
Introduction
Integrating modular cleanrooms into existing facilities presents moderate complexity, requiring careful coordination across structural, mechanical, and operational systems—yet it's far less disruptive than traditional construction. Research shows modular integration reduces project timelines by 40–60% compared to stick-built methods, with teams typically completing on-site assembly in weeks rather than months.
Successfully managing this requires facility managers paired with a design-build team that understands both modular construction principles and existing building constraints. ACH Engineering has delivered integrated cleanroom projects across pharmaceutical, biotech, medical device, semiconductor, and healthcare sectors throughout North America.
When integration is done incorrectly, the consequences compound quickly. Operational downtime extends beyond planned windows, compliance failures delay certification, and total costs can exceed new construction alternatives. The most common failure modes are:
- Structural incompatibilities — insufficient floor load capacity or ceiling clearance
- Pressure cascade failures — HVAC systems unable to maintain required differentials
- Contamination ingress — inadequate containment protocols during active construction
Understanding these risks is exactly why a structured integration approach matters. This guide covers the complete process—from pre-installation assessment through commissioning—following ISO 14644-1, FDA cGMP, and USP requirements.
TLDR
- Modular cleanroom integration requires comprehensive site assessment covering structural capacity, utility loads, and environmental baselines before design begins
- Integration follows a phased approach — assessment → design → preparation → installation → validation — completing 40–60% faster than traditional construction
- Success depends on coordination between cleanroom specialists, facility engineering teams, and regulatory consultants to minimize operational disruption
- Contamination control during construction requires active containment strategies to protect adjacent processes and personnel
- Post-integration validation through ISO 14644-1 testing and industry-specific certification confirms compliance before operations resume
Understanding Modular Cleanroom Integration
What Makes Modular Different
Modular cleanroom integration differs from traditional construction through prefabricated components built in controlled factory settings and assembled on-site. Unlike stick-built methods where raw materials (studs, drywall) are processed on-site with cutting, sanding, and finishing, modular systems arrive as precision-designed panels ready for assembly.
Key advantages include:
- On-site assembly takes days to weeks — not months — minimizing disruption to active operations
- Off-site fabrication eliminates construction dust, debris, and traffic from operational areas
- Panels can be relocated, removed, or reconfigured without complete reconstruction
- Factory manufacturing delivers consistent tolerances and sealing across every panel
Defining Integration Scope
Integration means connecting modular cleanroom systems—walls, ceilings, HVAC, controls—to existing building infrastructure (power, utilities, structural supports) while maintaining facility operations. In practice, this means mapping cleanroom specifications against what your building already provides: available electrical capacity, HVAC tie-in points, structural load ratings, and active utility routing.
When Modular Integration Is Optimal
Choose modular integration over traditional construction when:
- Retrofitting an existing space where shutting down operations isn't an option
- Phased expansions are planned or likely within 3-5 years
- The facility needs flexibility for future process changes or potential relocation
- Project timelines are fixed — clinical trials, regulatory submissions, or contract deadlines won't move
- The space is leased and cleanrooms must qualify as movable equipment rather than permanent improvements
Timeline Expectations
Modular integration typically runs 8-16 weeks from assessment to validation for standard installations, compared to 20-30 weeks for traditional construction. The gap is larger than it appears: modular components are fabricated off-site while site preparation runs in parallel, compressing the critical path (the overall project schedule).
Key Stakeholders
Successful integration pulls together several disciplines. A typical project involves cleanroom design-build specialists (such as ACH Engineering), facility engineering teams, operations managers, quality and compliance officers, regulatory consultants, and contractors handling utility upgrades. Aligning these stakeholders early — before design is finalized — is what separates on-schedule integrations from costly rework.
Pre-Integration Assessment and Planning
Prerequisites and Site Evaluation
Before design begins, verify these minimum structural and utility requirements. Gaps identified at this stage are far cheaper to address than mid-installation surprises.
Structural Prerequisites:
- Ceiling height: 10-12 feet minimum for ISO Class 5-8 installations, allowing at least one foot above the cleanroom ceiling for fixtures and ducts, plus two feet of vertical clearance above Fan Filter Units (FFUs) for maintenance access
- Floor load capacity: Standard cleanroom floors average 200 lbs/ft², but isolated areas for heavy process equipment may require up to 350 lbs/ft²
- Structural obstructions: Identify beams, ductwork, and conduit that may interfere with the cleanroom envelope
Utility Capacity Requirements:
Confirm available capacity before finalizing layout. Undersized electrical or HVAC infrastructure is a common cause of project delays.
- Electrical loads: FFUs typically draw 1.5A at 115VAC or 0.65A at 277VAC per unit; lighting adds similar amperage; a typical USP 797 room may require a dedicated 40A circuit
- HVAC capacity: Must support required air changes per hour (ACH) based on ISO classification:
- ISO 5: 240-360 ACH (unidirectional flow)
- ISO 7: 30-60 ACH
- ISO 8: 10-25 ACH
- USP <797> requires minimum 30 ACH for ISO 7 buffer rooms
- Process utilities: Availability of compressed air, specialty gases, DI water, and drainage

Environmental Baseline Assessment:
Document existing conditions before any work begins:
- Current airborne particle counts to establish improvement targets
- Temperature and humidity control capabilities
- Vibration and noise levels that may affect sensitive processes
- Contamination sources in adjacent areas
Access and Logistics:
- Equipment delivery paths with adequate clearance for large panels
- Installation access points that won't disrupt active operations
- Staging areas for component inspection and preparation
With site conditions documented, the next step is translating your process requirements into a defined cleanroom specification.
What ISO Classification Does Your Process Require?
Select the appropriate ISO class based on process requirements and applicable regulatory standards:
- ISO 5: Critical aseptic processing (filling zones, sterile compounding primary engineering controls)
- ISO 7: Buffer areas for sterile compounding, medical device assembly, pharmaceutical manufacturing
- ISO 8: Background zones, anterooms, less critical manufacturing steps
Industry-specific regulations overlay ISO classifications:
- Pharmaceutical: FDA cGMP (21 CFR Part 211) and Health Canada Good Manufacturing Practices (GUI-0029)
- Medical device: FDA Quality System Regulation (21 CFR Part 820)
- Compounding pharmacy: USP <797> for sterile preparations, USP <800> for hazardous drugs
- Semiconductor: SEMI standards for microelectronics manufacturing
Performance Criteria to Define:
- Particle counts at 0.5 µm and 5.0 µm (per ISO 14644-1)
- Temperature range (typically 20-22°C ±2°C)
- Relative humidity range (typically 30-50% ±5%)
- Air pressure differentials between zones (minimum 0.02 inches water column per USP <797>)
- Air change rates specific to classification
Regulatory Compliance Planning:
Identify all applicable standards early. Compliance requirements shape documentation, validation timelines, and ongoing operational costs.
- Documentation requirements: Installation Qualification, Operational Qualification, and Performance Qualification (IQ/OQ/PQ)
- Validation protocols specific to your industry
- Certification timelines and third-party testing needs
- Ongoing monitoring and recertification schedules (USP <797> requires certification every six months)
Managing Operational Continuity During Integration
A well-planned integration minimizes disruption to active production. The following strategies apply across pharmaceutical, biotech, and manufacturing environments.
Scheduling and Physical Controls:
- Schedule installation in phases timed around production cycles
- Use negative pressure zones and temporary barriers to contain construction dust and particulates
- Identify optimal shutdown windows through direct coordination with production teams
- Establish clear communication protocols for timeline updates and access restrictions
Stakeholder Expectations:
Set these expectations before work begins:
- Full project timeline with critical milestones
- Defined access restrictions to construction zones
- Scheduled windows for high-noise or high-vibration activities
- Emergency procedures if integration impacts live operations
Transition and Change Management:
Operational readiness goes beyond the physical build. Plan for:
- Staff training on new cleanroom gowning, entry, and operating protocols
- Workflow adjustments as processes move into the new controlled environment
- SOP updates reflecting new room classifications, pressure differentials, and monitoring requirements
Integration Process: Step-by-Step
The integration sequence follows a strict order: site preparation → utility rough-in → modular component installation → system integration → controls commissioning. Shortcuts in this sequencing cause validation failures and costly rework. Each phase below builds on the last — skipping ahead or overlapping stages is where most integration projects lose time and budget.
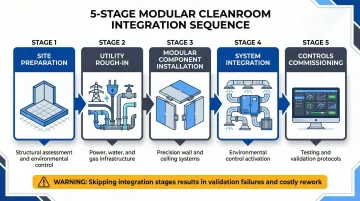
Site Preparation and Infrastructure Upgrades
Before a single panel goes up, the host facility needs to be ready. This phase addresses the surfaces, utilities, and structural elements that modular components will depend on.
Site preparation covers three areas:
- Apply epoxy coating or sealing to floors to create a cleanable, non-shedding surface
- Install or reinforce ceiling support systems rated for cleanroom component loads (approximately 20 lbs/ft² for walkable ceilings)
- Erect temporary containment walls with negative pressure to protect adjacent operations during construction
Utility upgrades run in parallel:
- Install dedicated electrical panels sized for cleanroom loads — typical installations require 200–400A service depending on room size and classification
- Add dedicated air handling units (AHUs) where existing systems lack capacity; size for required ACH rates plus 20–30% future expansion
- Extend compressed air, specialty gas lines, DI water, and drainage to cleanroom locations
Where the existing structure needs reinforcement:
- Ceiling support upgrades for walkable ceiling systems
- Floor reinforcement where heavy equipment exceeds the standard 200 lbs/ft² rating
- Wall anchoring preparations to support modular panel attachment loads
Modular Cleanroom Installation
With site prep complete, modular components can be installed. Precision matters here — tolerance errors in early stages compound through every subsequent phase.
Wall panels — typically 40mm or 60mm thick with aluminum honeycomb or rock wool cores and non-shedding uPVC or stainless steel facings — follow a defined installation sequence:
- Begin from corners, establishing square and plumb reference points
- Seal panel joints using rated gaskets or sealant
- Seal floor and ceiling interfaces to maintain pressure differentials
- Integrate doors, pass-throughs, and vision panels as panels are installed
- Install cleanroom coving at wall-floor junctions to eliminate particle traps
The ceiling system follows wall installation. Install rated grid support structures for walkable ceilings or HEPA filter module mounting. Seal all ceiling penetrations and route lighting, HVAC, and utility lines through the ceiling plane while maintaining full envelope integrity.
Flooring selection depends on application:
- Epoxy: Seamless and chemical-resistant — the standard choice for pharmaceutical and biotech
- Vinyl sheet: Cost-effective and easy to clean, well-suited for ISO 7–8 environments
- Raised access floor: Enables underfloor utility distribution for layouts requiring frequent reconfiguration
All flooring installs with seamless techniques and coved corners at wall junctions.
HVAC and Air Filtration Integration
HVAC integration is where most cleanroom projects either succeed or struggle. Connecting air systems to an existing facility infrastructure requires precise sequencing and leak-free execution.
AHU connection involves three elements:
- Route sealed ductwork from existing or new AHUs to the cleanroom supply points
- Install HEPA/ULPA filter housings with gel or gasket seals to prevent bypass leakage
- Seal all duct penetrations through the cleanroom envelope to maintain pressure integrity
Air distribution positioning determines whether the room meets its ISO classification in practice:
- Place FFUs or diffusers to achieve required airflow patterns — unidirectional for ISO 5, turbulent for ISO 7–8
- Locate return air grilles to sustain intended flow patterns without short-circuiting
- Install balancing dampers for precise volume control across multiple zones
Pressure differentials are verified against regulatory requirements before commissioning:
- Install pressure sensors and controllers in each zone
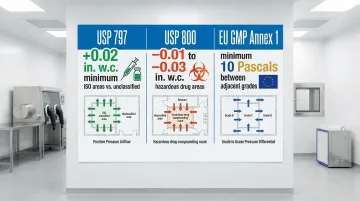
- Configure supply/exhaust balance to achieve required differentials:
- USP <797>: Minimum +0.02 in. w.c. between ISO areas and unclassified spaces
- USP <800>: -0.01 to -0.03 in. w.c. for hazardous drug areas
- EU GMP Annex 1: Minimum 10 Pascals between adjacent grades
- Confirm pressure cascade flows from cleanest to least clean areas
Electrical and Control Systems Integration
The final installation phase brings electrical systems and controls online. This stage also generates the documentation trail that validation depends on.
Electrical work covers:
- Cleanroom-rated lighting fixtures with sealed housings to prevent particle ingress
- Outlets and equipment connections positioned for process equipment with adequate circuit capacity
- Backup power connections for critical systems — HVAC and monitoring must stay live during facility power events
Control system integration ties the cleanroom into existing facility infrastructure:
- Install temperature, humidity, and pressure sensors with continuous data logging
- Integrate with Building Management Systems (BMS) via protocols like BACnet for centralized monitoring
- Configure alarm thresholds for out-of-specification conditions with defined escalation protocols
Documentation infrastructure runs alongside control system setup:
- Install continuous monitoring equipment required for validation and ongoing compliance
- Connect environmental data streams to existing QMS platforms
- Configure automated reporting outputs to satisfy regulatory documentation requirements
Post-Integration Validation and Commissioning
Validation follows a strict sequence: visual inspection → leak testing → airflow verification → particle count testing → environmental parameter verification. Skipping or reordering any step risks missing defects that become exponentially more expensive to fix after the cleanroom is operational.
Critical Validation Tests
HEPA Filter Integrity Testing:
Using ISO 14644-3 and IEST-RP-CC034 protocols, perform aerosol challenge tests:
- Introduce PAO or other test aerosol upstream of filters
- Scan filter faces and seals with photometer or particle counter
- Identify and repair any leaks exceeding 0.01% penetration
Airflow Verification:
- Smoke studies: Visualize airflow patterns to confirm unidirectional flow (ISO 5) or proper mixing (ISO 7-8)
- Velocity measurements: Verify face velocities meet specifications (typically 90 ±20 fpm for unidirectional flow)
- Air change rate verification: Confirm actual ACH meets design requirements
Particle Count Testing:
Per ISO 14644-1 classification requirements:
- Test at multiple locations throughout cleanroom
- Perform under three operational states: as-built (empty), at-rest (equipment installed), and operational (normal production)
- Verify particle concentrations meet ISO class limits (e.g., ≤352,000 particles/m³ ≥0.5 µm for ISO 7)
Pressure Differential Verification:
- Measure pressure differentials between all adjacent zones
- Verify cascade flows from cleanest to less clean areas
- Document that differentials meet regulatory minimums
Once testing is complete, results feed directly into the qualification documentation package required for regulatory approval and ongoing audits.
Documentation Requirements
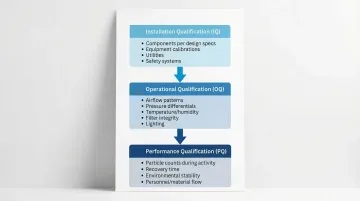
Installation Qualification (IQ):
- All components installed per design specifications
- Equipment calibrations are current
- Utilities meet design requirements
- Safety systems function properly
Operational Qualification (OQ):
- Airflow patterns and velocities
- Pressure differentials
- Temperature and humidity control
- Filter integrity
- Lighting levels
Performance Qualification (PQ):
- Particle counts during activity
- Recovery time after contamination events
- Environmental stability during process simulation
- Personnel and material flow impacts
These three qualification reports — IQ, OQ, and PQ — form the audit-ready evidence package that regulators and quality teams reference throughout the cleanroom's operational life.
Common Integration Challenges and Solutions
Utility System Overload
Problem: Electrical or HVAC systems prove undersized for cleanroom loads mid-integration — typically because load analysis underestimated demand, especially the high air change rates required for lower ISO classifications.
Solution:
- Install dedicated electrical panels or AHUs specifically for cleanroom
- Phase cleanroom operations to work within existing utility capacity
- Upgrade main service if required for full operation
Structural Clearance Issues
Problem: Insufficient ceiling height or floor-to-deck obstructions (beams, ductwork, conduit) block installation — usually traced to a site survey that didn't account for actual component dimensions and installation clearances.
Solution:
- Specify low-profile cleanroom components (reduced-depth ceiling systems)
- Reroute obstructing utilities before cleanroom installation
- Adjust cleanroom layout to avoid problem areas
- As a last resort, lower the floor slab or raise the roof deck
Operational Disruption Exceeds Plan
Problem: Installation work — drilling, utility tie-ins, material handling — creates contamination or noise that pushes disruption well beyond scheduled windows, stalling adjacent sensitive processes.
Solution:
- Implement enhanced contamination control barriers with negative pressure isolation
- Schedule high-impact work (core drilling, major utility tie-ins) during planned facility shutdowns
- Keep operations teams updated daily on scope changes and revised impact timelines
- Build schedule buffers specifically for unexpected disruptions
Pro Tips for Successful Integration
These three practices consistently separate smooth integrations from costly mid-project corrections:
Bring in specialists at the assessment stage — not after. Engage experienced modular cleanroom firms like ACH Engineering before facility teams finalize plans. Specialists catch utility conflicts, structural constraints, and compliance gaps early, when changes cost far less than they do mid-build.
Build in 20-30% excess capacity from day one. Oversize electrical panels, HVAC systems, and modular expansion zones during initial integration. Most facilities expand cleanroom space within 3-5 years — retrofitting later costs significantly more than planning for growth upfront.
Document as-builts, validation data, and procedures in real time. Maintain detailed drawings showing actual utility routing, calibration records, and operational procedures from the first day of operation. Keep physical and digital copies with version control. Regulators, auditors, and your own troubleshooting team will depend on this record when it matters most.
Frequently Asked Questions
How long does modular cleanroom integration typically take compared to traditional construction?
Modular integration typically requires 8-16 weeks from site preparation to validation versus 20-30 weeks for traditional stick-built construction. The advantage comes from parallel processing: modular components are fabricated off-site while site preparation occurs, compressing the overall schedule by 40-60%.
Can we integrate a cleanroom without completely shutting down adjacent operations?
Yes, with proper planning. Most integrations proceed with minimal operational disruption using phased approaches, contamination barriers with negative pressure containment, and careful scheduling. Typically only short shutdowns (4-8 hours) are required during critical tie-ins to existing utilities like electrical panels or HVAC systems. The key is working with experienced integrators who understand contamination control during construction.
What are the main cost differences between retrofitting with modular cleanrooms versus new construction?
Modular integration typically costs 15-30% less than traditional construction due to reduced labor, faster installation, and less operational downtime. Additional savings come from reduced material waste and the ability to depreciate modular cleanrooms over 7 years (as equipment) versus 39 years for traditional construction. However, complex retrofits requiring extensive utility upgrades may narrow this advantage.
Do modular cleanrooms meet the same ISO and regulatory standards as traditionally built cleanrooms?
Modular cleanrooms achieve identical ISO classifications and regulatory compliance as traditional construction when properly designed, installed, and validated. Compliance is performance-based—regulators measure particle counts, pressure differentials, and environmental controls, not construction methodology. Factory-manufactured precision often delivers more consistent performance than field-built alternatives.
What existing building conditions are required for successful cleanroom integration?
Minimum requirements include ceiling height of 10-12 feet, floor load capacity of 100-150 lbs/ft² (higher for heavy equipment zones), adequate electrical service (calculate based on cleanroom size and ISO class), and HVAC capacity for required air changes. However, experienced providers like ACH Engineering can engineer solutions for challenging sites through utility routing, structural reinforcement, or layout optimization.
How do we choose the right ISO classification for our cleanroom needs?
ISO classification must match your process requirements, product sensitivity, and regulatory standards. Over-specifying raises costs; under-specifying risks compliance failures — sterile filling requires ISO 5, while packaging may only need ISO 8. ACH Engineering works across pharmaceutical, biotech, medical device, and electronics industries to help match your processes to the right classification.
Need expert guidance on integrating a modular cleanroom into your existing facility? ACH Engineering specializes in controlled environment solutions across pharmaceutical, biotech, medical device, and other regulated industries throughout North America. Contact their team at +1 647-406-5721 or sales@achengineering.com for a site assessment and integration plan built around your facility and process needs.


