
Introduction
Contamination failures in cleanrooms don't just compromise product quality—they trigger regulatory enforcement actions, costly recalls, and production shutdowns. A review of U.S. FDA Warning Letters from 2018–2025 reveals that approximately 22% cite cleanroom design or construction defects as root causes — from inadequate airflow to pressure cascade failures. Health Canada enforcement trends echo the same pattern.
Facility teams often discover these gaps during qualification, when fixes are expensive and project timelines have already slipped.
ISO 14644-4:2022 provides a systematic framework that integrates contamination control from the earliest conceptual phase through operational handover — closing the gap between regulatory intent and practical design execution.
This guide breaks down the standard's key requirements for facility designers, project managers, and quality professionals. It covers:
- How ISO 14644-4 structures the design and construction process
- Critical requirements for airflow, pressurization, and envelope performance
- Commissioning and start-up documentation expectations
- How the standard aligns with FDA and Health Canada regulatory frameworks
TLDR: Key Takeaways
- ISO 14644-4:2022 establishes design, construction, and start-up requirements for cleanrooms across all controlled environment applications
- The standard introduces performance-based metrics like Air Change Effectiveness (ACE) and Contaminant Removal Effectiveness (CRE) rather than fixed air change rates
- Compliance requires ISO 14644-1 integration plus key documentation: URS, Design Qualification, and risk assessments
- Modular construction reduces on-site contamination by 30-40% compared to traditional stick-built methods
- The standard is effectively mandatory for pharmaceutical, biotech, and medical device manufacturers — FDA recognition and EU GMP Annex 1 both reference it
Understanding ISO 14644-4: Scope and Purpose
What the Standard Covers
ISO 14644-4:2022 specifies the complete process for creating cleanrooms and clean zones from initial requirements through design, construction, and start-up. Unlike ISO 14644-1, which defines cleanliness classifications based on particle counts, Part 4 focuses on how to build facilities that achieve and maintain those classifications. The standard applies to new construction, refurbishments, and modifications of controlled environments.
The standard's primary focus is airborne particle concentration control, though it acknowledges other contamination attributes like chemical vapors or surface cleanliness. It explicitly excludes operational activities (covered in ISO 14644-5), fire and safety regulations, and ongoing maintenance protocols.
Objective and Contamination Control Strategy
ISO 14644-4's core objective is establishing a contamination control strategy from the earliest design phase through construction completion. This proactive approach prevents the design deficiencies that commonly appear in FDA Warning Letters: cleanrooms that lack easily cleanable surfaces, cannot maintain pressure differentials, or contain structural gaps that create direct contamination pathways.
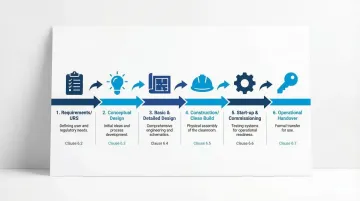
Standard Structure and Lifecycle Approach
The standard follows the project lifecycle through six key sections:
- Requirements (Clause 6): Defining User Requirements Specification and contamination control strategy
- Design (Clause 7): Conceptual, basic, and detailed design phases
- Construction (Clause 8): Clean build protocols and assembly procedures
- Start-up (Clause 9): Commissioning and operational handover
- Annexes A-D: Informative guidance on requirements, design, construction, and verification
Relationship to ISO 14644-1
ISO 14644-4 relies on ISO 14644-1 for cleanliness class definitions (ISO Class 1–9). Each design decision flows from the target classification the User Requirements Specification defines:
- Material selection: Surfaces must support the cleanability and outgassing requirements of the target ISO class
- HVAC configuration: Air change rates, filtration, and pressure cascades are sized to the classification
- Layout: Room adjacencies and traffic flows must prevent cross-contamination between zones
Start-up verification confirms the facility meets the ISO 14644-1 class through particle counting and other test methods defined in ISO 14644-3.
Industry Applicability
While technically voluntary, ISO 14644-4 is effectively mandatory for regulated industries:
Pharmaceutical and Biotech: EU GMP Annex 1 (2022) explicitly references ISO 14644-4 for cleanroom design, pressure-differential validation, and qualification, making compliance required for sterile manufacturers exporting to the EU.
Medical Devices: The FDA recognizes ISO 14644-4 (Recognition #14-589), meaning conformity can support premarket submissions.
Semiconductors and Microelectronics: The standard is the effectively the required standard for particulate control in wafer fabrication.
Aerospace, Food Processing, and Healthcare: Industries requiring contamination-sensitive assembly and processing increasingly adopt ISO 14644-4 principles.
Design Requirements and Considerations
Facility Layout and Flow Patterns
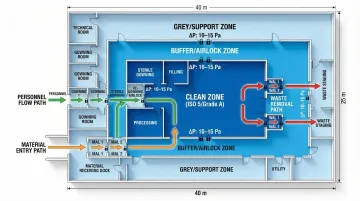
Effective contamination control begins with logical flow patterns that minimize cross-contamination risks. ISO 14644-4 requires designs that account for personnel movement, material handling, and waste removal—each of which creates potential transfer pathways between clean and dirty areas.
Key layout considerations include:
- Personnel, material, and waste flows: Separate pathways prevent contamination transfer between operations. FDA guidance specifies that rooms of higher air cleanliness must maintain 10–15 Pascals (Pa) positive pressure relative to adjacent lower-quality areas.
- Zoning strategies: Clean zones must be segregated from grey zones and support areas through physical barriers or aerodynamic separation. EU Annex 1 requires airlocks to be flushed with filtered air to minimize contamination entry during zone transitions.
- Airlock and buffer zone design: These transition spaces maintain pressure cascades and provide gowning areas. Proper design includes interlocked doors, adequate sizing for personnel or material carts, and appropriate air change rates to flush contaminants between door openings.
Materials Selection and Finishes
ISO 14644-4 mandates that internal surfaces be smooth, impervious, and free of gaps or pathways to uncontrolled areas. Material requirements include:
Surface Characteristics:
- Non-porous and non-shedding to prevent particle generation
- Cleanable and resistant to disinfectants and cleaning agents
- Smooth without recesses that accumulate contamination
- Seamless junctions between walls, floors, and ceilings
Wall Panel Systems: Options include High Pressure Laminate (HPL), powder-coated galvanized steel, and u-PVC panels. All must provide continuous, sealed surfaces without ledges or projecting features that collect particles.
Flooring Systems: Seamless epoxy, sheet vinyl, or other monolithic systems that eliminate grout lines and joints where contamination accumulates.
Ceiling Systems: Walkable ceilings allow maintenance access without contaminating the controlled space below, while providing continuous filtration through HEPA or ULPA filters.
Door Assemblies: Flush-mounted cleanroom doors with continuous seals maintain pressure differentials and cleanliness levels while accommodating personnel and material flow.
ACH Engineering addresses these requirements through prefabricated wall panels, seamless coving that eliminates sharp corners in line with cGMP requirements, and flush-mounted doors designed to hold internal pressure and cleanliness class.
HVAC and Environmental Control Systems
Once the cleanroom envelope is established, HVAC design determines whether it actually performs to spec. ISO 14644-4:2022 shifts to performance-based ventilation metrics rather than prescriptive air change rates—giving designers more flexibility but demanding more rigorous justification.
Filtration Requirements: Final filters (HEPA or ULPA) remove airborne particles. HEPA filters provide ≥99.97% efficiency, while ULPA filters achieve ≥99.9995% efficiency.
Airflow Patterns:
- Unidirectional (UDAF): Required for critical zones (Grade A/ISO 5) to sweep particles away from products with laminar flow
- Non-Unidirectional: Acceptable for supporting zones (ISO 7/8) where dilution is the primary control mechanism
Pressure Differentials: Both FDA and EU Annex 1 require 10–15 Pa positive pressure between adjacent rooms of differing classifications. Systems must include continuous monitoring with alarms for excursions.
Air Change Effectiveness (ACE) and Contaminant Removal Effectiveness (CRE): ISO 14644-4:2022 introduces these indices to optimize ventilation efficiency based on actual performance rather than fixed tables. This allows designers to demonstrate compliance through computational fluid dynamics (CFD) modeling or physical testing.
Temperature and Humidity Control: Setpoints are determined by product and process requirements, as well as operator comfort. FDA Warning Letters cite failures to maintain established limits—such as humidity excursions to 94%—as evidence of inadequate HVAC design. ACH Engineering's systems maintain temperatures between 18°C and 24°C and relative humidity between 30–60%, with tighter tolerances available for pharmaceutical and laboratory applications.
Utilities and Services Integration
All utility penetrations—pipes, ducts, electrical conduits—must be sealed to maintain cleanroom envelope integrity. Design considerations include:
- Smooth, cleanable penetrations without crevices
- Minimal projections into the controlled space
- Accessible service chases outside the cleanroom for maintenance
- Coordination between architectural, mechanical, and electrical systems
ACH Engineering integrates these elements from the conceptual design phase—coordinating material sourcing, vendor management, and construction oversight so utility systems reinforce contamination control rather than undermine it.
Documentation and Design Qualification
ISO 14644-4 Clause 6.3 mandates formal documentation that serves as the primary reference for regulatory inspections. Three documents form the core of this framework:
User Requirements Specification (URS): Defines intended use, regulatory requirements, target cleanliness class, environmental parameters, and critical process requirements.
Design Qualification (DQ): Verification that the proposed design meets the URS through systematic review of drawings, specifications, and calculations.
Risk Assessment: Contamination risk assessment identifies critical control points, informs design decisions, and establishes the basis for qualification protocols.
Construction Phase Requirements
Construction Sequencing and Clean-Build Protocol
ISO 14644-4 requires a "clean build protocol" that increases in stringency as construction progresses. This systematic approach minimizes contamination during construction.
The sequencing follows a strict progression, with cleanliness requirements tightening at each phase transition:
- Complete dirty work (framing, rough utilities, drilling) before installing finish materials
- Seal the cleanroom envelope before installing final filters
- Protect completed surfaces from ongoing construction activities
- Install and activate HVAC systems progressively to maintain positive pressure

Specific cleaning protocols apply before moving between phases — not after.
Material Handling and Storage
Components entering the construction zone must be:
- Cleaned before delivery to remove manufacturing residues
- Protected during transport and storage to prevent damage or contamination
- Staged in controlled storage areas separate from active construction
Inspection and Testing at Key Milestones
Critical verification activities during construction include:
- Verify the cleanroom envelope holds pressure differentials before installing finish materials
- Confirm installed HEPA/ULPA filters are leak-free to protect cleanliness classification
- Measure pressure cascades between zones to confirm design intent is met
Modular Construction Advantages
Modular cleanroom approaches reduce on-site particulate generation and accelerate commissioning by 30–40% compared to stick-built methods. That advantage comes from four concrete factors:
- Factory fabrication eliminates on-site cutting, sanding, and drywall dust that compromise cleanliness during traditional construction
- Controlled manufacturing environments produce components with consistent dimensional accuracy
- Simultaneous site preparation and component fabrication compress overall project timelines
- Prefabricated systems support easier modification as regulatory requirements or processes evolve
ACH Engineering applies this approach directly — factory-fabricating cleanroom components including wall panels, ceilings, and integrated systems to reduce on-site contamination risks while building in flexibility for future process changes.
Key Differences: ISO 14644-4 vs. Other Standards
ISO 14644-4 vs. EU GMP Annex 1
| Feature | ISO 14644-4:2022 | EU GMP Annex 1 (2022) |
|---|---|---|
| Scope | General cleanroom design & construction | Sterile medicinal product manufacturing |
| Pressure Differentials | Recommends pressure cascades | Mandates 10–15 Pa differential |
| Surface Requirements | "Smooth, impervious, cleanable" | "Smooth, impervious, unbroken" with no recesses |
| Qualification | Defines start-up & verification | Requires qualification per Annex 15 & ISO 14644 |
| Regulatory Status | Voluntary consensus standard | Legally binding for EU manufacturers |
EU Annex 1 references ISO 14644-4 as the technical basis for cleanroom design. In practice, this makes both frameworks effectively mandatory for pharmaceutical manufacturers exporting to Europe — ISO 14644-4 sets the engineering foundation, and Annex 1 defines the regulatory threshold.
ISO 14644-4 vs. FDA Guidance
FDA guidance aligns with ISO 14644-4 on airflow patterns (unidirectional for critical areas) and pressure differentials. For medical device manufacturing, the FDA recognizes ISO 14644-4 conformity as valid support for regulatory submissions.
Enforcement actions tell the same story. FDA 483 observations and warning letters frequently cite ISO 14644-4 deviations as cGMP violations, including:
- Inadequate pressure differential control between adjacent zones
- Poor facility design that creates contamination pathways
- Structural deficiencies in walls, floors, or ceilings that compromise cleanroom integrity
Relationship to Other ISO 14644 Parts
ISO 14644-4 doesn't operate in isolation — it works alongside other parts of the standard series, each covering a distinct phase of the cleanroom lifecycle:
- ISO 14644-1 sets the cleanliness classification system (particle count thresholds by class). Part 4 defines how to design and build a facility capable of achieving those classes.
- ISO 14644-2 covers ongoing operational monitoring to demonstrate sustained compliance. Part 4 addresses facility creation and initial start-up verification.
- ISO 14644-3 provides the specific test methods — particle counting, filter integrity testing, airflow uniformity — that Part 4 references during commissioning.
Start-up and Qualification
Start-up Phase Definition
ISO 14644-4 defines start-up as setting the installation to work and verifying its performance. This includes commissioning activities (balancing airflows, calibrating controls) and functional verification that the facility meets URS requirements.
Initial Qualification Testing
Required start-up verification activities include:
- Smoke studies verify airflow direction, identify stagnant zones, and confirm unidirectional patterns in critical areas
- Classification testing per ISO 14644-1 confirms the target cleanliness class under "at-rest" conditions (HVAC operating, no personnel or process equipment present)
- Pressure cascade measurements across all adjacent zones confirm the design intent is met
- Recovery testing measures how quickly the facility returns to specification after a contamination challenge
- Containment leak testing verifies envelope integrity and pressure differential stability during operations
- In-place HEPA/ULPA filter leak testing using aerosol challenge methods confirms zero bypass leakage

Documentation Requirements
These tests generate the documentation that regulators and auditors scrutinize most closely. Two formal qualification packages are required:
Installation Qualification (IQ) confirms equipment and systems are installed per design specifications — correct filter grades, sealed penetrations, and proper equipment placement.
Operational Qualification (OQ) confirms systems perform within established limits under "at-rest" conditions, including pressure differentials, temperature/humidity control, and particle counts.
Together, IQ and OQ form the regulatory foundation for Health Canada, FDA, and EMA inspections, providing auditable evidence that the facility was built and verified to its original design intent.
Frequently Asked Questions
What is ISO 14644-4 and what does it cover?
ISO 14644-4:2022 is the international standard specifying design, construction, and start-up requirements for cleanrooms and clean zones, covering everything from facility layout and material selection to HVAC design and initial qualification testing.
How does ISO 14644-4 differ from ISO 14644-1?
ISO 14644-1 defines cleanroom classification based on particle counts (ISO Class 1–9), while ISO 14644-4 specifies how to design, build, and start up facilities to achieve and maintain those classifications.
What are the key design requirements in ISO 14644-4?
Key requirements span materials, systems, and spatial planning:
- Select non-shedding, cleanable surfaces and finishes
- Design HVAC systems with appropriate filtration and pressure cascades
- Establish logical personnel and material flow patterns
- Implement zoning with airlocks to contain contamination
- Integrate contamination control strategies from the earliest design phase
Is ISO 14644-4 mandatory or voluntary?
ISO 14644-4 is a voluntary consensus standard, but regulatory bodies like FDA and EMA reference it extensively. EU GMP Annex 1 explicitly requires ISO 14644 compliance, making it effectively mandatory for pharmaceutical and biotech manufacturers in regulated markets.
What industries need to comply with ISO 14644-4?
Any industry operating in contamination-sensitive environments falls under its scope. The most common include:
- Pharmaceutical manufacturing and biotechnology
- Medical devices and compounding pharmacies
- Semiconductor fabrication and electronics
- Aerospace and defense
- Hospitals, healthcare facilities, and R&D laboratories
How does ISO 14644-4 relate to GMP requirements?
GMP guidelines like EU Annex 1 set the regulatory requirements, while ISO 14644-4 provides the technical framework for meeting them. In practice, facilities must satisfy both — the standard tells you how to build and qualify the cleanroom; the regulation tells you why it's required.


