
Introduction
A single cardboard box wheeled into a pharmaceutical cleanroom can release millions of cellulose fibers, each capable of transporting bacterial spores. That unauthorized pen in a technician's pocket? It's shedding graphite particles that will contaminate your ISO 5 environment for hours. One instance of non-compliant material can compromise months of sterile manufacturing, triggering costly product recalls, regulatory violations, and failed certifications.
Material selection directly impacts contamination control effectiveness, product quality, worker safety, and operational costs. The wrong choice creates particle generation, chemical contamination, or microbial harboring that undermines even the most sophisticated HVAC and filtration systems.
This guide explains what materials you can and cannot use in cleanrooms, why these restrictions exist, and how to make informed decisions based on your specific ISO classification and industry regulations.
TLDR
- Cleanrooms require strict material controls to prevent particulate, chemical, and biological contamination that compromise product quality
- Prohibited materials include cardboard, wood, unsealed metals, regular paper, personal items, and non-approved plastics
- Compliant materials must be non-shedding, non-reactive, easy to clean, and compatible with cleanroom disinfectants
- Material requirements vary significantly by ISO classification (3-8) and industry regulations (FDA cGMP, USP, SEMI standards)
- Incorrect material choices are one of the most common audit findings — and among the easiest to prevent with early-stage planning
Understanding Cleanroom Material Requirements
Cleanroom material restrictions are based on three fundamental contamination risks that threaten product quality and regulatory compliance.
- Particle generation: Cellulose-based materials like cardboard and paper shed fibers continuously, even when undisturbed. Cotton lint becomes a direct contamination vector, carrying microorganisms onto sensitive products.
- Chemical contamination: Some plastics and adhesives release volatile organic compounds (VOCs) that condense on wafer or pharmaceutical surfaces. Bare aluminum oxidizes; copper and brass corrode — both introduce chemical contaminants into the environment.
- Microbial harboring: Porous surfaces trap moisture and shelter bacteria, molds, and spores from disinfectants. Untreated paper pulp can contain 10⁸ to 10⁹ colony-forming units per gram, making it incompatible with any controlled environment.

ISO Classification Impact on Material Selection
ISO 14644-1:2015 establishes air cleanliness classifications from ISO Class 1 through ISO 9, with lower numbers indicating stricter particle concentration limits. Material requirements tighten considerably as class numbers decrease.
ISO Classes 3-5 (critical environments like semiconductor fabs and aseptic processing cores) require materials with near-zero particle shedding and ultra-low outgassing. Only non-porous materials resistant to abrasion can meet the particle concentration limits for sizes ranging from 0.1 µm to 5 µm.
ISO Classes 6-8 (support zones and general manufacturing areas) still require easily cleanable materials that don't contribute significantly to particle loads, but material restrictions are less stringent than in critical zones.
Industry-Specific Regulatory Layers
ISO classification sets the baseline — but industry-specific regulations layer additional material requirements on top:
- FDA cGMP (21 CFR 211.42): Requires aseptic processing areas to have floors, walls, and ceilings of smooth, hard surfaces that are easily cleanable
- USP <797>: Explicitly prohibits "nonessential objects that shed particles" such as cardboard, pencils, and cotton items from buffer areas
- USP <800>: Mandates surfaces in hazardous drug compounding areas be smooth, impervious, free from cracks, and non-shedding
- SEMI F21: Classifies airborne molecular contaminant levels for semiconductor manufacturing, driving selection of low-outgassing materials
Prohibited Materials in Cleanrooms
Knowing what to keep out is just as important as knowing what to bring in. The materials below are prohibited or restricted across most cleanroom classifications — from ISO 5 pharmaceutical suites to ISO 8 assembly areas.
Personal Items and Consumables
These items are strictly banned due to particle generation, bacterial harboring, and chemical contamination:
- Food, beverages, and tobacco products: Generate organic particles, introduce moisture, and harbor bacteria
- Cosmetics, perfumes, and colognes: Shed particles and release chemical contaminants that disrupt sensitive processes
- Jewelry (rings, watches, bracelets, earrings): Punctures gloves, harbors microorganisms in crevices, and generates particles through friction
Non-Compliant Construction and Work Materials
Construction material selection directly affects a cleanroom's long-term contamination control performance.
Construction Materials — Strictly Prohibited:
- Cardboard: Sheds cellulose fibers even when undisturbed and is a documented vector for Bacillus and fungal spores — one of the worst contamination sources in any cleanroom
- Unsealed wood and particle board: Porous surfaces allow microbial penetration that cannot be sanitized to cGMP standards
- Galvanized steel: Zinc coating flakes off, generating metallic particles
- Bare aluminum: Oxidizes over time, releasing particles and chemical contaminants
- Copper and brass: Corrode in cleanroom environments, generating particulate and chemical contamination
- Leather: Organic material that sheds particles and cannot be adequately disinfected
Work materials carry similar risks — even everyday office supplies can compromise air quality standards.
Work Materials — Strictly Prohibited:
- Regular paper and notebooks: Cellulose fibers shed with every page turn
- Standard pens and pencils: Graphite cores generate conductive particles; ballpoint ink can also shed during use
- Erasers: Rubber particles contaminate surfaces and are difficult to remove
- Duct tape and cloth tapes: Leave adhesive residue and shed fibers
- Aerosol sprays (except approved types): Propellants and particles disperse uncontrollably

Cleaning and Maintenance Materials
Prohibited Cleaning Items:
- Household vacuum cleaners: Recirculate particles through exhaust instead of capturing them — HEPA-rated cleanroom vacuums are required
- Cotton rags and towels: Generate significant lint with every use
- Abrasive cleaning pads (steel wool, Scotch-Brite): Release metal particles that contaminate surfaces
- Unapproved chemical cleaners: Leave residues incompatible with cleanroom surfaces or degrade materials
Restricted Materials Requiring Approval
Some materials fall into a "restricted" category — permitted only with cleanroom manager approval under controlled conditions:
- Certain adhesives for specific applications (must be low-outgassing and tested)
- Powders in sealed containers (must remain sealed except during controlled use)
- Open-cell foams if properly contained (generally avoided due to particle trapping)
- Books, magazines, and reference materials (cleanroom-compatible alternatives or digital versions strongly preferred)
Acceptable Cleanroom Materials
Cleanroom-Compatible Personal Protective Equipment
All PPE must be manufactured specifically for cleanroom environments — not repurposed from general industrial use:
- Lint-free coveralls made from polyester or Tyvek® (high-density polyethylene), tested for particle generation
- Nitrile or latex gloves — powder-free, rated for cleanroom use
- Hair covers and beard nets in synthetic materials that fully contain skin and hair particles
- Face masks with verified low particle-shedding performance
- Dedicated cleanroom footwear or shoe covers that won't track contaminants onto the floor
Approved Construction and Structural Materials
Stainless Steel
- 304 and 316 series are the standard — both offer strong corrosion resistance without generating particles
- Surface finish determines contamination risk: 2B (general purpose), No. 4 dairy finish (equipment), or electropolished (lowest risk)
- All stainless steel must be passivated or electropolished before installation
Other Approved Metals
- Anodized or powder-coated aluminum — the sealed finish blocks oxidation and particle release
- Chrome-plated metals — smooth, non-reactive, and easy to clean
Wall and Ceiling Systems
- Modular cleanroom wall panels with smooth, non-porous surfaces rated for frequent chemical cleaning
- Epoxy or polyurethane floor and wall coatings — seamless and chemical-resistant
- Cleanroom-grade ceiling tiles and grid systems that won't shed particles into HEPA airflow
ACH Engineering's modular cleanroom systems use materials pre-vetted for cleanroom performance — including High Pressure Laminate (HPL), powder-coated galvanized iron, and u-PVC panels. Each system is built to ISO Class 1–9 standards and designed from the start to meet FDA and GMP requirements.
Cleanroom-Approved Work Materials
Beyond the structure itself, everything brought inside the cleanroom for day-to-day work must meet the same standard.
Acceptable work supplies include:
- Synthetic paper notebooks (polyethylene or polypropylene) — no cellulose fibers to shed
- Indelible ballpoint pens tested for particle generation in controlled environments
- Cleanroom-compatible tape that leaves no residue and doesn't release fibers
- Low-outgassing adhesives and sealants tested for cleanroom compatibility
- Static-dissipative containers and trays for electronics and semiconductor environments
- Cleanroom-grade plastics: polypropylene, polycarbonate, or filler-free PVC
Cleaning and Maintenance Materials
Cleaning protocols are only as effective as the supplies used to execute them.
Approved cleaning supplies:
- HEPA-filtered cleanroom vacuums — capture particles without recirculating them
- Lint-free polyester or microfiber wipes tested for low particle generation
- Pre-saturated cleanroom mops for controlled, consistent application of cleaning agents
- Sterile disinfectants: isopropyl alcohol (IPA), hydrogen peroxide, and quaternary ammonium compounds, all verified compatible with cleanroom surfaces
- Cleanroom-grade detergents formulated to leave zero residue

Approved Chemicals and Process Materials
Any chemical brought into a cleanroom must meet these criteria, regardless of process:
- Documentation: Current Material Safety Data Sheets (MSDS) or Safety Data Sheets (SDS)
- Proper storage: Approved containers that don't shed particles or leach contaminants
- Designated use areas: Fume hoods or controlled zones if required by safety protocols
- Material compatibility: Verified compatibility with cleanroom materials and finishes
Critical Factors for Material Selection
Particle Generation and Shedding Potential
This is the PRIMARY consideration for cleanroom materials. Materials must demonstrate minimal particle shedding under normal use, friction, and wear.
Testing methods include:
- ASTM F51 standard test for sizing and counting particulate contaminant in cleanroom garments
- IEST-RP-CC004 for evaluating wiping materials
- Visual inspection under magnification
Particle generation directly affects ISO classification compliance and product contamination risk. Even materials that appear smooth can shed unacceptable particle levels under cleanroom conditions.
Chemical Compatibility and Outgassing
Materials must withstand repeated exposure to the cleaning agents used in your environment:
- Isopropyl alcohol (IPA)
- Hydrogen peroxide (including vaporized hydrogen peroxide for decontamination)
- Quaternary ammonium compounds
- Phenolic disinfectants
- Sporicidal agents
Beyond chemical resistance, outgassing is a separate concern that's easy to overlook. Some plastics and adhesives release volatile organic compounds (VOCs) that:
- Condense on wafer surfaces causing defects
- Contaminate pharmaceutical products
- Interfere with analytical processes
Material data sheets must be reviewed for outgassing characteristics. ISO 14644-8 establishes protocols for testing material emissions and classifying Air Chemical Cleanliness (ACC).
Surface Characteristics and Cleanability
Cleanroom materials must have smooth, non-porous surfaces that don't trap particles or harbor microorganisms.
Key surface requirements:
- Low surface roughness: Quantified by Roughness Average (Ra) values—lower is better
- Seamless construction: Eliminates particle accumulation points
- Coved corners: Preferred over square corners to prevent particle trapping
- Durability: Must withstand repeated cleaning and disinfection without degrading or shedding particles
FDA guidance explicitly mandates "smooth, hard surfaces that can be easily cleaned" for pharmaceutical manufacturing areas.
Electrostatic Properties
For electronics manufacturing:
- Materials must be static-dissipative or conductive to prevent electrostatic discharge (ESD) damage
- ANSI/ESD S20.20 establishes requirements for ESD control programs
- Worksurfaces must meet resistance limits (typically < 1.0 × 10⁹ ohms)
- Isolated conductors must be kept to < 35 volts
For pharmaceutical cleanrooms:
- Different electrostatic properties may be required to prevent particle attraction
- Static control must be balanced against other contamination control needs
Regulatory and Industry-Specific Requirements
FDA-regulated industries must use materials that:
- Comply with cGMP requirements
- Don't leach extractables or leachables into products
- Support validation and cleaning verification protocols
Aerospace and defense cleanrooms may have additional material restrictions for security or performance reasons, including requirements for vacuum outgassing testing per ASTM E1559.
Semiconductor manufacturing requires materials meeting SEMI standards for airborne molecular contamination and particle generation.
Because requirements vary significantly between industries—and sometimes between facilities in the same industry—material selection decisions should always be validated against your specific ISO classification, regulatory framework, and process chemistry before finalizing any specification.
How ACH Engineering Can Help
ACH Engineering is a specialized controlled environment solutions provider with deep expertise in material selection and cleanroom compliance across diverse industries. Their end-to-end approach covers material compliance from initial design through final validation.
Material Specification as Part of Design Services: ACH specifies materials during the design phase, ensuring all construction finishes and systems meet ISO 14644 standards from project inception. Their engineering team handles sourcing, vendor coordination, and price negotiation to secure pre-approved, compliant materials.
Pre-Approved Modular Cleanroom Solutions: ACH's modular cleanroom systems come with pre-approved materials including:
- High Pressure Laminate (HPL) wall panels for durability and customization
- Powder-coated galvanized iron for corrosion-resistant construction
- u-PVC panels for chemical resistance and cost-effectiveness
- Stainless steel frameworks and appropriate coatings
All materials are prefabricated offsite, ensuring consistent quality control and reducing onsite cleanroom installation to a few weeks after component delivery.
Operational Materials Guidance: ACH extends compliance guidance beyond structural materials to operational consumables. This includes:
- PPE selection appropriate to your ISO classification
- Cleaning agents and disinfectants compatible with wall and floor finishes
- Work materials and packaging approved for your contamination control requirements
Industry-Specific Expertise: With experience serving pharmaceutical, biotech, electronics, food & beverage, and healthcare sectors, ACH understands nuanced material requirements for:
- Health Canada GMP and FDA cGMP compliance for pharmaceutical manufacturing
- USP <797>/<800> standards for compounding pharmacies
- ESD-safe environments for semiconductor and electronics manufacturing
- Biological Safety Level requirements for biotech research
- Food safety regulations for processing facilities
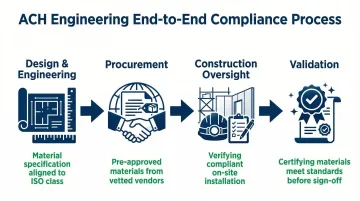
End-to-End Compliance Support: ACH manages material compliance across every project phase:
- Design & Engineering — material specification aligned to your ISO class and regulatory framework
- Procurement — sourcing pre-approved materials from vetted vendors
- Construction Oversight — verifying compliant installation on site
- Validation — certifying that all materials meet specified standards before final sign-off
For teams navigating material compliance for the first time — or managing complex multi-standard environments — ACH's structured process removes the guesswork at every stage.
Conclusion
Successful cleanroom operation starts with rigorous material control. Every item brought into the cleanroom either supports or undermines your contamination control efforts. A single non-compliant material can compromise product quality, trigger regulatory violations, and waste months of careful work.
Material selection is not a one-size-fits-all checklist. Your specific ISO classification, industry regulations, and process requirements determine what's acceptable. For example:
- An ISO 7 medical device assembly room has very different material tolerances than an ISO 4 semiconductor fab with strict airborne molecular contamination limits
- Materials approved for general pharmaceutical manufacturing may not survive vaporized hydrogen peroxide decontamination cycles required in sterile compounding
- Electrostatic properties that are irrelevant in a biotech lab become critical in battery dry rooms and electronics manufacturing
Balancing particle generation, chemical compatibility, cleanability, electrostatic properties, and regulatory requirements is genuinely complex work. ACH Engineering brings end-to-end expertise to this process — from initial design through material specification and construction — helping clients across pharmaceutical, semiconductor, medical device, and other regulated industries make compliant, durable material decisions the first time.
Frequently Asked Questions
Can I bring my phone or laptop into a cleanroom?
Standard personal electronics are restricted — ports, seams, and screens shed particles and trap contaminants that can't be adequately cleaned. Only cleanroom-compatible models with sealed, non-shedding surfaces may enter, subject to management approval.
Why is cardboard prohibited in cleanrooms?
Cardboard is highly fibrous and continuously sheds cellulose particles even when undisturbed, making it one of the worst contamination sources. It's also porous, retaining moisture that supports bacterial and mold growth. Use cleanroom-approved plastic or metal containers instead.
What's the difference between cleanroom-approved paper and regular paper?
Cleanroom paper is made from synthetic materials (polyethylene, polypropylene) that don't shed fibers like cellulose-based regular paper. It's specifically tested for particle generation and designed to withstand cleanroom handling without contamination.
Are all stainless steel grades acceptable for cleanrooms?
No. 300-series stainless steel (304 or 316) is preferred — both offer strong corrosion resistance and won't generate particles, with 316 providing added durability against aggressive disinfectants. Other grades risk corrosion or particle shedding under cleanroom conditions.
How do I know if a material is cleanroom-compatible?
Start by confirming manufacturer certification and reviewing particle generation test data (ASTM F51 or IEST standards). Then verify chemical compatibility with your cleaning agents. ACH Engineering can assess any material against your specific ISO classification and regulatory requirements.
Can I use regular cleaning supplies in a cleanroom?
No. Regular cleaning supplies are prohibited because they shed lint, may contain additives incompatible with cleanroom surfaces, and lack particle generation testing. Only cleanroom-certified cleaning materials—including HEPA-filtered vacuums, lint-free wipes, and approved disinfectants—should be used to maintain contamination control.


