
Introduction: Understanding ISO 7 Cleanroom Classification
ISO 7 stands as one of the most widely specified cleanroom classifications across pharmaceutical manufacturing, biotech research, and medical device production.
Its popularity comes from a practical balance: stringent enough to protect sensitive processes, yet economically feasible to build and maintain at scale.
Why ISO 7 matters: Many pharmaceutical manufacturers struggle with contamination risks during non-sterile processing, buffer zone operations, and material preparation. ISO 7 provides the contamination control needed for these critical support areas without the cost intensity of ISO 5 environments.
ISO 6 and ISO 7 cleanrooms represent nearly 47% of the global cleanroom market, driven primarily by pharmaceutical and medical device applications.
This guide covers:
- ISO 14644-1 standard that defines ISO 7
- Technical design requirements and regulatory equivalencies (EU GMP Grade C, FS 209E Class 10,000)
- Industry applications and practical compliance strategies for achieving and maintaining certification
TLDR:
- ISO 7 limits particles to 352,000/m³ (≥0.5 µm), equivalent to FS 209E Class 10,000 and EU GMP Grade C
- Requires 30-60 air changes per hour with HEPA filtration and +10-15 Pa pressure differentials
- Serves as buffer zones for ISO 5 aseptic areas in pharmaceutical manufacturing
- Modular construction reduces costs by 30-40% compared to traditional methods
- Validation includes IQ/OQ/PQ protocols with annual recertification requirements
What is ISO 7 Cleanroom Classification?
Defining ISO 7 by Particle Count Limits
ISO 14644-1:2015 strictly defines ISO 7 based on maximum allowable airborne particle concentrations. The classification sets clear thresholds at three particle size ranges:
- ≥0.5 µm: 352,000 particles/m³ (10,000 particles/ft³)
- ≥1.0 µm: 83,200 particles/m³ (2,356 particles/ft³)
- ≥5.0 µm: 2,930 particles/m³ (83 particles/ft³)
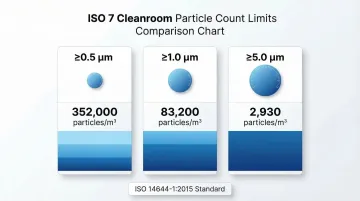
These limits define the maximum contamination allowed when the cleanroom is tested in the "at rest" state (fully operational systems, no personnel present).
Understanding the FS 209E Relationship
ISO 7 directly corresponds to FS 209E Class 10,000, the older US Federal Standard withdrawn in 2001. Despite ISO 14644-1 replacing FS 209E two decades ago, many North American facilities still reference "Class 10,000" in specifications and conversations. Understanding both naming systems remains essential for navigating industry documentation and legacy facility upgrades.
Air Change Rates and Airflow Patterns
ISO 7 environments typically require 30-60 air changes per hour (ACH) to maintain particle count limits through continuous dilution.
Recent ISPE guidance indicates that optimized HVAC systems can achieve compliance at the lower end of this range (20-40 ACH), potentially reducing energy consumption by 36-67% compared to conservative 60+ ACH designs.
Airflow characteristics:
- Non-unidirectional (turbulent) flow is standard for ISO 7, relying on mixing and dilution rather than displacement
- Laminar flow is rarely required except in specific high-risk zones within the ISO 7 space
- Proper air distribution prevents dead zones where particles accumulate
Position in the ISO Hierarchy
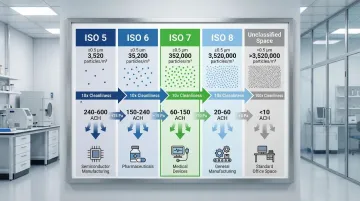
ISO 7 occupies a strategic middle position in the nine-tier ISO classification system:
- 100x cleaner than ISO 8 (3,520,000 particles/m³), which often serves as gowning or ante-room space
- 100x less stringent than ISO 5 (3,520 particles/m³), the standard for aseptic processing zones
- 10x less stringent than ISO 6 (35,200 particles/m³), used for sensitive device assembly
This positioning makes ISO 7 ideal as a buffer or background environment protecting more critical ISO 5 zones from contamination entry.
ISO 7 Cleanroom vs. Other Classifications: Key Comparisons
Particle Count and ACH Comparisons
Understanding how ISO 7 compares to adjacent classifications helps determine the appropriate environment for specific processes:
| ISO Class | Particles/m³ (≥0.5 µm) | ACH Range | FS 209E Equivalent | Typical Applications |
|---|---|---|---|---|
| ISO 5 | 3,520 | 240-480 (unidirectional) | Class 100 | Aseptic filling, critical sterile operations |
| ISO 6 | 35,200 | 90-180 | Class 1,000 | Optical assembly, implantable device manufacturing |
| ISO 7 | 352,000 | 30-60 | Class 10,000 | Buffer rooms, solution prep, compounding |
| ISO 8 | 3,520,000 | 15-25 | Class 100,000 | Gowning rooms, non-sterile packaging |
EU GMP Grade C Equivalency
For pharmaceutical facilities operating under European regulations, ISO 7 corresponds to EU GMP Grade C in the "at rest" state.
This alignment simplifies global compliance strategies, allowing facilities to adopt unified classification approaches across US and EU markets.
Critical distinction: Grade C allows relaxation to ISO 8 levels (3,520,000 particles/m³) during operations, but the environment must demonstrate recovery to ISO 7 within 15-20 minutes after activities stop.
Key operational requirements:
- Recovery time dictates HVAC system capacity
- Air change rates must support rapid particle clearance
- Monitoring systems track real-time recovery performance
ISO 7 as a Buffer Environment
In aseptic pharmaceutical manufacturing, ISO 7 serves a critical protective function:
- Creates a contamination barrier around ISO 5 critical zones
- Maintains pressure differentials that direct airflow outward
- Provides controlled environment for personnel movement and material transfer
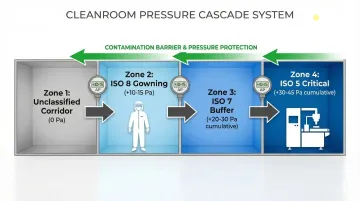
FDA cGMP guidance requires that ISO 5 aseptic processing zones must be located within an ISO 7 background environment. The pressure cascade prevents contamination from entering critical zones:
Typical pressure cascade:
- Unclassified corridor → ISO 8 gowning (+10-15 Pa) → ISO 7 buffer (+10-15 Pa) → ISO 5 critical zone
The graduated pressure increase creates airflow barriers that continuously push air from cleaner to less clean areas, preventing backflow contamination.
Comparison Table: ISO 7 vs. Adjacent Classes
| Factor | ISO 5 | ISO 6 | ISO 7 | ISO 8 |
|---|---|---|---|---|
| Max particles (≥0.5 µm) | 3,520 | 35,200 | 352,000 | 3,520,000 |
| Cleanliness relative to ISO 7 | 100x cleaner | 10x cleaner | Baseline | 10x dirtier |
| Typical ACH | 240-480 | 90-180 | 30-60 | 15-25 |
| Airflow pattern | Unidirectional | Non-unidirectional | Non-unidirectional | Non-unidirectional |
| HEPA ceiling coverage | 80-100% | 25-40% | 15-25% | 5-15% |
| Personnel gowning | Full sterile gown, double gloves | Full coverall, boots, hood | Full coverall, boots, hood | Lab coat or coverall |
ISO 7 Cleanroom Design and Technical Requirements
HVAC System Specifications
HEPA filtration requirements:
- Filters rated H13 (≥99.95% efficiency) or H14 (≥99.995% efficiency) per EN 1822 standard
- Ceiling coverage of 15-25% with filter fan units (FFUs) to achieve necessary air distribution
- Terminal filters require integrity testing every 6-12 months per ISO 14644-3
Pressure differential control:
- Maintain +10 to +15 Pascals (Pa) relative to adjacent lower-classification areas
- EU GMP Annex 1 specifies minimum 10 Pa between different grades
- Continuous monitoring systems required to detect pressure loss events
- Control loops should target +10 Pa to balance compliance with energy efficiency
Air distribution strategies:
- Supply air through ceiling-mounted HEPA filter arrays
- Low-level return air grilles to promote top-to-bottom airflow
- Avoid dead zones through computational fluid dynamics (CFD) modeling during design
- Balance supply and return to maintain precise pressure differentials
Gowning and Airlock Requirements
ISO 7 spaces frequently serve as ante-rooms before personnel enter ISO 5 or ISO 6 zones. Because these areas act as the last barrier before critical environments, proper gowning room design is essential to prevent contamination transfer.
Gowning room features:
- Separate clean and dirty sides with bench or step-over barrier
- Adequate space for donning full coveralls, hoods, boots, and gloves without contamination risk
- Often maintained at ISO 8 classification with step-up to ISO 7 through airlock
Airlock design principles:
- Interlocked doors prevent simultaneous opening
- Adequate volume for personnel or material carts
- Pressure cascade maintained even during door operation
- Air showers optional but not required for ISO 7 entry
Surface Material Requirements
All cleanroom surfaces must be smooth, non-shedding, impervious, and easy to clean. This requirement rules out standard gypsum board, acoustic tiles, and wood finishes commonly used in conventional construction.
Wall systems:
- Epoxy-coated gypsum or modular metal wall panels
- Seamless joints with flush surfaces
- Chemical-resistant to withstand repeated disinfection
Ceiling systems:
- Walkable ceiling grids for maintenance access without contamination risk
- Integrated HEPA filter mounting
- Smooth, sealed surfaces between filter units
Flooring:
- Seamless vinyl sheet or epoxy coating
- Coved corners (4-6 inch radius) eliminating wall-floor joints
- Chemical-resistant to cleaning agents and process chemicals
Modular cleanroom systems increasingly use prefabricated wall panels and walkable ceiling grids that meet these specifications while reducing installation time. This approach allows facilities to achieve cGMP compliance faster and adapt to future process changes.
Environmental Monitoring Requirements
Particle counting protocols:
- ISO 14644-2 mandates risk-based monitoring plans
- Continuous monitoring not strictly required by ISO but common in pharmaceutical applications
- Periodic classification testing at least annually
- Strategic sampling locations based on risk assessment
Viable (microbial) monitoring: For EU GMP Grade C (ISO 7) applications:
- Air samples: 100 cfu/m³ maximum
- Settle plates (4-hour exposure): 50 cfu maximum
- Contact plates: 25 cfu maximum
Temperature and humidity control:
- Temperature: 17-20°C typically specified (lower than standard comfort to compensate for heat generated by full gowning)
- Relative humidity: 30-60% (below 30% risks static electricity; above 60% promotes microbial growth)
- Continuous monitoring with alarmed excursions
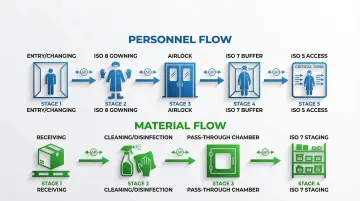
Personnel and Material Flow Patterns
Beyond environmental controls, proper traffic flow patterns are critical to ISO 7 performance. These pathways prevent contamination introduction and maintain the pressure cascades that keep classification integrity intact.
Cascade zoning strategy:
- Unclassified → ISO 8 (gowning) → ISO 7 (buffer) → ISO 5 (critical)
- Each transition through airlocks or pass-through chambers
- Pressure differentials maintained at each interface
Material transfer:
- Pass-through chambers with interlocked doors
- Cleaning and disinfection protocols before material entry
- Separate flows for raw materials vs. finished products where possible
Effective ISO 7 designs integrate airlock placement, pass-through chambers, and material flows early in the planning process. This ensures the layout supports both current operations and future workflow adaptations without compromising contamination control.
ISO 7 Cleanroom Applications Across Industries
Pharmaceutical Manufacturing
ISO 7 environments support multiple pharmaceutical operations:
- Surrounding ISO 5 biological safety cabinets for sterile compounding per USP <797> and <800> requirements
- Final packaging operations for tablets, capsules, and other solid dosage forms
- Preparation of culture media and buffers before sterilization
- Washing and staging of containers, closures, and equipment before introduction to aseptic zones
Medical Device Manufacturing
Medical device production uses ISO 7 for controlling bioburden before terminal sterilization:
- Assembly of non-implantable devices like surgical instruments, diagnostic equipment, and Class II devices
- Final packaging of sterile devices after terminal sterilization
- Microbiological testing and inspection areas requiring contamination control
Biotech and Research Applications
Biotechnology facilities rely on ISO 7 for sensitive but non-critical operations:
- Protein purification and formulation after initial fermentation
- Growing cell lines for research or therapeutic applications
- Research spaces requiring contamination control for reproducible results
Industry-Specific Solutions
ACH Engineering provides ISO 7 modular cleanroom solutions for pharmaceutical manufacturers, compounding pharmacies, biotech companies, medical device producers, and research laboratories across North America.
These modular systems meet industry-specific regulatory requirements including USP standards for pharmaceutical applications and FDA requirements for medical device manufacturing.
Compliance, Validation, and Ongoing Maintenance
Validation Process: IQ, OQ, PQ
ISO 7 cleanrooms must undergo formal qualification following the V-model validation approach:
Installation Qualification (IQ):
- Verifies equipment installed per design specifications
- Documents HEPA filter installation and leak testing
- Confirms surface finishes meet non-shedding requirements
- Validates pressure differential sensors and control systems
Operational Qualification (OQ):
- Tests cleanroom "at rest" (systems running, no personnel)
- Measures airflow velocity and volume at each supply point
- Verifies pressure differentials across all interfaces
- Conducts particle count testing to confirm ISO 7 classification
- Documents recovery time (return to specification after disturbance)
Performance Qualification (PQ):
- Tests cleanroom "in operation" with personnel and equipment
- Verifies ISO 7 maintained during actual manufacturing activities
- Validates gowning procedures and personnel flow patterns
- Confirms environmental monitoring systems function correctly
Ongoing Compliance Requirements
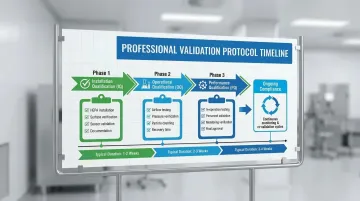
Once validated, maintaining ISO 7 classification requires consistent monitoring and documentation.
Routine testing frequencies include:
- Particle count testing: Quarterly or semi-annual based on risk
- HEPA filter integrity testing: Annually (6-12 months per WHO)
- Pressure differential monitoring: Continuous with alarms
- Airflow volume verification: Annual recertification
- Microbial monitoring: Continuous or per batch (pharmaceutical)
Documentation protocols require:
- Written procedures for cleaning, maintenance, and sanitization (FDA cGMP 21 CFR 211.42)
- Records demonstrating consistent ISO 7 under dynamic conditions
- Deviation investigations for readings outside specification
- Annual summary reports showing environmental trends
Regulatory Frameworks Referencing ISO 7
FDA cGMP references ISO 14644-1 for cleanroom classification and requires ISO 7 background environments for ISO 5 aseptic zones.
EU GMP Annex 1 defines Grade C (ISO 7 at rest) for pharmaceutical sterile manufacturing. The updated 2022 version strengthens contamination control requirements.
USP <797>/<800> requires buffer rooms for sterile compounding to meet at least ISO 7, while <800> adds requirements for hazardous drug handling with negative pressure.
Common Challenges and Best Practices for ISO 7 Cleanrooms
Design Mistakes to Avoid
Inadequate air change rates:
- Defaulting to minimum ACH without validating recovery time
- Design for 40-50 ACH initially, then validate if reduction to 30 ACH maintains compliance
Improper pressure cascade:
- Pressure reversals due to unbalanced supply/return or door operation
- Install continuous pressure monitoring with alarms and size airlocks adequately for pressure recovery
Insufficient gowning space:
- Cramped gowning rooms increase contamination risk from personnel contact with surfaces
- Provide minimum 40-50 square feet per gowning station with clear separation of clean/dirty sides
Poor filter placement:
- Dead zones where air circulation is inadequate
- Use CFD modeling during design to optimize filter and return grille locations
Advantages of Modular Construction
Once construction is complete, maintaining compliance requires ongoing attention to four critical areas.
Proper gowning procedures:
- Personnel represent the largest contamination source
- Validate gowning procedures and train all personnel before cleanroom access
- Regular competency assessments and retraining
Regular maintenance schedules:
- Preventive maintenance for HVAC systems, filters, and monitoring equipment
- Documented cleaning procedures following strictly controlled SOPs
- Equipment calibration and verification on defined schedules
Staff training programs:
- Initial qualification training for all cleanroom personnel
- Ongoing education on contamination control principles
- Refresher training at least annually
Environmental monitoring best practices:
- Trend analysis to detect gradual degradation before failures occur
- Investigation thresholds set below action limits to enable proactive response
- Regular review of monitoring data by quality assurance
Frequently Asked Questions
What are the requirements for an ISO 7 cleanroom?
ISO 7 cleanrooms limit particles to 352,000/m³ (≥0.5 µm) through 30-60 air changes per hour with HEPA filtration, +10-15 Pa pressure differential, and continuous environmental monitoring including particle counting and microbial sampling.
How does ISO 7 compare to ISO 5, ISO 8, Class 10,000, and Grade C?
ISO 7 is 100x cleaner than ISO 8 and 100x less stringent than ISO 5, equivalent to Class 10,000 and EU GMP Grade C at rest. Unlike ISO 5's unidirectional airflow, ISO 7 uses turbulent mixing ventilation.
What industries commonly use ISO 7 cleanrooms?
Pharmaceutical manufacturing, medical device production, biotech research, compounding pharmacies (USP <797>/<800> compliance), and electronics manufacturing use ISO 7 for contamination-sensitive operations like sterile compounding, assembly, and cell culture.
How much does it cost to build an ISO 7 cleanroom?
Costs range from $250-350 CAD/ft² for modular construction to $350-500 CAD/ft² for traditional stick-built methods, with project size, complexity, and location significantly impacting final expenses.
What is the difference between ISO 7 and USP 797 buffer room requirements?
USP <797> requires buffer rooms to meet ISO 7 classification minimum per ISO 14644-1. USP <800> adds hazardous drug handling requirements, including negative pressure relative to adjacent areas.
How often should ISO 7 cleanrooms be tested and validated?
Initial validation follows IQ/OQ/PQ protocols. Ongoing monitoring includes particle counting quarterly or semi-annually, annual HEPA filter testing, and continuous pressure monitoring, with complete requalification every 12 months for EU GMP Grade C.


