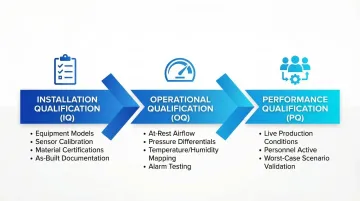
Commissioning follows the IQ/OQ/PQ sequence to verify and document cleanroom performance:
- Installation Qualification (IQ): Verifies correct installation per specifications — equipment models, sensor calibration, material certifications, and as-built documentation.
- Operational Qualification (OQ): Tests system performance under "at-rest" conditions (no personnel or active processes). Covers airflow velocity, pressure differentials, temperature/humidity mapping, and alarm functions.
- Performance Qualification (PQ): Demonstrates consistent operation during live production with personnel and equipment active. Validates classification under worst-case scenarios.
Required Testing Protocols
ISO 14644-3:2019 specifies mandatory tests for certification:
| Test | What It Measures | Key Threshold |
|---|---|---|
| Particle Count | Concentrations at "at-rest" and "operational" states, worst-case locations | Per ISO classification limits |
| Airflow Velocity & Uniformity | Air change rates and filter face velocity | 0.45 m/s ±20% for ISO 5 unidirectional |
| HEPA Filter Integrity | Aerosol challenge (PAO/DOP) for media and seal leaks | Leaks >0.01% require repair/replacement |
| Pressure Differential | Cascade function with doors closed and during personnel movement | Per design specifications |
| Recovery | Time for particle counts to return to baseline post-contamination | Faster = more effective air change rate |
Certification Requirements and Frequency
ISO 14644-2 mandates ongoing requalification to prove continued control:
- ISO 5 and cleaner: Maximum 6-month intervals
- ISO 6-9: Maximum 12-month intervals
- After modifications: Requalification required after any significant changes to facility or equipment
Certification isn't legally required for every cleanroom, but it's standard practice and typically mandated by FDA, Health Canada, ISO 13485, and contractual obligations. Plan to allocate 3-5% of total project cost for comprehensive commissioning and validation.
Common Challenges and Solutions
Most cleanroom projects run into the same three problems: budget overruns, schedule delays, and performance failures at commissioning. Each is preventable with the right approach.
Budget Overruns
Challenge: Incomplete User Requirements Specification or scope creep during design and construction phases.
Solution: Front-load planning to lock in scope before a single drawing is issued:
- Invest 2-4 weeks in upfront planning to document every equipment requirement, utility need, and operational parameter
- Require documented cost impacts before approving any scope modification
- Reserve 10-15% contingency for unforeseen site conditions — not scope additions
Schedule Delays
Challenge: Long-lead equipment (12-20 weeks for specialized air handlers) combined with multi-trade coordination gaps.
Solution: Time and sequencing are everything on a cleanroom build:
- Procure critical equipment 6-9 months ahead of installation dates
- Run weekly coordination meetings with all trades against a detailed sequencing plan
- Consider modular construction to compress timelines through parallel off-site fabrication
Performance Failures
Challenge: Commissioning deficiencies discovered after construction completion require costly remediation.
Solution: Engage your commissioning agent during design, not at turnover. Budget 3-5% of project cost for testing and validation. Implement Stage 2-3 construction protocols early — latent contamination is far harder to resolve than a deficiency caught on a drawing.
Conclusion
Successful cleanroom construction demands equal attention to planning, design, and execution. The significant investment—ranging from $200 to over $1,000 per square foot—requires thorough understanding of ISO classifications, regulatory requirements, and specialized construction protocols. Shortcuts in any phase compromise the entire investment and can lead to regulatory failures or costly retrofits.
That risk makes the choice of partner critical. Working with experienced cleanroom specialists who understand your industry's classification requirements helps you navigate the complex intersection of mechanical systems, construction techniques, and regulatory compliance without costly missteps.
ACH Engineering provides end-to-end cleanroom solutions across North America, handling design, construction, and commissioning under one roof. Their modular systems install faster than traditional builds, adapt to future changes, and are backed by project management that runs from initial planning through validation and certification.
Frequently Asked Questions
How much does it cost to build a cleanroom?
Cleanroom costs range from $200 to over $1,000 per square foot depending primarily on ISO classification. ISO 8 facilities typically cost $200-400/sq ft, ISO 6-7 mid-range facilities run $400-700/sq ft, and ISO 5 or cleaner environments cost $700-$1,000+/sq ft. HVAC systems represent 25-50% of total project costs, making mechanical design the largest single expense.
What are the FDA guidelines for cleanrooms?
In the US, FDA requires pharmaceutical cleanrooms to comply with cGMP regulations under 21 CFR Parts 210-211, mandating environmental monitoring programs, documented cleaning procedures, and cleanroom qualification/validation. In Canada, Health Canada enforces equivalent GMP standards under the Food and Drug Regulations, with both frameworks aligned to ICH Q10 principles.
What is not allowed in a cleanroom?
Prohibited items include cardboard (sheds fibers), wood (releases particles), paper products, pencils (graphite sheds), non-approved adhesives, and personal items including jewelry, makeup, and perfume. Any materials that shed particles or cannot be properly cleaned and sanitized are restricted. During construction, Stage 2 protocols ban these materials to prevent latent contamination.
Do cleanrooms need to be certified?
While certification isn't legally required for all cleanrooms, it's industry best practice and often required by contract or regulation. Pharmaceutical and medical device cleanrooms must be validated per FDA and ISO 13485 requirements. ISO 14644-2 requires initial certification and requalification every 6 months for ISO 5 environments and annually for ISO 6-8.
How long does it take to build a cleanroom?
Typical timelines run 6-18 months: 2-4 months for design, 1-3 months for permitting, 4-12 months for construction, and 1-2 months for commissioning. Modular cleanroom systems can cut construction time by 30-50% through off-site fabrication running parallel to site preparation.
What ISO classification do I need for my cleanroom?
Classification depends on product sensitivity and regulatory requirements. Pharmaceutical and medical device manufacturing typically requires ISO 7-8 (Class 10,000-100,000), semiconductor fabrication needs ISO 5-6 (Class 100-1,000), and sterile filling operations require ISO 5 (Class 100) or better. A cleanroom engineering specialist can confirm the right classification for your specific process and applicable regulations.


