
Introduction
Selecting the right wall coating for your cleanroom is a critical contamination control decision affecting compliance, product quality, and operational efficiency. The wrong choice leads to particle shedding, failed audits, and costly downtime.
In 2023-2024, FDA warning letters explicitly cited wall surface deficiencies like peeling coatings and non-cleanable finishes in aseptic areas. Wall integrity isn't optional—it's a regulatory imperative.
This guide covers selection criteria and application best practices, helping you balance technical performance, compliance, and budget.
TL;DR
- Cleanroom wall coatings provide seamless, non-porous barriers that prevent particle generation and enable rigorous cleaning
- Selection criteria include cleanroom classification, chemical resistance, durability, substrate type, and cure time
- Application success depends equally on surface prep, environmental controls, and post-application validation
- Pharmaceutical cleanrooms need aggressive chemical resistance; semiconductor facilities require low outgassing materials
- Proactive maintenance and recoating preserve compliance with ISO, FDA, and GMP standards
What is Cleanroom Wall Paint?
Cleanroom wall coatings are specialized systems designed to create smooth, seamless, non-particle-shedding surfaces that withstand rigorous cleaning while maintaining contamination control.
Unlike conventional paints, these coatings must meet stringent requirements for low VOC emissions (often <50 g/L), non-outgassing properties, and resistance to harsh sanitizing agents like hydrogen peroxide vapor and bleach solutions.
Primary Coating Types
Epoxy-Based Systems: These two-component coatings offer superior chemical resistance, excellent adhesion to various base materials (substrates), and durability under aggressive cleaning protocols. Epoxies deliver exceptional hardness and structural integrity.
However, they require longer cure times—typically 7-14 days for full chemical resistance—and can yellow under UV exposure.
Polyurethane Systems: Flexible coatings with faster cure times (24-48 hours for light operations), good UV resistance, and moderate chemical resistance. They offer superior color stability and flexibility compared to epoxies, making them ideal for topcoats where appearance matters and quick turnaround is essential.
Hybrid Systems: These polymer combinations (epoxy-urethane blends) balance the benefits of both chemistries. A common approach: fiber-reinforced epoxy build coats for structural durability, topped with polyurethane for chemical resistance and color retention.

Core Components and Properties
Regardless of which coating type you select, understanding the layering system is essential for performance.
A typical multi-layer system includes:
- Primer coat — Promotes adhesion and seals base material (substrate) porosity
- Build coats — Builds thickness, impact resistance, and smooth foundation (often 10-15 mils in high-traffic areas)
- Topcoat — Offers primary chemical resistance, cleanability, and surface finish
Essential properties for all cleanroom coatings:
- Seamless finish with no pinholes
- Non-particle shedding characteristics
- Complete barrier (impermeability) to moisture and contaminants
- Resistance to microbial growth
- Prevention of Airborne Molecular Contamination (AMC) in semiconductor applications
Benefits of Proper Cleanroom Wall Coating Systems
Choosing the right wall coating system directly impacts your cleanroom's performance, regulatory standing, and long-term operational costs. The benefits extend across three critical areas.
Contamination Control:
- Prevents particle generation from substrate materials
- Creates easily cleanable surfaces that don't harbor bacteria or mold
- Maintains air quality by preventing outgassing of volatile compounds
- Eliminates gaps and joints where contaminants accumulate
Meeting regulatory standards isn't optional in controlled environments. Proper coating systems provide the documentation and performance auditors expect.
Regulatory Compliance:
- Meets FDA 21 CFR 211.42 pharmaceutical facility requirements for "smooth, hard surfaces that are easily cleanable"
- Supports ISO 14644-1 cleanroom classification standards through reduced surface particle emission
- Aligns with EU GMP Annex 1 requirements for "smooth, impervious, and unbroken" surfaces in sterile manufacturing
The operational advantages of premium coating systems become clear within the first year of use.
Operational Efficiency:
- Extends time between deep cleaning cycles through smooth, non-porous surfaces that resist particle adhesion
- Reduces maintenance costs via durability (15-20 years for premium systems)
- Minimizes unplanned downtime from coating failures
- Reduces risk of audit failures and warning letters
- Delivers ROI within 3-5 years through reduced cleaning and maintenance expenses

What to Consider When Selecting Cleanroom Wall Paint
Coating selection must align with specific operational requirements, cleanroom classification, industry regulations, and budget constraints.
The following six factors help facilities connect technical coating specifications to measurable business outcomes like compliance, longevity, and total cost of ownership.
Cleanroom Classification and Industry Standards
ISO 14644-1 classifications (Class 1-9) determine coating requirements based on allowable airborne particle concentration. Stricter classifications demand more robust systems:
- ISO Class 3-5 (pharmaceutical aseptic, semiconductor): Requires virtually zero particle generation, seamless application, and resistance to frequent intensive cleaning
- ISO Class 6-8 (medical device, compounding): Still requires non-shedding, cleanable surfaces but with less aggressive cleaning frequency
Industry-Specific Requirements:
| Industry | Key Requirement | Regulatory Driver |
|---|---|---|
| Pharmaceutical | Chemical resistance, microbial control | FDA 21 CFR 211.42, EU GMP Annex 1 |
| Semiconductor | Low outgassing, zero particle generation | ISO 14644, IEST-RP standards |
| Medical Device | Non-porous, cleanable surfaces | FDA 21 CFR 820, ISO 13485 |
| Food & Beverage | Washdown resistance, non-toxic | FDA 21 CFR 117.20 |
Chemical Resistance Requirements
Match your coating system to the cleaning agents and disinfectants used in daily operations:
- Vaporized Hydrogen Peroxide (VHP) - Requires specialized urethane topcoats; standard epoxies may degrade under 600 ppm exposure
- Isopropyl Alcohol (70%) - Most high-performance epoxies and urethanes show excellent resistance
- Sodium Hypochlorite (Bleach) - Can discolor epoxies; urethanes offer better color stability
- Quaternary Ammonium Compounds - Generally compatible with both epoxy and polyurethane systems
Testing and Selection:
- Obtain chemical resistance charts from coating manufacturers
- Conduct compatibility testing with your specific cleaning protocols
- Pharmaceutical and healthcare cleanrooms requiring frequent sterilization need epoxy or specialized polyurethane systems
- Less aggressive environments can use standard polyurethanes

Durability and Abrasion Resistance
Consider impact and abrasion resistance based on operational demands:
Performance Specifications:
- Abrasion resistance (ASTM D4060): High-performance urethanes show 68-84 mg weight loss after 1,000 cycles
- Impact resistance (ASTM D2794): Systems should withstand 140+ in-lbs without cracking
- Hardness (ASTM D3363): Pencil hardness ratings of 4H indicate hard, scratch-resistant finish
High-traffic corridors with cart traffic require thicker mil builds (10-15 mils) or reinforced systems. Low-traffic areas can use thinner applications.
Damaged coatings expose substrate material that generates particles and harbors contaminants.
Surface Preparation and Substrate Compatibility
Coating performance depends heavily on proper substrate preparation. Most coating failures trace back to inadequate surface prep.
Common Substrates and Requirements:
- Concrete/CMU - Clean per ASTM D4258, profile per ASTM D4259 (abrasive blasting), test moisture per ASTM D4263
- Drywall - Requires specific primer to seal porous surface before high-performance coatings
- Metal Panels - Prepare per SSPC-SP 16 (minimum 0.75 mil profile for adhesion)
- Existing Painted Surfaces - Verify compatibility, mechanically abrade if outside recoat window
Moisture testing is critical for concrete substrates.
Use ASTM D4263 (plastic sheet method) or ASTM F2170 (in-situ relative humidity) to prevent delamination from capillary moisture.
Curing Time and Operational Downtime
Cure times directly impact facility shutdown duration and production losses:
| System Type | Tack-Free | Light Traffic | Full Chemical Cure |
|---|---|---|---|
| Standard Epoxy | 2-5 hours | 16-24 hours | 7 days |
| Fast-Cure Epoxy | 1-2 hours | 4-8 hours | 4-7 days |
| Polyurethane | 4-9 hours | 12-24 hours | 5-7 days |
| VHP Resistance | N/A | N/A | Up to 14 days |
Planning Considerations:
- Faster cure times often compromise chemical resistance or durability
- Plan phased applications to minimize disruption
- Control temperature and humidity during curing (ideal 50-90°F, <85% RH)
- Conduct validation testing before returning room to service
Budget and Total Cost of Ownership
Consider all cost components beyond material price:
- Material costs (typically $2-8 per square foot)
- Surface preparation expenses (often the largest labor component)
- Labor for application
- Downtime costs (frequently exceeds contract value)
- Expected lifespan (15-20 years for premium vs. 5-7 years for budget systems)
Hidden Costs of Failure:
- Emergency repairs at premium rates
- FDA warning letters and compliance issues
- Contamination events leading to product recalls
- Production losses during unplanned shutdowns
Higher-quality systems have higher upfront costs but lower total cost of ownership. Calculate the full lifecycle cost, not just initial investment.

Application Best Practices for Cleanroom Wall Coatings
Proper application technique is as critical as material selection—poor application of premium coatings yields worse results than expert application of mid-grade products. Following manufacturer specifications and industry best practices ensures optimal performance and longevity.
Surface Preparation Requirements
Substrate Assessment:
- Conduct moisture testing for concrete (calcium chloride or relative humidity testing per ASTM standards)
- Evaluate surface profile to ensure adequate mechanical anchor
- Identify defects requiring repair (cracks, spalling, previous coating delamination)
Cleaning Protocols:
- Remove dust, oils, previous incompatible coatings, and contaminants
- Use mechanical preparation (grinding, shot blasting) for concrete to achieve proper profile
- Apply chemical cleaning for metal substrates per SSPC-SP 16
Inadequate surface preparation causes most premature coating failures, including delamination, bubbling, and poor adhesion.
Environmental Controls During Application
Optimal Application Conditions:
- Temperature: 50-90°F (10-32°C)
- Relative humidity: Below 85%
- Dew point: Surface temperature must be 5°F above dew point to prevent condensation
Critical Application Controls:
- Control airborne particulate through temporary containment and filtration
- Provide adequate ventilation to manage VOC levels and ensure proper cure
- Monitor temperature closely—low temperatures slow cure rates significantly
- Watch humidity levels—high humidity can cause amine blush in epoxies
Application Techniques and Quality Control
Recommended Application Methods:
- Airless spray: Preferred for large areas, delivers uniform seamless coverage
- Roller application: Used for smaller areas, requires strict adherence to wet film thickness targets
Quality Control Measures:
- Use wet film thickness gauges (ASTM D4414) to verify specified thickness during application
- Conduct visual inspection for uniformity, runs, sags, and thin spots
- Document application conditions (temperature, humidity) and material batch numbers
- Monitor environmental conditions continuously to ensure parameters stay within TDS limits
Achieving specified wet film thickness ensures proper dry film thickness and performance. Most systems target 4-8 mils WFT for primers and topcoats.
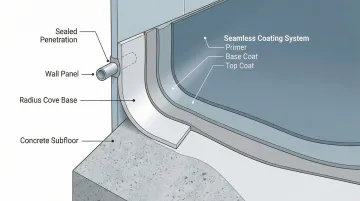
Cove Base and Seam Details
Beyond flat surface application, proper detailing at transitions and penetrations ensures complete cleanroom integrity.
Integral Cove Bases: Create radius coves at floor-to-wall transitions using trowelable epoxy systems or preformed cove base materials. This eliminates 90-degree corners where contaminants accumulate and creates easily cleanable surfaces compliant with GMP requirements.
Penetration Sealing: Properly coat around doors, windows, electrical boxes, and HVAC penetrations to maintain seamless barrier. Use compatible, cleanroom-grade sealants to maintain air seal and prevent particle ingress. Gaps in these areas are frequent citations in FDA inspections.
Post-Application Validation and Testing
Visual Inspection Criteria:
- Uniform colour and gloss appearance
- Freedom from pinholes, blisters, peeling, or cracking
- Proper coverage at edges, corners, and details
Testing Protocols:
- Dry film thickness - Verify using magnetic (SSPC-PA 2) or ultrasonic gauges to ensure system meets specification
- Adhesion testing - Conduct ASTM D3359 (tape test) or ASTM D4541 (pull-off test) if required, though these are destructive
Allow full cure before validation testing and returning the cleanroom to service.
While light traffic may be permitted within 24 hours, full chemical resistance to aggressive agents like VHP typically requires 7-14 days depending on product and environmental conditions.
How ACH Can Help
ACH Engineering is a specialized controlled environment solutions provider with expertise in cleanroom design, construction, and optimization across pharmaceutical, biotech, medical device, and other regulated industries throughout North America. Operating across Canada and North America, ACH brings proven engineering capabilities with memberships in the International Society for Pharmaceutical Engineering (ISPE) and Professional Engineers Ontario (PEO).
ACH specifies appropriate wall coating systems based on your cleanroom classification, operational requirements, and budget. The engineering team ensures wall surfaces meet critical requirements:
- Smooth, non-porous finish for easy cleaning
- Non-shedding materials that won't contaminate processes
- Chemical compatibility with your cleaning protocols
- ISO classification and FDA GMP compliance support
Working with trusted coating suppliers and applicators, ACH ensures proper material selection, surface preparation, and application techniques that meet industry standards.
The process starts with understanding your specific requirements, then delivers solutions that balance contamination control, regulatory compliance, and operational efficiency. From initial design through validation and certification, the cleanroom project management team coordinates execution while minimizing downtime.
Conclusion
Selecting the right cleanroom wall coating system requires balancing technical performance requirements, regulatory compliance needs, and budget constraints. The goal isn't simply choosing the most expensive or popular coating, but identifying the system that aligns with your specific cleanroom classification, cleaning protocols, and operational priorities.
Application technique is equally critical. Premium materials applied poorly will underperform mid-grade systems applied expertly. Surface preparation, environmental controls, and proper curing are non-negotiable for long-term success.
To maximize coating system longevity and performance:
- Monitor coating condition through regular visual inspections and maintenance programs
- Plan recoating proactively rather than reactively to avoid compliance gaps
- Document performance trends to optimize future coating selection
With proper selection, application, and maintenance, high-quality coating systems deliver 15-20 years of reliable performance while supporting your contamination control and regulatory compliance objectives.
Frequently Asked Questions
What type of paint is best for pharmaceutical cleanrooms?
Epoxy-based or high-performance polyurethane systems are best due to frequent exposure to aggressive cleaning agents like hydrogen peroxide vapor and isopropyl alcohol. Multi-layer systems with fiber-reinforced epoxy build coats and urethane topcoats offer optimal chemical resistance while meeting FDA GMP requirements.
How often should cleanroom walls be repainted or recoated?
High-quality epoxy systems typically last 10-20 years, while polyurethane systems last 7-15 years, depending on cleaning frequency and operational wear. Conduct annual visual inspections to plan maintenance proactively rather than waiting for coating failure.
Can you paint over existing cleanroom wall coatings?
Yes, if the existing coating is compatible, well-adhered, and properly prepared. If outside the recoat window (typically 24 hours), mechanically abrade the surface to ensure proper adhesion and prevent delamination from incompatible chemistries.
What is the difference between epoxy and polyurethane wall coatings for cleanrooms?
Epoxies offer superior chemical resistance and hardness but cure slowly (7 days) and yellow under UV. Polyurethanes cure faster (24-48 hours) and resist UV better but have lower resistance to aggressive cleaners. Hybrid systems combine both strengths.
How long does cleanroom wall paint take to cure before the room can be operational?
Fast-cure polyurethanes may allow light operations in 24-48 hours, while epoxies typically require 7-14 days for full chemical resistance, particularly against aggressive agents like VHP. Always follow manufacturer specifications for your specific coating system.
Do cleanroom wall coatings need to meet specific FDA or ISO requirements?
FDA and ISO don't specify coating products but require smooth, cleanable, non-shedding surfaces compatible with cleaning agents. FDA 21 CFR 211.42 requires "smooth, hard surfaces that are easily cleanable" in aseptic areas, and coatings must support ISO classification and cleaning validation.


