
Introduction
The pharmaceutical, biotechnology, and medical device manufacturing sectors face mounting pressure to maintain contamination-free environments while adapting to rapidly changing production demands. The global cleanroom technology market is projected to reach $10.82 billion USD by 2030, driven by increased biopharmaceutical production and tightening regulatory requirements.
Yet traditional cleanroom construction presents significant obstacles: projects typically require 6-18 months to complete, cost between $400-$700 USD per square foot, and lack the flexibility to accommodate evolving business needs. When production timelines are measured in weeks and regulatory compliance is non-negotiable, these constraints can severely impact competitiveness.
Modular cleanrooms change that equation. These pre-designed, factory-built controlled environments deploy 40-60% faster and cost 30-50% less than stick-built construction — without compromising compliance with USP, cGMP, FDA, and ISO standards. What follows covers what modular cleanrooms are, how they work, and what industries stand to gain most from making the switch.
TLDR: Key Takeaways
- Pre-designed systems install in 4–12 weeks versus 6–18 months for traditional construction
- ISO Class 3–8 compliance at $200–$400 CAD per square foot — a fraction of conventional build costs
- Available in hardwall, softwall, and rigidwall configurations for different permanence needs
- Reconfigure, expand, or relocate the cleanroom as your operations grow or processes evolve
- Full regulatory compliance with USP 797/800, cGMP, FDA 21 CFR Part 11, and ISO 14644-1
What Are Modular Cleanrooms?
Modular cleanrooms are pre-designed, factory-built controlled environment systems designed to maintain specific cleanliness levels according to ISO 14644-1 classification standards. Unlike traditional stick-built construction, these systems use prefabricated components manufactured in controlled factory conditions and assembled rapidly on-site.
Modular Cleanroom Construction and Components
The prefabrication process begins in controlled manufacturing facilities where panels, frames, and systems are built to precise specifications. This approach eliminates the variability and contamination risks common with on-site construction.
Key components include:
- Wall panels: Non-shedding, non-reactive materials such as powder-coated steel, stainless steel, or vinyl-clad panels
- Ceiling grids: Walkable systems that allow maintenance access without compromising the controlled environment
- HEPA/ULPA filtration: High-efficiency filters capturing 99.97% of particles at 0.3 µm
- HVAC units: Integrated systems providing precise temperature (18-24°C) and humidity control (30-60% RH)
- Lighting systems: Flush-mounted fixtures that maintain surface integrity
- Flooring: Seamless epoxy or vinyl systems with coved transitions to eliminate particle accumulation points

Types of Modular Cleanroom Systems
| Type | Construction | Best For | Limitations |
|---|---|---|---|
| Hardwall | Solid aluminum or steel-framed panels | Pharmaceutical manufacturing, semiconductor production, medical device assembly — anywhere strict ISO classification is required | Higher upfront cost; less flexible to relocate |
| Softwall | Flexible vinyl curtain walls with lightweight aluminum frames | Temporary applications, budget-sensitive projects, areas where visual access is a priority | Not ideal for strict temperature and humidity control |
| Rigidwall | Clear acrylic or composite polymer panels | Applications needing strong visual monitoring combined with solid environmental control | Mid-range environmental performance; sits between hardwall and softwall |
ISO Classification Capabilities
The type of modular system you choose directly affects which ISO class you can achieve. The ISO 14644-1 standard defines classifications based on maximum allowable particle concentrations per cubic meter of air. Modular systems can achieve ISO Class 3 through Class 8, with Class 5-7 being most common for pharmaceutical applications.
| ISO Class | Particles ≥0.5 µm/m³ | Air Changes/Hour | Typical Applications |
|---|---|---|---|
| ISO 3 | 35 | 540-600+ | Semiconductor manufacturing |
| ISO 4 | 352 | 400-540 | Optics, biotech research |
| ISO 5 | 3,520 | 240-600 | Aseptic processing, sterile filling |
| ISO 6 | 35,200 | 70-160 | Medical device assembly |
| ISO 7 | 352,000 | 30-60 | Pharmacy buffer areas |
| ISO 8 | 3,520,000 | 10-25 | Gowning rooms, packaging |
Key Benefits of Modular Cleanrooms
Rapid Installation and Deployment
Modular systems demonstrate 40-60% faster deployment compared to traditional construction. Typical installation timelines range from 4-12 weeks versus 6-18 months for stick-built projects.
A lean manufacturing case study documented a 41% reduction in field hours for a modular facility, with over 85% of project hours moved off-site. This dramatic shift reduces on-site disruption and accelerates time-to-operation.
Pre-certification and factory testing further accelerate validation. Components arrive with documentation packages that simplify IQ/OQ/PQ protocols, reducing the validation timeline by weeks.
Cost-Effectiveness and Budget Predictability
Modular cleanrooms typically cost $200-$400 CAD per square foot compared to $400-$700 CAD per square foot for traditional construction—a potential savings of 30-50%.
Cost advantages stem from:
- Reduced labor through factory assembly
- Shorter project timelines minimizing carrying costs
- Minimal site disruption to ongoing operations
- Lower material waste through controlled manufacturing
- Predictable fixed pricing eliminating cost overruns

These savings extend beyond construction costs. Modular units are often classified as equipment rather than building improvements, which can unlock more favorable depreciation treatment and meaningfully improve ROI and cash flow—consult your tax advisor for jurisdiction-specific guidance.
Flexibility and Scalability
Modular panels can be easily reconfigured, expanded, or relocated as business needs evolve. A 500-square-foot cleanroom can expand to 5,000+ square feet without complete reconstruction.
This adaptability proves particularly valuable for:
- Growing companies anticipating capacity increases
- Organizations with changing product portfolios
- Facilities requiring seasonal production adjustments
- Companies consolidating or relocating operations
The modular approach allows businesses to preserve their infrastructure investment rather than starting from scratch when requirements change.
Regulatory Compliance and Validation
Modular cleanrooms meet USP 797/800, cGMP, FDA 21 CFR Part 11, and ISO 14644 standards through factory-controlled manufacturing and standardized designs.
Pre-certification processes simplify validation documentation. Manufacturers provide comprehensive packages including:
- Design Qualification (DQ) documentation
- Installation Qualification (IQ) protocols
- Operational Qualification (OQ) test procedures
- Performance Qualification (PQ) acceptance criteria
This pre-packaged documentation significantly reduces the regulatory submission burden. Inspectors and quality teams receive audit-ready records from day one, rather than assembling evidence retrospectively after construction.
Minimal Operational Disruption
Modular installation doesn't require facility shutdowns or extensive construction activities. Since components are prefabricated off-site, on-site work primarily involves assembly rather than construction from raw materials.
This approach dramatically reduces:
- Construction noise and dust
- Number of workers required on-site
- Contamination risks to existing operations
- Scheduling conflicts with ongoing production
Turnkey providers manage design, engineering, installation, and validation as a single coordinated process—a critical advantage for facilities that must maintain sterile or controlled conditions throughout the build.
Quality Control and Consistency
Factory-controlled manufacturing ensures superior quality compared to on-site construction variables. Controlled environments eliminate weather exposure, dust contamination, and inconsistent workmanship.
Testing procedures conducted before delivery include:
- Pressure differential verification
- HEPA filter integrity testing per EN 1822 and IEST-RP-CC001 standards
- Particle count measurements
- Airflow pattern validation
- Surface cleanliness assessment
Industries and Applications
Pharmaceutical and Biotechnology Applications
Pharmaceutical and biotech sectors account for over 65% of cleanroom demand. Applications include:
- Sterile compounding: USP 797-compliant facilities requiring ISO Class 7 buffer rooms with ISO Class 8 anterooms
- Cell and gene therapy manufacturing: ISO Class 5 biosafety cabinets within ISO Class 7 buffer rooms
- Vaccine production: Aseptic processing areas with ISO Class 5 critical zones
- Aseptic processing: Sterile drug production requiring ISO Class 5 or better air quality for critical areas
Most pharmaceutical applications require ISO Class 5-7 environments with continuous monitoring of temperature, humidity, and pressure differentials per FDA cGMP and Health Canada GMP requirements.
Medical Devices and Healthcare
Medical device manufacturing requires compliance with ISO 13485 and FDA 21 CFR 820. Applications include:
- Medical device assembly (typically ISO Class 5-7)
- Surgical instrument manufacturing
- Hospital compounding pharmacies
- Tissue banks and cell culture facilities
Assembly areas range from ISO Class 5 to ISO Class 7 depending on device risk profile, with surfaces required to be smooth, impervious, and non-shedding to facilitate cleaning and disinfection.
Other High-Tech Industries
Beyond pharma and healthcare, cleanroom standards apply across a wide range of industries with equally demanding contamination control needs:
- Semiconductors: ISO Class 1-4 environments with tightly controlled humidity and temperature to prevent microelectronics failure
- Aerospace: Assembly and inspection areas where even trace contamination can cause component failure in critical systems
- Battery Dry Rooms: Low-humidity environments (typically <1% RH) for lithium-ion battery electrode manufacturing
- Food Processing: Hygienic processing and packaging zones that meet food safety standards without sacrificing throughput
- Cannabis Cultivation: Controlled grow rooms, extraction facilities, and packaging areas for agricultural and medical cannabis operations
Design and Technical Considerations
HVAC and Air Filtration Systems
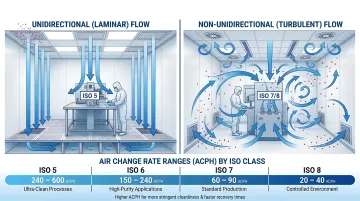
HEPA filtration is the baseline for cleanroom air quality control. Standard HEPA filters capture 99.97% of particles at 0.3 µm, while ULPA filters used for ISO 3–5 environments deliver even higher efficiency for ultra-critical applications.
Air change rates vary by ISO classification:
- ISO 5: 240-600+ ACH
- ISO 6: 70-160 ACH
- ISO 7: 30-60 ACH
- ISO 8: 10-25 ACH
Select airflow patterns based on your ISO zone requirements:
- Unidirectional (laminar) flow: Required for ISO 5 critical zones in aseptic filling to sweep particles away from products
- Non-unidirectional (turbulent) flow: Acceptable for ISO 7/8 areas where air mixing dilutes particulate concentration
HVAC systems typically account for 25-40% of total project cost, making proper design essential for both performance and budget management.
Pressure Differentials and Cascading
Pressure control prevents contamination migration between adjacent spaces. The table below summarizes key regulatory thresholds:
| Standard | Application | Pressure Requirement |
|---|---|---|
| USP <800> | Hazardous drug buffer rooms | Negative: 0.01–0.03 in. w.c. relative to adjacent areas |
| USP <797> | Non-hazardous sterile compounding buffer rooms | Positive: ≥0.02 in. w.c. vs. anterooms and unclassified areas |
Pressure cascade principles ensure airflow from cleanest to less clean areas, protecting critical zones. Continuous monitoring devices must track and record pressures daily to maintain compliance.
Material Selection and Surfaces
Cleanroom materials must be non-porous, non-shedding, chemical-resistant, and easy to clean per ISO 14644-9 surface cleanliness standards.
Wall panel materials:
- Powder-coated steel: Durable, chemical-resistant option for standard cleanrooms
- Stainless steel: Ideal for ISO 5 and aseptic environments requiring maximum chemical resistance
- Vinyl-clad panels: Cost-effective solution with static-dissipative properties
- FRP (Fiberglass Reinforced Plastic): Chemical-resistant and durable for medical device and pharma applications
Flooring options:
- Epoxy coatings: Seamless, chemical-resistant, available in ESD formulations
- Heat-welded vinyl: Seamless installation with coved transitions to walls
- Seamless PVC: Chemical-resistant with excellent cleanability
All flooring must be seamless and coved to walls to prevent particle accumulation and allow effective cleaning protocols.
Monitoring and Control Systems
Environmental monitoring requirements per ISO 14644-2 include:
- Particle counters: Routine monitoring to prove continued compliance
- Temperature/humidity sensors: Continuous tracking of critical parameters
- Pressure differential monitors: Real-time verification of cascading pressure
- Viable monitoring: Microorganism detection on surfaces and in air
Integration with Building Management Systems (BMS) enables real-time monitoring, automated alarming, and electronic records compliant with FDA 21 CFR Part 11 for data integrity and audit trails.
When specifying a BMS, confirm that your monitoring vendor can provide validated software and 21 CFR Part 11-compliant audit trail documentation before installation begins.
Personnel and Material Flow
Proper gowning room design and personnel flow minimize contamination. IEST-RP-CC003 provides guidance on garment systems for cleanrooms.
Best practices include:
- Airlocks: Personnel enter through designated anterooms with cascading pressure
- Pass-throughs: Material transfer chambers minimize personnel traffic between zones
- Unidirectional flow: In aseptic areas, personnel must avoid interrupting airflow over open products
- Strict access control: Only properly trained personnel permitted in controlled areas
Getting the layout right from the start — with clear separation between gowning areas, buffer rooms, and production zones — is far less costly than retrofitting flow paths after construction.
Modular vs. Traditional Cleanrooms: Making the Right Choice
Choosing between modular and traditional cleanroom construction comes down to your timeline, budget, and how your facility needs might evolve. Here's a practical breakdown of where each approach fits best.
When Modular Cleanrooms Are the Best Choice
Modular systems are well-suited for:
- Completing a project in under 6 months, where construction speed is a priority
- Reducing upfront capital costs by 30–50% compared to traditional builds
- Scaling capacity or reconfiguring layouts as your production needs shift
- Short-term or phased operations such as pilot production, clinical trial manufacturing, or seasonal runs
- Installing within an occupied building where minimizing structural disruption matters
- Facilities under 10,000 square feet, where modular construction offers the strongest cost-to-value ratio

That said, modular isn't the right fit for every scenario. Traditional stick-built construction holds advantages in specific situations.
When Traditional Construction May Be Preferred
Stick-built construction is worth considering for:
- Large-scale facilities exceeding 20,000 square feet, where traditional economies of scale become more competitive
- Projects with complex architectural integration requirements tied to existing building infrastructure
- Permanent installations where reconfiguration or relocation is not anticipated
- Sites in jurisdictions with building codes that mandate traditional construction methods
If you're unsure which approach fits your project, ACH Engineering's team can assess your requirements and recommend the right path — from initial scoping through full turnkey delivery.
Frequently Asked Questions
How much does a modular cleanroom cost?
Modular cleanroom costs typically range from $200–$400 per square foot (USD) depending on ISO class and specifications. Higher classifications like ISO 5 can reach $1,000+ per square foot due to greater HVAC capacity and HEPA coverage requirements. Key cost drivers include size, classification, HVAC complexity, and wall panel materials. Modular systems typically run 30–50% less than traditional construction.
What industries use modular cleanrooms?
Primary industries include pharmaceutical/biotech (sterile manufacturing, compounding), medical devices (assembly, packaging), compounding pharmacies (USP 797/800 compliance), semiconductors (microelectronics fabrication), aerospace (component assembly), food/beverage (hygienic processing), cannabis cultivation (extraction and packaging), and research laboratories (controlled experimentation).
How long does it take to install a modular cleanroom?
Installation timelines vary by project complexity: 4–8 weeks for small systems under 1,000 square feet, 8–12 weeks for medium installations (1,000–5,000 square feet). This is 40–60% faster than traditional construction.
What ISO classifications are available for modular cleanrooms?
Modular systems can achieve ISO Class 3 through Class 8, with Class 5–7 being most common for pharmaceutical applications. Classification depends on filtration efficiency, air change rates, and room design. Higher classifications require more sophisticated HVAC systems and tighter environmental controls.
Can modular cleanrooms be relocated or expanded?
Yes, modular panels can be disassembled, relocated, and reassembled at new locations. Rooms can be expanded by adding panels and adjusting HVAC capacity — preserving your infrastructure investment as requirements evolve.
What is the difference between hardwall, softwall, and rigidwall cleanrooms?
Hardwall uses rigid metal-framed panels for permanent installations requiring maximum environmental control. Softwall uses flexible vinyl curtains for temporary or adaptable setups — good visibility, but lower contamination resistance. Rigidwall uses durable polymer panels that balance permanence with adaptability, offering tighter control than softwall with reasonable transparency.


