
Introduction
In pharmaceutical manufacturing, even microscopic contamination can compromise product safety, trigger costly batch failures, and result in serious regulatory violations. Many facilities experience contamination events despite having cleaning procedures in place. The gap usually comes down to inconsistencies in cleanroom classification, cleaning agents, technique, frequency, and validation protocols.
The regulatory pressure is real. Health Canada's GMP guidelines — mirrored by FDA requirements for facilities with cross-border operations — require that ISO Class 5 areas yield no detectable microbiological contaminants under aseptic conditions. Inspection findings routinely flag the same failures: inadequate cleaning validation, non-sterile agents used in critical zones, and gaps in environmental monitoring.
Getting it right requires more than a checklist. This guide covers:
- Cleanroom classifications and how they shape cleaning requirements
- Step-by-step cleaning procedures for pharmaceutical environments
- Essential requirements: validated disinfectants and trained personnel
- Critical parameters that determine cleaning effectiveness
- Common mistakes that lead to contamination events
- Troubleshooting strategies for persistent problems
TL;DR
- Validated cleanroom cleaning combines physical removal of contaminants first, then disinfection to eliminate microorganisms
- Protocols must match ISO Class 5–8 classification — stricter areas need sterile materials, sporicidal agents, and higher frequency
- Rotate disinfectants, follow validated contact times, wear correct PPE, and document everything
- Most failures trace back to skipping cleaning before disinfection, wrong dilutions, or unvalidated GMP procedures
- ISO Class 5 areas require sterile water for disinfectant dilution to avoid introducing bioburden
Understanding Pharmaceutical Cleanroom Classifications
Pharmaceutical cleanrooms are classified by ISO 14644-1:2015 standards based on maximum allowable airborne particles per cubic meter, and ACH Engineering's pharmaceutical cleanroom services cover the full design-to-validation lifecycle. ISO Class 5 represents the cleanest environment for sterile product exposure, while Class 8 is suitable for less critical areas like change rooms.
Key Classification Standards:
| ISO Class | 0.5 µm particles/m³ | 1.0 µm particles/m³ | Pharmaceutical Use |
|---|---|---|---|
| ISO 5 | 3,520 | 832 | Sterile compounding, aseptic filling |
| ISO 6 | 35,200 | 8,320 | Background environment for ISO 5 |
| ISO 7 | 352,000 | 83,200 | Non-sterile compounding, packaging |
| ISO 8 | 3,520,000 | 832,000 | Warehousing, gowning rooms |
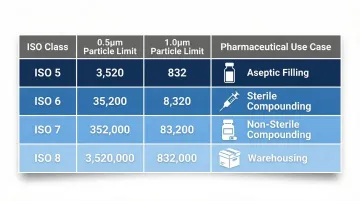
Classification determines cleaning requirements directly. ISO Class 5 areas demand sterile cleaning materials, daily protocols, and sporicidal agents. FDA guidance explicitly states that ISO Class 5 areas should normally yield no microbiological contaminants, establishing zero tolerance for cleaning failures.
FDA, Health Canada, and EU GMP Annex 1 all link air quality classifications to specific cleaning and disinfection requirements. Your facility's ISO class determines not just how often you clean, but which agents, materials, and verification methods are acceptable — making classification the logical starting point for any cleaning program.
Step-by-Step Cleanroom Cleaning Process
Step 1: Preparation and Safety Setup
Don appropriate PPE matching the cleanroom class: sterile coveralls for ISO 5, gowns for ISO 6-7, with bouffant caps, masks, sterile gloves, and shoe covers. Humans are the largest contamination source in any cleanroom—proper gowning is non-negotiable.
Gather validated cleaning materials:
- Lint-free wipes or mops (polyester or microfiber for ISO 5)
- Approved cleaning solutions at proper dilutions
- Sterile water for ISO 5 areas (USP <797> mandates sterile water for irrigation)
- Graduated cylinders for accurate dilution
- Labeled spray bottles with preparation and expiration dates
Review cleaning logs to confirm previous completion and check for contamination alerts requiring special attention.
Step 2: Remove Visible Contamination (Cleaning Phase)
Begin by removing gross contamination using dry methods: sweep or vacuum with HEPA-filtered equipment, or use tacky rollers on floors to capture particles before wet cleaning.
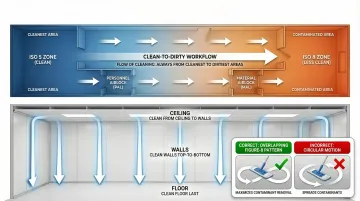
Apply detergent-based cleaning solution to all surfaces using lint-free wipes or mops. Work from cleanest to dirtiest areas (ISO 5 to ISO 8) and top to bottom (ceilings to floors). This directional approach prevents cross-contamination and particle redistribution.
Use an overlapping figure-8 pattern when mopping or unidirectional strokes when wiping to ensure complete coverage. Change wipes or mop heads every 600 ft² for ISO 5/6 areas, every 1,000 ft² for ISO 7/8. Reusing a contaminated mop head redistributes bioburden across surfaces you've already cleaned—undoing the work entirely.
Critical point: Applying disinfectant to visibly soiled surfaces is ineffective because organic matter shields microorganisms from antimicrobial action. Always clean first.
Step 3: Apply Disinfectant (Sanitization Phase)
After cleaning, apply the validated disinfectant until all surfaces are fully saturated. Common pharmaceutical disinfectants include:
- 70% Isopropyl Alcohol (IPA): Fast-drying, bactericidal and fungicidal, but not sporicidal. Must be sterile in ISO 5 areas.
- Sodium Hypochlorite (0.5% bleach): Sporicidal with 10-60 minute contact time. Corrosive to stainless steel; requires thorough rinsing.
- Hydrogen Peroxide (3-6%): Sporicidal at higher concentrations with good material compatibility.
- Quaternary Ammonium Compounds: Bactericidal and fungicidal but non-sporicidal. Good cleaning properties.
Maintain the required contact time specified in your SOP—typically 5-10 minutes depending on the product. Surfaces must remain visibly wet for the entire contact time. If alcohol evaporates before that window closes, disinfection has failed and the surface must be re-treated.
For ISO Class 5 areas, follow with a sterile water rinse if the disinfectant requires it. Allow to air dry or wipe down with sterile lint-free wipes before returning the area to service.
Step 4: Documentation and Verification
Complete cleaning logs documenting:
- Date and time
- Areas cleaned
- Cleaning agents used with lot numbers
- Personnel performing the work
- Any observations or deviations
Once logs are complete, run through this verification sequence before releasing the area:
- Visual inspection — Check all surfaces for residues, streaks, or missed areas and perform touch-up cleaning as needed.
- Environmental monitoring — Collect settle plates, contact plates, and air samples per your monitoring schedule.
- Log sign-off — Have a second qualified person review and countersign the cleaning record.
Gaps in any of these steps are among the first things FDA or Health Canada auditors flag during a site inspection.
When Should You Clean Your Pharmaceutical Cleanroom
Cleaning frequency is determined by cleanroom classification, production schedule, and regulatory requirements.
Cleaning Schedules by ISO Class
ISO Class 5 areas:
- At the start of each shift
- Before each batch or compounding session
- Every 30 minutes during continuous compounding
- At day's end
- After any spill or suspected contamination
Supporting areas (ISO 6–8):
- Daily: Counters, work surfaces, and floors
- Weekly: Deep cleaning of walls and equipment
- Monthly: Ceilings, storage shelving, and sporicidal treatment
Trigger Events Requiring Immediate Cleaning
Some situations require cleaning outside the regular schedule:
- Product spills or equipment maintenance
- Failed environmental monitoring results
- Suspected contamination events
- Extended periods of non-use (rooms must be re-cleaned and re-certified before resuming production)
- Environmental data indicating spore-forming organisms
USP <797> requires validated, documented cleaning schedules followed consistently. Document and justify any deviations from the schedule.
With your schedule established, the next step is understanding which cleaning agents and methods apply to each area.
Essential Requirements for Effective Cleaning
Successful cleanroom cleaning depends on having the right materials, properly trained personnel, and validated procedures working together as a system.
Cleaning Agents and Disinfectants
Understanding the difference between cleaning agents and disinfectants is fundamental. Cleaning agents (detergents) remove soil, particles, and organic matter. Disinfectants are antimicrobial agents that kill microorganisms. Cleaning must always precede disinfection—organic matter can neutralize disinfectant activity.
Disinfectant Rotation Strategy:
Current best practice calls for routine use of a broad-spectrum disinfectant augmented by periodic sporicidal treatment, with frequency driven by environmental monitoring data rather than a fixed calendar schedule.
Use at least three disinfectants with distinct mechanisms of action:
- Daily/routine: Broad-spectrum agent (70% IPA or quaternary ammonium)
- Weekly/monthly: Different broad-spectrum agent (hydrogen peroxide)
- Monthly: Sporicidal agent (sodium hypochlorite or high-concentration hydrogen peroxide)
This approach prevents the development of resistant organisms and targets the full range of microbes typically found in pharmaceutical environments.
Approved Disinfectants with Specifications:
| Agent | Concentration | Contact Time | Activity | Limitations |
|---|---|---|---|---|
| 70% IPA | 70% | 30s-1 min | Bactericidal, fungicidal, virucidal | Not sporicidal; evaporates quickly |
| Sodium Hypochlorite | 0.5% (5000 ppm) | 10-60 min | Sporicidal, bactericidal, virucidal | Corrosive; unstable (24-hour shelf life) |
| Hydrogen Peroxide | 3-6% | 10-60 min | Sporicidal (high conc.), bactericidal | Requires proper storage |
| Quaternary Ammonium | 0.1-2% | 10 min | Bactericidal, fungicidal | Not sporicidal; can leave residues |

Cleaning Tools and Equipment
All cleaning tools must be lint-free, dedicated to specific cleanroom classes, and made of materials compatible with disinfectants.
Required equipment:
- Polyester or microfiber wipes for ISO 5 areas
- Autoclavable mop heads and handles for floors
- Bucketless mop systems to reduce contamination risk
- Graduated cylinders for accurate dilution
- Spray bottles labeled with preparation date, expiration date, and lot numbers
- Separate tools for floors versus walls/ceilings
Clean, sanitize, and store all equipment properly after each use. In ISO 5 areas, opt for disposable items wherever practical — this eliminates the recontamination risk that comes with reusing tools.
Trained Personnel and Validated Procedures
Cleaning staff must receive comprehensive training before performing cleanroom cleaning:
- Follow gowning procedures specific to each cleanroom class
- Apply correct wiping patterns and directional techniques to avoid cross-contamination
- Prepare and dilute disinfectants accurately per approved specifications
- Observe required contact times — cutting these short compromises microbial kill
- Handle chemicals safely, including spill response and PPE use
- Complete documentation records per GMP requirements
Both FDA 21 CFR Part 211.67 and Health Canada's Good Manufacturing Practices (GUI-0001) require written cleaning procedures that must be established, followed, and documented with records maintained per GMP standards.
All cleaning procedures must be validated to demonstrate they consistently achieve acceptable levels of cleanliness and microbial control. Validation includes worst-case scenarios and requires periodic revalidation when procedures, materials, or facility bioburden changes.
Key Parameters and Common Mistakes Affecting Cleaning Effectiveness
Even with proper materials and training, cleaning can fail if critical parameters are not controlled or common mistakes are made.
Disinfectant Contact Time and Concentration
Disinfectants require specific contact times (the time a surface remains wet) to be effective — typically 5–10 minutes depending on the product. If surfaces dry before contact time ends, disinfection fails.
Alcohol presents a particular challenge because it evaporates rapidly. To achieve proper contact time, surfaces may need to be re-wetted multiple times during the required period.
Improper dilution is a frequent failure point:
- Too concentrated: damages surfaces, leaves residues, and creates safety hazards
- Too dilute: fails to kill microorganisms and wastes time and money
The concentration exponent determines how rapidly efficacy drops with dilution. Phenolic disinfectants have a concentration exponent of 6, meaning a small dilution error causes massive potency loss. Use automated dilution systems or test strips to verify concentrations regularly.
Water Quality and Temperature
USP <797> explicitly mandates sterile water for irrigation or injection when diluting disinfectants in ISO Class 5 areas. Using non-sterile water introduces bioburden during the cleaning process, reintroducing contamination during the process.
For ISO 6–8 areas, Purified Water, USP is generally acceptable provided it meets microbial limits.
Water temperature affects both disinfectant efficacy and cleaning agent performance. Higher temperatures increase antimicrobial activity but can also accelerate evaporation and degradation of active ingredients. Always follow manufacturer specifications for the optimal range.
Surface Material Compatibility
Certain disinfectants can damage cleanroom materials over time, compromising surface integrity and creating refuges for microbes.
Common compatibility issues:
- Sodium hypochlorite on 304L stainless steel causes pitting and corrosion — thorough rinsing after application is essential
- Repeated alcohol use can crack acrylics and polycarbonate surfaces
- Phenolics leave sticky residues that are difficult to remove and may cause skin irritation
Validate all cleaning agents against your cleanroom's surface materials — stainless steel, epoxy floors, acrylic windows, and wall panels — before implementation. When selecting or upgrading cleanroom systems, confirm that materials like HPL panels, powder-coated galvanized iron, and u-PVC are rated for repeated chemical exposure.
Cleaning Direction and Technique
The critical rule: clean from clean to dirty (ISO 5 to ISO 8) and top to bottom (ceiling to floor). This prevents cross-contamination and particle redistribution.
Proper technique includes:
- Overlapping figure-8 patterns for mopping
- Unidirectional strokes for wiping (pull and lift, don't spread)
- Frequent wipe/mop changes
- Avoiding circular motions that spread rather than remove contamination
Skipping the Cleaning Step Before Disinfection
Applying disinfectant to visibly soiled surfaces is ineffective because organic matter, oils, and particles shield microorganisms from antimicrobial action. Sodium hypochlorite is particularly susceptible to inactivation by organic material.
When staff skip cleaning to save time, they reduce efficacy and waste expensive disinfectants — without achieving the microbial control they assumed they had. The clean-then-disinfect sequence cannot be shortened.
Using Expired or Improperly Stored Cleaning Solutions
Diluted cleaning solutions have limited shelf life:
- Diluted bleach (0.5%): 24 hours maximum
- Diluted bleach (higher concentration): up to 14 days
- Diluted alcohol: approximately 1 year, but evaporation alters concentration
- Commercial bleach containers: dispose 1 year after manufacture
Using expired solutions provides no microbial control while giving false confidence. Improper storage — wrong temperature, light exposure, or contaminated containers — degrades disinfectants further.
All solutions must be labeled with preparation date, expiration date, and lot numbers. This documentation is required during regulatory inspections.
Troubleshooting and Advanced Cleaning Solutions
Even well-designed cleaning programs encounter challenges such as persistent contamination, failed environmental monitoring, or difficulty cleaning complex equipment.
Problem: Recurring Microbial Contamination Despite Following Protocols
Likely causes:
- Resistant microorganisms due to lack of disinfectant rotation
- Inadequate contact time (surfaces drying too quickly)
- Biofilm formation on surfaces
- Contamination sources outside cleaning scope (HVAC, personnel, incoming materials)
Recommended solutions:
- Implement a triple-clean protocol: clean with detergent, disinfect with a broad-spectrum agent, apply a sporicidal treatment, then disinfect with a different agent. This sequence breaks down biofilms and eliminates resistant organisms.
- Increase environmental monitoring frequency to pinpoint contamination sources — sample HVAC diffusers, door handles, and high-touch surfaces.
- Retrain personnel on technique: observe actual cleaning procedures against SOPs. Even small deviations can compromise effectiveness.
- Assess facility design: persistent contamination may signal root-cause issues like inadequate airflow, difficult-to-clean surfaces, or inappropriate materials.

Problem: Difficulty Cleaning Hard-to-Reach Areas or Complex Equipment
Standard mops and wipes cannot effectively clean inside equipment, tight corners, or overhead areas, leading to contamination reservoirs.
Specialized tools and approaches:
- Isolator cleaning wands for reaching inside equipment
- Extension poles for ceiling access
- Small foam swabs for crevices and joints
- Walkable ceiling systems that provide safe maintenance access
Modern modular cleanroom designs address this proactively. ACH Engineering's walkable ceiling systems eliminate contamination risks by allowing personnel to safely access overhead areas for thorough cleaning and maintenance without introducing contaminants into the space below.
Cleanroom coving provides seamless wall-ceiling and wall-floor connections that eliminate sharp corners where contaminants accumulate. These GMP-compliant design features make thorough cleaning practical, not just planned.
When to Consider Professional Cleanroom Design or Upgrades
Certain situations indicate that cleaning challenges stem from facility design issues rather than procedural failures:
- Persistent contamination despite proper protocols and aggressive remediation
- Excessive cleaning time impacting production schedules
- Difficulty maintaining cleanroom classification between monitoring periods
- Upcoming regulatory inspections revealing design deficiencies
- Frequent equipment cleaning failures or hard-to-reach areas
Modern modular cleanroom systems build cleanability into the design phase — smooth surfaces, minimal crevices, and layouts that support efficient workflows. ACH Engineering's turnkey cleanroom solutions factor in cleaning protocols, material compatibility, and maintenance access from day one, with modular systems that can be expanded or reconfigured as production needs change.
Advanced Approaches: Automated and Technology-Enhanced Cleaning
Emerging technologies supplement manual cleaning but do not replace validated procedures.
UV-C Disinfection: UV-C light (254 nm) disrupts microbial DNA, achieving >1 log reduction for bacteria and viruses. However, UV-C is a "line-of-sight" technology—shadowed areas receive no disinfection. It does not remove physical soil and must be used as an adjunct to manual cleaning.
Hydrogen Peroxide Vapor (HPV): HPV systems achieve >6 log reduction of bacterial spores, making them highly effective for terminal decontamination. Applications include room decontamination and isolator sterilization. Limitations include long cycle times (1.5-8 hours) and the requirement for sealed HVAC systems. Like UV-C, HPV does not remove dust or stains—manual cleaning must precede vapor treatment.
Real-Time Particle Monitoring: Continuous monitoring of non-viable particles (0.5 µm and 5.0 µm) provides immediate feedback on cleaning effectiveness and can detect contamination events as they occur. EU Annex 1 requires continuous monitoring for Grade A zones to capture transient events that periodic sampling would miss.
While these technologies enhance contamination control, they cannot replace the need for proper manual cleaning protocols, trained personnel, and validated procedures.
Conclusion
Effective pharmaceutical cleanroom cleaning comes down to five fundamentals:
- Understanding cleanroom classifications and their contamination thresholds
- Following validated, step-by-step cleaning and disinfection procedures
- Using appropriate materials and correctly prepared disinfectants
- Controlling critical parameters such as contact time, concentration, and coverage
- Maintaining thorough, audit-ready documentation
Most cleaning failures stem from preventable causes: inadequate training, rushing through procedures, improper disinfectant preparation or application, or failing to validate and monitor effectiveness. The two-step process of cleaning followed by disinfection is fundamental—skipping cleaning renders disinfection ineffective .
Investing in proper cleaning protocols, quality materials, and staff training protects product quality and keeps your facility in regulatory compliance. When cleaning challenges persist despite sound procedures, the root cause is often the facility itself.
Design improvements—seamless surfaces, accessible ceiling systems, appropriate wall and floor materials—can solve problems that procedural changes alone cannot. Modular cleanroom specialists like ACH Engineering design controlled environments with cleanability built in from the start, reducing the ongoing burden on cleaning teams and minimizing contamination risk at the source.
Frequently Asked Questions
Frequently Asked Questions
How to clean a pharmacy clean room?
Pharmacy cleanrooms follow USP <797> requirements: don appropriate sterile PPE, clean surfaces with detergent to remove particles and organic matter, then apply disinfectant (70% sterile IPA or 0.5% bleach) with adequate contact time. Document all activities with dates and lot numbers, and rotate disinfectants monthly with a sporicidal agent to prevent resistance.
What are the GMP guidelines for clean rooms?
FDA 21 CFR Part 211.67 requires written procedures, validated cleaning methods, documented schedules, and trained personnel with documented competency. EU GMP Annex 1 adds that cleaning must precede disinfection — organic surface contamination can inactivate disinfectants before they reach microorganisms.
How often should pharmaceutical cleanrooms be cleaned?
ISO Class 5 areas require cleaning at shift start, before each production run, every 30 minutes during continuous compounding, and immediately after spills. Supporting areas need daily surface cleaning, weekly deep cleaning of walls and equipment, and monthly sporicidal treatments. Exact frequencies must be validated for each facility using environmental monitoring data.
What is the difference between cleaning and disinfection in cleanrooms?
Cleaning removes visible dirt, particles, and organic matter using detergents and physically removes contaminants from surfaces. Disinfection kills microorganisms using antimicrobial agents like alcohol, bleach, or hydrogen peroxide. Cleaning must always be performed before disinfection because organic matter shields microorganisms from disinfectants and can neutralize their antimicrobial activity.
What are the most common cleanroom cleaning mistakes?
The most frequent mistakes are skipping cleaning before disinfecting, incorrect disinfectant dilution, and insufficient contact time (surfaces drying before the required dwell period). Other common failures include using expired solutions, neglecting disinfectant rotation, and inadequate documentation that cannot demonstrate compliance during an inspection.
Do I need to validate my cleanroom cleaning procedures?
Yes. FDA GMP regulations require validation of all cleaning procedures to demonstrate consistent removal of contaminants and prevention of cross-contamination. Studies must cover efficacy testing against standard and facility-specific organisms, worst-case scenarios, and in-situ environmental monitoring verification. Revalidation is required whenever procedures, materials, or facility bioburden change significantly.


