
Introduction
Maintaining a clean ISO Class 7 environment is one of the most operationally demanding — and consequential — responsibilities in pharmaceutical, biotech, and medical device facilities. These cleanrooms hold to a strict standard of ≤352,000 particles (≥0.5 microns) per cubic meter, functioning as buffer areas that protect sensitive processes from contamination while supporting sterile operations in adjacent ISO Class 5 zones.
This guide covers the cleaning procedures, agent selection, frequency requirements, and documentation practices that keep ISO Class 7 environments compliant and operational.
Inadequate cleaning procedures carry serious financial and regulatory consequences. Contamination events cost the pharmaceutical industry between $2.5 billion and $5 billion annually in product recalls and lost sales.
Failed compliance audits trigger FDA warning letters, facility shutdowns, and regulatory penalties. In one documented case, cleanroom contamination resulted in $18.2 million in criminal and civil penalties. Remediation costs escalate quickly: from $100,000 for minor incidents to over $2 billion for systemic failures requiring consent decrees.
TL;DR
- ISO Class 7 cleanrooms must maintain ≤352,000 particles (≥0.5 µm) per cubic meter through systematic cleaning
- Use only Health Canada- or EPA-approved disinfectants and cleanroom-grade low-lint supplies, with all personnel in full PPE
- Follow clean-to-dirty and top-to-bottom cleaning patterns with documented procedures and proper contact times
- Daily disinfection of work surfaces and floors, weekly deep cleaning, monthly comprehensive sanitization
- Log all cleaning activities and retain records for 3+ years per regulatory requirements
Why Proper Cleaning is Critical for ISO Class 7 Cleanrooms
ISO Class 7 cleanrooms function as buffer areas supporting sterile compounding, pharmaceutical packaging, device assembly, and biotechnology processes. These controlled environments bridge the gap between general manufacturing spaces and critical ISO Class 5 zones, making contamination control essential for product safety and regulatory compliance.
Direct Impact on Particle Counts
Cleaning procedures directly determine whether facilities maintain classification standards. Improper cleaning introduces contaminants through:
- Non-compliant materials that shed particles
- Incorrect techniques that redistribute rather than remove contamination
- Inadequate disinfection that allows microbial growth
- Cross-contamination from dirty-to-clean cleaning patterns
Effective cleaning protocols actively reduce particle counts, prevent microbial proliferation, and maintain the controlled environment necessary for sensitive operations.
Regulatory Compliance Requirements
Multiple regulatory frameworks mandate documented cleaning procedures:
- USP <797>: Requires specific cleaning frequencies and documentation for sterile compounding areas
- USP <800>: Mandates hazardous drug handling protocols including specialized cleaning procedures
- FDA cGMP (21 CFR 211): Requires written equipment cleaning records and validated procedures
- ISO 14644-5: Establishes operational standards for cleanroom maintenance and cleaning
Non-compliance results in FDA warning letters, failed inspections, operational shutdowns, and potential product recalls. Facilities must demonstrate both that cleaning occurred and that procedures effectively maintain classification standards.
Cost Analysis: Prevention vs. Remediation
Preventive cleaning programs cost a fraction of remediation, which ranges from $100,000 for minor contamination events to over $2 billion for systemic failures. That 10-100x cost differential makes validated cleaning procedures an essential risk management investment.
Cleanroom Design Impact on Cleaning Efficiency
Facility design directly affects how easy — or difficult — a cleanroom is to clean. ACH Engineering builds modular cleanrooms — with designs that address cleanroom particle contamination management — using seamless coving, flush-mounted doors, and accessible ceiling systems specifically to reduce particle accumulation points and cut cleaning time.
These design choices improve contamination control outcomes compared to traditional construction, without adding complexity to daily maintenance routines.
ISO Class 7 Cleanroom Cleaning Standards & Requirements
Particle Count Specifications
ISO 14644-1:2015 defines ISO Class 7 as maintaining a maximum of 352,000 particles ≥0.5 microns per cubic meter under at-rest conditions. Cleaning procedures directly impact these counts by removing particle-generating contaminants before they become airborne.
Regulatory Framework
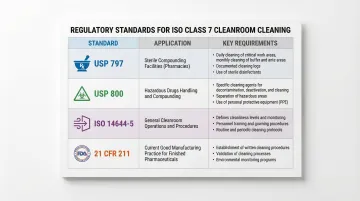
Multiple standards govern ISO Class 7 cleaning:
| Standard | Application | Key Requirements |
|---|---|---|
| USP <797> | Sterile compounding facilities | Daily floor/counter cleaning, monthly wall/ceiling disinfection |
| USP <800> | Hazardous drug handling | Sporicidal agent use, specialized PPE for cleaning personnel |
| ISO 14644-5 | Cleanroom operations | Validated cleaning procedures, material specifications |
| 21 CFR 211 | Pharmaceutical manufacturing | Written cleaning records, equipment logs, validated procedures |
Understanding occupancy states matters here. ISO 14644-1 distinguishes three conditions under which a cleanroom may be classified:
- As-built: Facility complete with services connected, no equipment or personnel present
- At-rest: Equipment installed and operating, no personnel present
- Operational: Normal functioning with personnel actively working
FDA guidance requires classification to prioritize data from operational conditions. Cleaning schedules must account for contamination generated during actual use — not just maintain cleanliness in an empty room.
Personnel Training Requirements
Because contamination risk is highest under operational conditions, who performs the cleaning matters as much as how it's done. Only staff with documented competency should clean ISO Class 7 spaces. Training programs must cover:
- Proper gowning sequences and PPE use
- Cleaning technique execution (directional patterns, contact times)
- Chemical handling and safety protocols
- Documentation requirements and deviation reporting
- Contamination risks and prevention strategies
Documentation Standards
21 CFR 211.180 requires record retention for at least 1 year after batch expiration, while USP <797> mandates 3-year retention for sterile compounding records. All cleaning activities must be logged with:
- Date and time of cleaning
- Personnel performing the work
- Products used (including lot numbers)
- Surfaces cleaned
- Any deviations encountered
- Verification signatures
Essential Cleaning Supplies & Equipment for ISO Class 7
Approved Disinfectant Chemistries
All disinfectants used in North American cleanrooms must carry the appropriate regulatory registration — EPA registration numbers in the U.S. or Health Canada DIN numbers in Canada. Approved chemistries for ISO Class 7 include:
- Accelerated Hydrogen Peroxide (AHP): Fast-acting and broad-spectrum with minimal surface residue
- Sodium Hypochlorite (Bleach): Effective sporicidal agent for monthly deep cleaning
- Isopropyl Alcohol (70%): Rapid disinfection for frequently touched surfaces
- Quaternary Ammonium Compounds: Daily surface disinfection with residual activity
- Phenolics: Broad-spectrum activity for environmental surfaces
- Iodophors: Effective against bacteria, viruses, and fungi
Critical requirement: Confirm the appropriate registration number before use. Products must be used according to labelled contact times — if a label specifies 10 minutes, surfaces must remain visibly wet for the full duration.
Cleanroom-Grade Tools and Materials
Selecting the right disinfectant is only half the equation — the tools used to apply it matter just as much. IEST-RP-CC004 establishes standards for cleanroom wiping materials, requiring evaluation for particle generation, extractables, and absorption rates.
Required cleaning tools:
- Sterile or low-lint microfiber wipes, preferably pre-saturated for consistent disinfectant concentration
- Polyester mop heads and handles — non-shedding and dedicated exclusively to the cleanroom
- Cleanroom-grade buckets: Non-particulating containers for solution preparation
- HEPA-filtered vacuums — HEPA or ULPA exhaust filters are required per IEST-RP-CC044 to prevent particle re-contamination during cleaning
- Sterile water for irrigation in any ISO Class 5 areas nested within the Class 7 space
Personal Protective Equipment (PPE)
With the right tools in hand, the people using them need equal attention. 21 CFR 211.28 mandates appropriate protective apparel for cleanroom personnel. Unlike ISO Class 5 or stricter environments, Class 7 typically allows single-layer gowning — though hazardous drug handling areas require additional protection:
- Disposable cleanroom gloves (double-gloved for hazardous drug areas)
- Lint-free shoe covers
- Bouffant caps (beard covers if applicable)
- Face masks
- Safety glasses
- Cleanroom gowns or frocks
Donning sequence: Shoe covers first, then gown, bouffant, mask, and gloves last.

Step-by-Step ISO Class 7 Cleaning Procedures
Preparation Phase
Proper preparation prevents contamination from entering the cleanroom before cleaning even begins. Complete both steps below before crossing the gowning threshold.
Don PPE in this exact sequence — order matters for contamination control:
- Remove outer garments and jewelry
- Don shoe covers first
- Put on cleanroom gown or frock
- Secure bouffant cap (and beard cover if needed)
- Position face mask properly
- Put on safety glasses
- Don cleanroom gloves last
Once garbed, prepare your cleaning supplies outside the cleanroom before entry:
- Gather all pre-approved cleaning supplies outside the cleanroom
- Prepare cleaning solutions per manufacturer's instructions
- Use sterile water for critical ISO Class 5 surfaces
- Document solution preparation time and concentration
- Verify all materials are within expiration dates
Pre-Cleaning Phase
Before applying any disinfectants, remove loose contamination sources. Skipping this step means disinfectants will be working against a particle load — reducing their efficacy.
- Remove all portable equipment and materials from the cleanroom
- Dispose of waste in designated receptacles
- Use a HEPA vacuum to remove loose particles
- Start from highest points (ceiling, light fixtures)
- Work systematically downward to avoid re-contaminating cleaned surfaces
With loose particles cleared, the environment is ready for systematic disinfection.
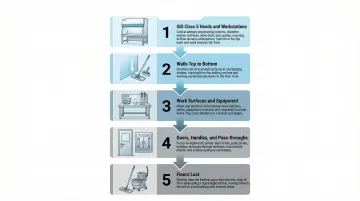
Surface Disinfection Sequence
Sequence discipline is what separates a compliant clean from one that simply looks clean. Always work from the highest-classification zone outward, and from ceiling to floor — never reverse this order.
ISO Class 5 hoods/workstations first
- Use sterile wipes and sterile water solutions
- Clean all interior surfaces thoroughly
- Allow proper contact time before resuming use
Walls (top to bottom)
- Start at ceiling junction
- Use overlapping horizontal strokes
- Work downward in sections
- Never skip areas or clean in circular motions
Work surfaces, counters, and equipment exteriors
- Clean all horizontal surfaces
- Include equipment housings and control panels
- Pay attention to frequently touched areas
Doors, handles, and pass-throughs
- Disinfect both sides of doors
- Clean handles, frames, and viewing windows
- Include interlock systems and switches
Floors last
- Use clean-to-dirty pattern
- Start from farthest corner from exit
- Work systematically toward the door
- Never walk on freshly cleaned areas
Contact Time and Wiping Technique
Contact time is non-negotiable. Many disinfectants require the surface to remain visibly wet for their full kill time — wiping dry too early renders the disinfectant ineffective, even if the surface looks clean. Unidirectional wiping matters for the same reason: circular patterns simply redistribute particles rather than removing them.
- Maintain visible surface wetness for the full manufacturer-specified contact time (typically 1–10 minutes)
- Use unidirectional wiping motions only — circular patterns redistribute contamination
- Fold wipes to expose a clean surface as you progress
- Discard wipes when fully soiled
- Never re-use wipes or return them to solution
Final Inspection and Documentation
Before exiting, complete a structured close-out check:
- Visually inspect all surfaces for cleanliness and residue
- Verify no cleaning materials remain in the controlled environment
- Confirm waste has been properly removed
- Complete the cleaning log immediately with all required information
- Document any deviations or issues encountered
Documentation completed at the time of cleaning — not reconstructed afterward — is what stands up to regulatory audit.
Post-Cleaning Protocol
Once cleaning is complete, allow the environment to recover before resuming production:
- Allow 15–20 minutes settling time before resuming operations (validate the actual recovery time for your specific facility)
- Store all cleaning equipment outside the cleanroom in designated areas
- Launder or dispose of used mop heads and wipes per protocol
- Never store dirty cleaning materials inside the controlled environment
ISO Class 7 Cleaning Schedule & Frequency
USP <797> establishes minimum cleaning frequencies that apply regardless of activity level. The schedule below breaks requirements into three tiers — daily, weekly, and monthly — with guidance on when to increase frequency based on contamination risk.
Daily Cleaning Requirements
- Disinfect all ISO Class 5 work surfaces (hoods, biosafety cabinets, laminar flow workstations)
- Clean and disinfect sinks and countertops
- Empty waste receptacles and replace liners
- Mop floors using clean-to-dirty pattern starting from farthest corner
- Wipe high-touch surfaces: door handles, light switches, phones, pass-through controls
Weekly Cleaning Requirements
Complete all daily tasks, plus:
- Deep clean walls from top to bottom using approved disinfectant
- Clean windows and viewing panels (both sides)
- Sanitize storage shelving and bins (empty before cleaning)
- Clean exterior surfaces of all equipment and furniture
- Disinfect waste containers thoroughly
Monthly Cleaning Requirements
Complete all daily and weekly tasks, plus:
- Clean ceilings and overhead structures (light fixtures, sprinkler heads, HEPA housings)
- Deep clean and sanitize refrigerators and incubators
- Inspect and clean HEPA filter housings (exterior only—never compromise filter integrity)
- Comprehensive cleaning of all horizontal surfaces including tops of equipment and storage units
- Use sporicidal agents for comprehensive microbial control
Frequency Adjustments
These three tiers set the baseline, but several conditions require you to clean more often. Bump frequency when activity levels rise (more personnel means more particles), when spills occur, or when environmental monitoring exceeds action levels (10 cfu/m³ for ISO 7 air). Seasonal factors — such as increased HVAC load or higher humidity — can also elevate contamination risk, as can process changes that introduce new sources of particulates.

Common Mistakes to Avoid in ISO Class 7 Cleaning
Using Non-Approved Products or Incorrect Dilutions
Household cleaners, unapproved disinfectants, or improperly diluted solutions don't just underperform — they actively compromise the controlled environment. These products can leave chemical residues, introduce particles or volatile organic compounds (VOCs), and fail to kill target organisms at wrong concentrations.
Before using any cleaning product in an ISO Class 7 space:
- Verify the EPA registration number or Health Canada DIN number
- Follow dilution instructions exactly as written on the label
- Record lot numbers and preparation details for every solution batch
- Never substitute products without formal change control approval and validation
Improper Cleaning Direction and Cross-Contamination
Cleaning from dirty to clean areas redistributes contamination across previously cleaned surfaces. Using the same mop for floors and walls — or moving equipment between classification areas without decontamination — can undo an entire cleaning cycle.
Always work clean-to-dirty: start near ISO Class 5 zones and move toward exits. Clean top-to-bottom so gravity carries dislodged particles away from already-cleaned surfaces. Equipment should be dedicated to specific zones — tools don't cross classification boundaries without documented cleaning and revalidation.
Inadequate Contact Time and Documentation Failures
Two separate failures often occur together here. First, wiping surfaces without adequate dwell time renders disinfectants ineffective — the chemistry requires sustained contact with microorganisms to achieve its rated kill claims. Second, incomplete documentation creates compliance gaps that make contamination investigations nearly impossible to resolve.
For dwell time, the standard is straightforward: if a product requires 10 minutes of contact, the surface must remain visibly wet for 10 minutes. Reapply as needed.
For documentation, complete cleaning logs immediately after finishing — never from memory. Each log entry should capture:
- Date, time, and personnel
- Products used with lot numbers
- Preparation details and dilution ratios
- Any deviations from standard procedure
Conclusion
Systematic, documented cleaning procedures are non-negotiable for maintaining ISO Class 7 cleanroom classification and regulatory compliance. The particle count standard of ≤352,000 particles (≥0.5 microns) per cubic meter can only be achieved through strict adherence to validated protocols using approved materials, trained personnel, and proper techniques.
With contamination events costing the pharmaceutical industry billions annually, a well-executed cleaning program is one of the highest-return investments a facility can make. The benefits extend across the operation:
- Protects product quality and prevents costly batch failures
- Reduces regulatory risk and supports audit readiness
- Maintains brand reputation through consistent, compliant output
Treat your cleaning program as a core operational function — not an afterthought — and your ISO Class 7 classification will hold up under the scrutiny it was designed to withstand.
Frequently Asked Questions
What are the requirements for ISO 7 cleaning?
ISO Class 7 cleaning requires approved disinfectants (carrying EPA registration numbers in the US or Health Canada DIN numbers in Canada) with verified contact times, cleanroom-grade low-lint supplies meeting IEST standards, and trained personnel in proper PPE. Procedures must follow clean-to-dirty and top-to-bottom patterns, maintaining particle counts at or below 352,000 particles ≥0.5 microns per cubic meter, with documentation retained for 3+ years.
How often should an ISO Class 7 cleanroom be cleaned?
At minimum: daily disinfection of work surfaces, sinks, and floors; weekly cleaning of walls, windows, and equipment exteriors; and monthly sanitization of ceilings and overhead structures. Increase frequency if environmental monitoring shows trending particle counts or if activity levels rise significantly.
What cleaning agents are approved for ISO Class 7 cleanrooms?
Approved disinfectant chemistries include Accelerated Hydrogen Peroxide, Sodium Hypochlorite (bleach), 70% Isopropyl Alcohol, Quaternary Ammonium Compounds, Phenolics, and Iodophors. All products must carry EPA registration numbers (U.S.) or Health Canada DIN numbers (Canada) and must be used according to manufacturer's labeled contact times to ensure efficacy.
What PPE is required for cleaning ISO Class 7 cleanrooms?
Personnel must wear cleanroom-grade gloves, lint-free shoe covers, bouffant caps, face masks, safety glasses, and cleanroom gowns or frocks (plus beard covers if applicable). Donning order matters: shoe covers go on first, gloves last. All items must be cleanroom-grade to avoid particle shedding.
How do you document ISO Class 7 cleanroom cleaning activities?
Each cleaning event must be logged immediately (never backdated) with date, time, personnel names, products and lot numbers, surfaces cleaned, contact times, deviations, and verification signatures. Retention periods vary by regulation: typically 3 years under USP <797> for sterile compounding, or 1 year post-expiration under Health Canada GMP and FDA cGMP requirements.
What is the difference between cleaning an ISO Class 7 vs Class 8 cleanroom?
ISO Class 7 (352,000 particles/m³) requires more frequent cleaning, stricter material requirements with low-lint supplies, more rigorous PPE protocols, and sterile water for critical ISO Class 5 surfaces within the space. ISO Class 8 tolerates 10x more particles per cubic meter, so procedures are somewhat less stringent—though minimum cleaning frequencies remain comparable under USP <797> guidelines.


