
Introduction
Medical device manufacturers face a critical challenge: establishing cleanroom environments that meet strict regulatory requirements while maintaining operational efficiency. Determining the appropriate ISO classification, designing HVAC systems that balance contamination control with energy costs, and navigating the complex validation requirements that FDA and ISO 13485 audits demand remains a persistent challenge.
Manufacturers producing sterile or high-purity medical devices—implantable components, surgical instruments, and drug delivery systems—face particularly high stakes. Even microscopic contamination can compromise patient safety and trigger costly recalls.
The technical specifications matter significantly: ISO Class 7 cleanrooms require 30-60 air changes per hour, while over-classifying a facility can increase HVAC energy consumption by up to 80%.
To address these challenges, this guide covers ISO classifications, planning considerations, step-by-step setup procedures, validation requirements, and ongoing compliance strategies—providing the technical foundation needed to build a compliant, cost-effective cleanroom for medical molding operations.
TL;DR
- ISO Class 7 (30-60 ACH) or Class 8 (10-25 ACH) cleanrooms are standard for medical molding
- Modular cleanroom costs: $120-$250/sq ft (ISO 7), $50-$120/sq ft (ISO 8); 4-12 week install
- Electric molding presses cut particle generation versus hydraulic systems
- Validation includes IQ/OQ/PQ phases with annual recertification mandatory for regulatory compliance
Understanding ISO Cleanroom Classifications for Medical Molding
Particle Count Standards
Cleanroom classifications for medical molding depend on precise particle control. ISO 14644-1:2015 defines these classifications based on maximum allowable airborne particle concentrations per cubic meter, with the critical measurement being particles ≥0.5 µm in diameter.
ISO Classification Limits:
| ISO Class | Max Particles/m³ (≥0.5 µm) | Typical Medical Molding Application |
|---|---|---|
| ISO 5 | 3,520 | Implantable device assembly, sterile filling |
| ISO 7 | 352,000 | General device assembly, molding of sterile-path devices |
| ISO 8 | 3,520,000 | Non-sterile device molding, packaging, gowning rooms |

Most medical molding operations require ISO Class 7 or Class 8, depending on device classification and whether molding occurs under sterile conditions.
FDA guidance recommends that supporting clean areas meet at least ISO Class 8 under dynamic conditions, while critical areas require ISO Class 5.
Air Change Requirements
The classification you select drives significant differences in HVAC system design and operating costs:
- ISO Class 7: Requires 30-60 air changes per hour (ACH)
- ISO Class 8: Requires 10-25 ACH
This difference translates directly to capital and operational expenses—ISO 7 cleanrooms typically cost 2-3x more to build and operate than ISO 8 facilities due to increased filtration, higher fan capacity, and greater energy consumption.
Operational States Matter
Beyond selecting the right classification, understanding occupancy states determines testing success. ISO 14644-1 defines three states:
- As-built: Facility complete but no equipment or personnel
- At-rest: Equipment installed and operating, no personnel
- Operational: Facility functioning with personnel present
Your cleanroom must meet particle count limits in the operational state—the most challenging condition. Many facilities pass at-rest testing but fail when personnel and molding equipment introduce particle loads during actual manufacturing.
Regulatory Alignment
FDA and ISO 13485 require manufacturers to justify their cleanroom classification through documented risk analysis. This assessment must consider device classification (Class I, II, or III), patient contact surfaces, sterility requirements, and bioburden control strategies.
When full-facility cleanrooms prove cost-prohibitive, some manufacturers use "clean room machines"—molding equipment with localized Class 7 enclosures. This approach provides stringent control exactly where needed while reducing overall facility requirements.
Planning and Design Considerations
Risk Assessment First
Begin with a thorough risk assessment that determines:
- Required cleanroom class based on device risk profile
- Necessary room size and layout for current and future production
- Whether modular or traditional construction suits your scalability needs
- Integration requirements for existing or planned molding equipment
This assessment drives every downstream decision and prevents costly rework.
Site Selection Factors
Critical infrastructure requirements:
- Ceiling height: Minimum 9-12 feet to accommodate HEPA filter plenums and ductwork
- Structural capacity: Adequate load-bearing for HVAC systems and equipment
- Utility access: Proximity to electrical, water, and compressed air services
- Isolation: Distance from high-vibration areas or contamination sources
Architectural Requirements
Cleanroom construction demands specialized materials and design:
- Surfaces: Non-porous, easily cleanable materials—epoxy-coated steel, aluminum honeycomb, or high-pressure laminate for walls
- Floors: Seamless epoxy or heat-sealed vinyl to prevent bioburden accumulation
- Corners: Coved radius corners eliminate particle traps
- Penetrations: All wall, ceiling, and floor penetrations must be sealed to maintain pressure integrity
HVAC and Filtration Design
The HVAC system represents 25-40% of total project cost and determines whether you achieve certification.
Critical design elements:
- Filtration: HEPA filters (99.97% efficiency at 0.3 µm) for Class 7/8; ULPA filters for higher classes
- Pressure control: Minimum 10-15 Pascals positive pressure differential between adjacent zones to prevent contamination ingress
- Environmental control: Temperature 68-72°F, humidity 30-50% RH to control microbial growth and static
- Air changes: 20-40 ACH for ISO Class 7, 40-60 ACH for ISO Class 6 during operation

Controlled Entry Protocols
Design traffic flow to minimize contamination introduction:
- Gowning areas: Separate rooms with appropriate storage, hand washing stations, and sticky mats
- Airlocks: Interlocked doors that prevent simultaneous opening and pressure loss
- Material pass-throughs: Dedicated transfer chambers for supplies and components
- Segregated flow: Separate paths for personnel, materials, and waste removal
Equipment Integration
Molding equipment selection directly impacts cleanroom performance:
- All-electric presses preferred: Hydraulic presses generate significantly higher particle concentrations due to oil mist and mechanical wear
- Equipment placement: Position machines to optimize airflow patterns and avoid dead zones
- Utility connections: Route power, cooling, and material feeds to maintain room integrity
Environmental Monitoring Systems
Install continuous monitoring for compliance documentation:
- Particle counters at multiple locations
- Differential pressure monitors with alarms
- Temperature and humidity sensors
- Data logging systems for audit trails
Specialized providers like ACH Engineering offer turnkey solutions that handle design, engineering, and installation, ensuring all elements integrate properly for medical molding compliance.
Step-by-Step Cleanroom Setup Process
Cleanroom installation follows a defined sequence where each phase must be completed and verified before proceeding. Shortcuts or improper sequencing often lead to costly rework and certification delays.
1. Site Preparation and Structural Work
Complete all structural modifications before cleanroom construction begins:
- Finish building envelope work and weatherproofing
- Install utility rough-ins (electrical, HVAC, plumbing)
- Ensure the space is clean, dry, and ready to maintain environmental conditions
- Level floors to within specified tolerances (typically ±1/8 inch)
2. Cleanroom Construction and Sealing
With the site prepared, the next phase focuses on building the cleanroom envelope itself:
- Erect wall panels, ceiling grid systems, and flooring
- Seal all joints, penetrations, and interfaces—every gap is a contamination pathway
- Install coved corners at all wall-ceiling and wall-floor intersections
- Apply sealant to all panel connections and service penetrations
3. HVAC and Filtration System Installation
Once the physical structure is sealed, the critical environmental control systems come next. Commission the air handling system to verify performance:
- Install air handling units, ductwork, and return air plenums
- Mount HEPA filter housings and filters
- Verify proper air changes per hour using calibrated instruments
- Test pressure differentials between zones (minimum 10-15 Pa)
- Confirm uniform airflow patterns without dead spots using smoke visualization
4. Lighting, Electrical, and Monitoring Systems
Integrate all electrical and monitoring infrastructure:
- Install cleanroom-rated LED lighting (flush-mounted to avoid particle accumulation)
- Connect electrical outlets and equipment power
- Mount environmental monitoring sensors (particle counters, pressure, temperature, humidity)
- Integrate systems with building management controls for continuous oversight
5. Gowning Room and Airlock Setup
Establish contamination control protocols:
- Configure gowning areas with appropriate storage for cleanroom garments
- Install hand washing and sanitizing stations
- Position sticky mats at entry points
- Set up airlocks with interlocked doors to prevent simultaneous opening
6. Equipment Installation and Integration
The final phase involves bringing in production equipment while maintaining cleanroom integrity. Coordinating this stage requires careful planning to avoid compromising the controlled environment:
- Position molding machines to maintain optimal airflow patterns
- Connect utilities (power, cooling, material feeds) through sealed penetrations
- Install auxiliary equipment (dryers, conveyors, robots)
- Verify that equipment placement doesn't create dead zones or disrupt air patterns

Validation, Testing, and Certification
Cleanroom validation follows a structured three-phase approach, with each phase requiring specific test protocols and documented acceptance criteria.
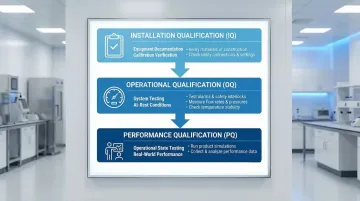
The IQ/OQ/PQ Framework
Installation Qualification (IQ):
- Documents that equipment installed per design specifications
- Records HEPA filter ratings, duct connections, and system components
- Confirms all instruments are calibrated and traceable
Operational Qualification (OQ):
- Tests that systems operate within specified limits under at-rest conditions
- Measures ACH rates, pressure differentials, temperature, and humidity
- Performs HEPA filter integrity testing using aerosol challenge methods
Performance Qualification (PQ):
- Verifies cleanroom maintains ISO classification under operational conditions
- Tests with personnel present and equipment running
- Documents that the facility can consistently meet requirements during actual manufacturing
Once these qualification phases are complete, ongoing testing procedures verify continued compliance.
Critical Testing Procedures
Particle Count Testing:
- Use calibrated optical particle counters compliant with ISO 21501-4
- Sample at multiple locations based on statistical grid (determined by room area)
- Test in both at-rest and operational states
- Document that particle concentrations remain below classification limits
Airflow Visualization:
- Use smoke or fog to identify dead zones, turbulent areas, or reverse flow
- Verify unidirectional flow in critical areas
- Confirm proper air movement from clean to less clean zones
HEPA Filter Integrity:
- Challenge filters with aerosol (typically PAO or DOP)
- Scan downstream face with photometer
- Ensure no leaks exceed 0.01% of upstream concentration
Additional Validation Tests:
- Pressure differential verification between adjacent zones
- Temperature and humidity mapping throughout the space
- Recovery time testing (how quickly room returns to specification after disturbance)
- Air change rate verification using tracer gas or other methods
Qualified personnel must perform all validation testing using calibrated instruments. Complete documentation packages are required for regulatory submissions and compliance audits.
Common Setup Challenges and Solutions
Pressure Differential Failures
Maintaining the required 10-15 Pa differential between cleanroom and adjacent spaces proves challenging when HVAC capacity falls short, door seals wear down, or multiple entry points open simultaneously. Filter loading also increases system resistance over time, gradually degrading pressure control.
Addressing these issues requires a multi-point approach:
- Upgrade air handling capacity if the system is undersized for the actual space volume
- Install high-quality door seals with automatic closers to prevent pressure loss
- Implement airlock protocols with interlocked doors that prevent simultaneous opening
- Deploy continuous pressure monitors with alarms to catch failures immediately
ACH Engineering's cleanroom project management approach addresses pressure control during the initial design phase, sizing HVAC systems with appropriate margins and specifying commercial-grade door hardware that maintains seal integrity under continuous use.
Operational Particle Count Failures
Even cleanrooms that pass at-rest testing often fail operational qualification when real-world activities introduce contamination. Personnel movement, material handling, and molding equipment all generate particles that static testing doesn't capture.
The most effective solutions target specific contamination sources. Inadequate gowning allows personnel to shed particles, so enhanced training and stricter protocols make an immediate difference. Material introduction requires decontamination procedures—wiping with isopropyl alcohol or passing through air showers before entry.
Equipment selection matters significantly. Hydraulic molding presses can generate particles through fluid mist and mechanical wear, while all-electric presses eliminate these sources entirely. When equipment upgrades aren't feasible, adding localized HEPA filtration near high-generation areas contains contamination at the source. If particle counts still exceed specifications, increasing air change rates provides additional dilution, though this requires HVAC capacity margins built into the original design.
Thermal Control Issues
Thermal Control Issues
Temperature and humidity hot spots frequently develop when heat-generating molding equipment concentrates in one area or when air distribution fails to provide adequate mixing. These variations create zones outside specification that compromise product quality.
Corrective approaches depend on root cause:
- Rebalance airflow if duct design creates uneven distribution
- Add supplemental cooling for concentrated equipment clusters
- Install zone controls to manage different areas independently
Equipment placement decisions made during initial layout significantly impact long-term thermal control. Distributing heat-generating machinery throughout the space, rather than clustering it, reduces localized thermal loads. When relocation isn't practical, enclosing heat sources with dedicated exhaust prevents thermal pollution of the broader cleanroom environment.
Ongoing Maintenance and Compliance
Preventive Maintenance Schedule
Establish routine maintenance to preserve cleanroom performance:
Filter maintenance:
- Replace pre-filters every 3-6 months to protect HEPA filters
- Replace HEPA filters every 3-5 years or when pressure drop doubles
- Replacing filters based on pressure drop rather than calendar intervals can reduce energy costs by approximately 5%
System maintenance:
- Clean ductwork and plenums annually
- Calibrate monitoring instruments per manufacturer specifications
- Inspect and replace door seals as needed
- Service HVAC equipment per maintenance schedules
Environmental Monitoring Protocols
Implement tiered monitoring appropriate to your classification:
Daily monitoring:
- Particle counts at designated locations
- Pressure differential readings
- Temperature and humidity verification
- Visual inspections for cleanliness
Weekly/monthly monitoring:
- Expanded particle count testing
- Surface sampling for bioburden (if required)
- Equipment performance verification
- Documentation review
Document all results and investigate any deviations immediately.
Trending data helps identify problems before they cause certification failures, which directly impacts your recertification timeline.
Recertification Requirements
ISO 14644-2 mandates routine testing to prove continued compliance:
- ISO Class 7 & 8: Recertification every 12 months
- ISO Class 5: Recertification every 6 months
Recertification uses the same validation protocols performed during initial qualification, including particle counting, airflow verification, and pressure differential testing.
Maintain comprehensive documentation for regulatory audits and FDA inspections.
Cost Considerations and ROI
Capital Expenditure Ranges
Based on 2024-2026 market data, modular cleanroom costs vary significantly by classification:
Modular Cleanroom Costs:
- ISO Class 8: $65-$155 CAD per square foot
- ISO Class 7: $155-$320 CAD per square foot
- ISO Class 6: $320-$640 CAD per square foot
Traditional stick-built construction typically costs 20-40% more than modular systems but offers greater customization for permanent installations. Modular systems from providers like ACH Engineering offer faster installation timelines and flexibility for future expansion while reducing upfront costs.
Cost Breakdown
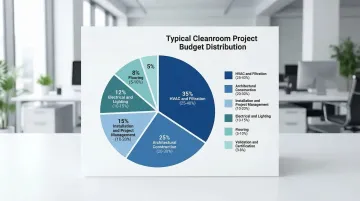
A typical cleanroom project budget breaks down as:
- HVAC and filtration: 25-40% (largest single component)
- Architectural construction: 20-30%
- Installation and project management: 10-20%
- Electrical and lighting: 10-15%
- Flooring: 5-10%
- Validation and certification: 3-8%
Operational Expenditures
Beyond initial construction, ongoing operations represent a significant investment.
Annual operating costs typically equal 15-25% of initial capital investment. Major expenses include:
- Energy costs: Approximately 40% of OPEX, mainly HVAC operation
- Filter replacement: Pre-filters and HEPA filters on schedule
- Maintenance: Preventive and corrective maintenance
- Monitoring and validation: Annual recertification and ongoing testing
- Personnel: Cleaning staff and environmental monitoring
Return on Investment
Calculate ROI based on multiple factors:
Risk mitigation:
- Prevents costly product recalls from contamination
- Protects regulatory approval and market access
- Reduces rejection rates and rework costs
Market access:
- Enables production of higher-value medical devices
- Opens sterile and implantable device markets
- Demonstrates quality commitment to customers and regulators
Operational efficiency improvements contribute to bottom-line savings. Cleanrooms reduce contamination-related downtime, improve process consistency and yield, and support lean manufacturing initiatives.
For a 2,000 sq ft ISO Class 7 cleanroom at $260/sq ft CAD ($520,000 CAD capital investment), typical payback periods range from 2-5 years depending on product portfolio and contamination risk profile.
Conclusion
Cleanroom setup for medical molding represents a significant investment that directly impacts product quality, regulatory compliance, and patient safety. Proper planning—including accurate risk assessment, appropriate classification selection, and disciplined design execution—is essential for success.
Critical decisions that impact both capital and operational costs include:
- ISO Class 7 versus Class 8 classification selection
- Modular versus traditional construction approach
- All-electric versus hydraulic molding equipment
- HVAC system design and filtration specifications
Facilities that cut corners during design or construction inevitably face costly rework, certification delays, or ongoing compliance challenges.
These risks underscore the value of working with experienced controlled environment specialists. Early engagement with cleanroom design and engineering teams—particularly those offering turnkey solutions and end-to-end project management—helps avoid common pitfalls while ensuring your facility meets both current regulatory requirements and future expansion needs. The investment in proper cleanroom setup protects your ability to manufacture and sell medical devices while safeguarding the patients who depend on them.
Frequently Asked Questions
What ISO cleanroom class is required for medical device molding?
ISO Class 7 is standard for sterile or implantable devices, while Class 8 suits non-sterile devices requiring contamination control. Conduct a risk assessment per ISO 13485 and consult Health Canada or FDA guidance for your specific device classification and sterility requirements.
How much does it cost to build a cleanroom for medical molding?
A 1,000 sq ft ISO Class 8 modular cleanroom typically costs C$50,000-$120,000, while Class 7 ranges C$120,000-$250,000+. Traditional construction costs 20-40% more. Ongoing operational costs run 15-20% of initial investment annually for utilities, maintenance, and monitoring.
How long does cleanroom setup and certification take?
Modular cleanrooms take 4-6 months total (2-4 months design, 4-12 weeks construction, 2-4 weeks validation), while traditional stick-built construction requires 8-14 months from initial planning to operational certification.
What's the difference between modular and traditional cleanrooms?
Modular cleanrooms use prefabricated panels for faster installation (weeks vs. months), easier reconfiguration, and typically 20-40% lower costs. Traditional stick-built cleanrooms offer greater customization for permanent installations or unusual layouts.
Do I need a cleanroom for all medical device manufacturing?
No. Conduct a risk-based assessment considering device classification (Class I, II, III), patient contact type, and sterility requirements. Low-risk, non-sterile devices may require only controlled environments—not classified cleanrooms.
What maintenance is required to maintain cleanroom certification?
Daily environmental monitoring (particle counts, pressure, temperature, humidity), weekly cleaning, pre-filter changes every 3-6 months, HEPA filter changes every 3-5 years, and annual recertification testing are mandatory. Continuous documentation is required for regulatory compliance.


