
Introduction
Contamination control is the cornerstone of medical device manufacturing quality, and human operators remain the single largest contamination source—emitting between 50,000 and 180,000 particles per person per minute. This biological burden accounts for approximately 80% of all particulate contamination in cleanrooms. Automation offers the only viable path to drastically reduce this variable while meeting stringent FDA 21 CFR 820 and ISO 14644 standards.
Cleanroom automation improves consistency, compliance, and throughput — but only when implementation is approached correctly. This guide covers system selection, validation protocols, integration planning, and the scenarios where automation delivers the greatest return for medical device manufacturers.
TL;DR
- Cleanroom automation integrates robotic systems, automated material handling, and environmental monitoring within controlled environments that meet ISO 14644 and FDA compliance standards
- Delivers the greatest value in repetitive precision tasks — assembly, dispensing, inspection, and packaging — where human presence raises contamination risk
- Successful implementation depends on cleanroom design built around automation equipment, validated environmental controls, and cGMP requirements from the outset
- Most facilities reach ROI within 14–20 months, with reported labor cost reductions of 40–60% and throughput gains of 2–3x
How to Implement Cleanroom Automation in Medical Manufacturing
Step 1: Assess Current Processes and Define Automation Scope
Begin with a thorough evaluation of existing manual processes to identify automation candidates based on repetitiveness, precision requirements, contamination risk, and production volume. Not every process benefits equally from automation—focus on operations where human variability directly impacts quality or where contamination risk is highest.
Define specific, measurable automation objectives:
- Throughput improvement targets (e.g., 200–300% increase)
- Contamination reduction goals (e.g., scrap reduction from 3–5% to below 0.5%)
- Regulatory compliance enhancement requirements
Conduct a risk assessment using FMEA (Failure Mode and Effects Analysis) to identify potential failure modes in automated systems and their impact on product quality and patient safety. Completing this before procurement decisions locks in your compliance posture early, when changes are still inexpensive.
Step 2: Design Cleanroom Infrastructure to Support Automation
Ensure your cleanroom classification (ISO Class 5, 6, 7, or 8) matches the medical device risk category and that physical space can accommodate automated equipment. Critical infrastructure considerations include:
Spatial Requirements:
- Adequate ceiling height for robotic reach envelopes
- Floor loading capacity for heavy automation equipment
- Maintenance access for troubleshooting and repairs
Environmental Controls: Plan for adequate air changes per hour (ACH)—ISO Class 5 requires 240–600 ACH, Class 7 needs 60 ACH, and Class 8 requires 20 ACH. HEPA filtration capacity must offset heat loads from automation equipment while maintaining airflow patterns that prevent contamination.
Modular vs. Traditional Construction: Modular cleanroom designs can be assembled on-site in weeks rather than months, with 20–30% lower total costs due to predictable pricing and accelerated tax depreciation (5–7 years vs. 39 years for stick-built facilities). ACH Engineering's modular cleanroom solutions are designed with automation integration in mind, giving medical manufacturers the flexibility to add or reconfigure equipment without extensive facility modifications or production downtime.
Step 3: Select and Integrate Automation Technologies
Evaluate automation technologies appropriate for specific medical manufacturing tasks:
Common Automation Systems:
- Robotic arms for multi-axis assembly, pick-and-place, and repetitive fastening tasks
- Automated dispensing systems for precise adhesive, coating, and fluid application
- Vision systems for inline inspection, dimensional verification, and defect detection
- Automated material handling for contamination-free inter-process transport
Cleanroom Compatibility Requirements: Selected equipment must meet strict cleanroom standards:
- Low particle generation (tested per ISO 14644-14:2016)
- Smooth, crevice-free surfaces for effective cleaning and disinfection
- Materials that don't shed particulates or outgas volatile compounds
- PFPE (perfluoropolyether) lubricants for low vapor pressure
- NSF H1 grade lubricants for medical contact applications
Develop an integration plan connecting automation systems with environmental monitoring, building management systems (BMS), and manufacturing execution systems (MES) for real-time data collection and process control.
Step 4: Validate and Qualify Automated Systems
Validation determines whether your automated systems consistently produce devices that meet specifications—and it's a regulatory requirement, not optional. FDA 21 CFR 820.70(i) explicitly requires validation of computer software used in production or quality systems.
IQ/OQ/PQ Protocol:
- Installation Qualification (IQ): Confirm the equipment is installed per manufacturer specifications, utilities are connected correctly, and documentation is complete
- Operational Qualification (OQ): Test the system across its full operating range to verify it performs as designed under both normal and worst-case conditions
- Performance Qualification (PQ): Run production batches under real-world conditions to demonstrate the process reliably yields conforming product

Perform process validation studies with statistically significant sample sizes. ISO 11462-2 provides tools for determining appropriate sample sizes and implementing statistical process control (SPC).
Establish ongoing monitoring protocols and revalidation schedules. FDA 21 CFR 820.75(b) mandates monitoring and control of process parameters for validated processes, with revalidation triggered by facility, equipment, or process changes.
Step 5: Train Personnel and Establish SOPs
Even the most capable automated system depends on well-trained operators to run correctly. Develop comprehensive training programs covering:
- Automated system operation and troubleshooting
- Emergency procedures and safety protocols
- Cleanroom protocol adherence during automation operation
- Documentation requirements for regulatory compliance
SOPs translate that training into consistent, auditable practice. Create detailed procedures for:
- Automated system startup and shutdown sequences
- Routine maintenance and calibration schedules
- Cleaning and sanitization protocols
- Change control and deviation management
When Should You Implement Cleanroom Automation in Medical Manufacturing
Automation isn't the right fit for every facility. The decision should be driven by specific operational, quality, and business factors rather than technology trends alone.
Ideal Scenarios for Automation:
- High-volume production of Class II/III medical devices requiring consistent precision
- Processes with high contamination risk from human handling
- Operations experiencing labor shortages or high turnover
- Products requiring extensive documentation for regulatory traceability
- Manufacturing lines where scrap rates exceed 3-5% from inconsistent manual processes
Signs Automation May Be Premature:
- Low-volume custom device manufacturing with frequent design changes
- Early-stage R&D with evolving processes
- Operations with limited capital investment capacity
- Processes where human judgment and adaptability are critical to quality decisions

Once production volumes are stable and consistent, the financial case tends to move quickly. Manufacturers typically achieve payback in 14-20 months through labor cost reduction, throughput improvements, and scrap elimination.
What You Need Before Implementing Cleanroom Automation in Medical Manufacturing
Most cleanroom automation projects that stall do so before a single robot is installed — not because of technology gaps, but because the facility, regulatory, and organizational groundwork wasn't laid first.
Infrastructure and Environmental Requirements
Your cleanroom must meet the appropriate ISO classification with validated HVAC systems and documented environmental monitoring. Physical space planning must account for automation equipment footprint, movement ranges, and maintenance access corridors — dimensions that modular cleanroom designs can be configured around from the start.
Critical Environmental Parameters:
- Temperature: 18-24°C (±1°C tolerance)
- Humidity: 30-60% RH (±5% tolerance)
- Pressure: Minimum 10-15 Pascals differential between adjacent rooms
Prefabricated modular cleanroom systems — like those ACH Engineering designs for medical manufacturers — are particularly well-suited for automation integration. Because the structural layout is configurable before installation, teams can plan equipment placement, power routing, and service access without retrofitting an existing space. Installation typically takes weeks rather than months, and full ISO Class 1-9 compliance is maintained throughout.
Technical and Regulatory Readiness
Establish a quality management system (QMS) compliant with ISO 13485, with documented manufacturing processes defining critical parameters. Your regulatory strategy must address validation requirements for automated systems under FDA 21 CFR Part 11 (electronic records) and applicable device-specific regulations. Canadian manufacturers must also address Health Canada's Medical Device Regulations (SOR/98-282) and software validation guidance under GUI-0029.
Automated systems operating under Part 11 must demonstrate controls across three areas:
- Access limitations: Restricting system use to authorized personnel only
- Operational checks: Enforcing correct sequencing of process steps
- Authority checks: Verifying user permissions before executing critical functions
Every action in these categories requires a secure, time-stamped audit trail.
Organizational and Financial Capability
Required capabilities include:
- Qualified personnel with expertise in automation, cleanroom operations, and validation
- Committed leadership support for the implementation timeline
- Realistic budget covering equipment costs (40-60% of total), integration (25-40%), validation (15-25%), training, and ongoing maintenance
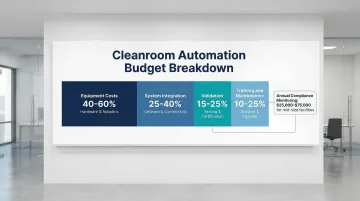
Annual compliance costs for monitoring and calibration range from $25,000 to $75,000 for mid-size facilities.
Key Parameters That Affect Automation Success in Cleanrooms
Get any one of these parameters wrong, and automated systems that should improve consistency can instead introduce new failure points — triggering regulatory findings, product rejects, or process shutdowns.
Environmental Control Consistency
Maintaining stable environmental conditions is critical because automated systems may generate heat or disrupt airflow patterns. Deviations affect both equipment performance and product quality.
Impact on Precision:
- Temperature fluctuations cause thermal expansion in robot linkages, affecting positioning accuracy
- Humidity variations alter fluid viscosity in automated dispensing systems
- Low humidity increases electrostatic discharge risk, damaging sensitive components
Typical tolerances for medical device cleanrooms maintain temperature at 18-24°C (±1°C) and humidity at 30-60% RH (±5%). Automated dispensing and robotic positioning require these tight controls for consistent results.
Contamination Control and Material Compatibility
Particle generation from moving automation components directly impacts contamination risk and the ability to maintain ISO classification during automated operations.
Contamination Sources:
- Belts, cables, and hoses shedding particles
- End-effector movements generating dust
- Lubricants outgassing or degrading
ISO 14644-14:2016 provides methodology for assessing equipment suitability based on particle emission. Leading manufacturers like ABB offer collaborative robots certified to ISO Class 4 particle emission limits.
Material Selection:
- Use PFPE lubricants for low outgassing and chemical stability
- Select 316L stainless steel or anodized aluminum for construction
- Choose PEEK for vacuum chucks to prevent residue transfer
- Ensure all materials withstand required cleaning chemicals
Process Validation and Control Strategy
Validation planning for automated cleanroom processes requires defining and actively monitoring several control layers:
- Identify critical process parameters (CPPs) and critical quality attributes (CQAs) before commissioning
- Establish real-time monitoring and feedback control at each automated step
- Implement statistical process control (SPC) to catch process drift early
- Set alert limits that trigger review before non-conforming product reaches the line
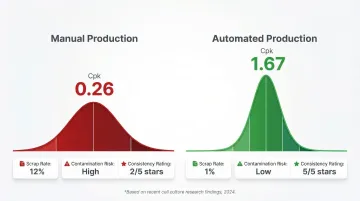
Research comparing manual vs. automated cell culture production found manual processes had poor capability (Cpk = 0.26) compared to automated processes showing significantly less variability and higher consistency.
System Integration and Data Integrity
Connecting automation controllers, environmental monitoring systems, MES, and quality management databases ensures:
- Complete traceability for regulatory compliance
- FDA 21 CFR Part 11 compliance for electronic records
- Data-driven decision making for continuous improvement
Integration Success Stories: A global manufacturer integrated 42Q MES with fully automated lines to produce 10 million devices annually, supporting ISO 13485 compliance. Terumo Americas implemented Siemens Opcenter to create a paperless environment, reducing non-conformances by 40-60%.
Common Mistakes When Implementing Cleanroom Automation in Medical Manufacturing
Three implementation errors account for the majority of failed cleanroom automation projects in medical manufacturing. Each one is preventable with early planning.
Underestimating Validation Complexity
Inadequate IQ/OQ/PQ protocols and validation studies without statistical rigor delay production starts and create regulatory exposure. Budget validation at 15–25% of equipment costs, and add 6–12 months to your timeline if validation planning starts late.
Selecting Incompatible Equipment
Automation equipment selected without a cleanroom compatibility assessment can generate excess particulates, corrode under cleaning chemicals, or disrupt environmental controls. A 2024 FDA Warning Letter to Integra LifeSciences cited particulate excursions near equipment and inadequate investigation of environmental failures — a direct consequence of skipping this step.
Excluding Quality and Regulatory Teams
Bringing in quality assurance and regulatory affairs after design decisions are made leads to GMP gaps and documentation deficiencies that surface during inspections. These teams need a seat at the table from the initial assessment phase, not after the equipment specs are finalized.
Troubleshooting Issues During Cleanroom Automation Implementation
Even well-planned automation projects encounter challenges during commissioning and early operation that need methodical diagnosis.
Problem: Automated System Causing Cleanroom Classification Failure
Likely Causes:
- Excessive particle generation from moving components
- Inadequate ventilation for heat dissipation
- Improper equipment placement disrupting airflow patterns
Solutions:
- Run particle count testing during active equipment operation and review airflow visualization studies
- Modify HVAC capacity or add equipment enclosures to restore ISO classification
- Upgrade to cleanroom-certified robots tested to ISO 14644-14 standards if particle generation persists
Problem: Inconsistent Automated Process Results Despite Environmental Control
Likely Causes:
- Inadequate equipment calibration or worn components
- Software programming errors
- Material variability not accounted for in automation parameters
Solutions:
- Increase calibration frequency and conduct measurement system analysis (MSA) to isolate drift sources
- Review and refine automation recipes using process capability studies
- Add incoming material inspection protocols to reduce variability before it reaches the line
Problem: Integration Failures Between Automation and Facility Systems
Likely Causes:
- Incompatible communication protocols
- Network security restrictions preventing data flow
- Inadequate IT infrastructure for data volume
Solutions:
- Engage automation vendors and IT teams jointly to map and resolve network architecture gaps
- Apply cybersecurity controls (such as role-based access and segmented VLANs) that protect without throttling data flow
- Size data storage and backup capacity to match continuous output volumes before go-live
Alternatives to Full Cleanroom Automation in Medical Manufacturing
Full automation isn't the right fit for every operation. Depending on production volume, product complexity, and budget, three scaled alternatives—semi-automated systems, collaborative robots, and modular phased rollouts—can deliver meaningful gains without the capital commitment of a full deployment.
Semi-Automated Systems with Manual Loading/Unloading
Best For:
- Production volumes that don't justify fully automated material handling
- Product variability requiring human inspection and decision-making
- Limited capital budgets
Trade-offs: Maintains some labor costs and contamination risk from human intervention while gaining consistency in critical process steps. Initial investment runs 30–50% less than full automation.
Collaborative Robots (Cobots) in Cleanroom Environments
For operations that need flexibility without sacrificing cleanliness, cobots offer a practical middle ground.
Ideal For:
- Low-to-medium volume operations where processes shift frequently
- Space-constrained facilities where full industrial robot guarding isn't feasible
Cobots typically deliver ROI under 18 months with a smaller footprint than traditional industrial robots. Manufacturers like KUKA offer cleanroom-rated cobots certified to ISO Class 2 standards.
Trade-offs: Cycle times are slower than dedicated automation, and an operator must remain present—but deployment is faster and costs are significantly lower than industrial robots requiring full guarding.
Modular Automation Approach with Phased Implementation
Most Appropriate When:
- Starting with high-impact processes while building internal expertise
- Spreading capital across multiple fiscal periods
- Reducing risk through incremental implementation takes priority
Success Example: Multiply Labs implemented modular robotic clusters for cell therapy manufacturing, achieving a 74% cost reduction and 100x more patient doses per square foot compared to manual processes.
Trade-offs: The timeline to realize full benefits is longer, and integration between phases can introduce complexity. The upside: reduced financial exposure and the opportunity to optimize each stage before scaling further.

Conclusion
Cleanroom automation for medical manufacturing delivers significant benefits in consistency, compliance, and efficiency when implemented with proper planning, appropriate infrastructure, and rigorous validation. Successful automation requires matching technology to specific process needs, ensuring cleanroom design supports automation integration, and maintaining focus on regulatory compliance throughout implementation.
Balancing those benefits against implementation complexity and organizational readiness is what separates successful programs from stalled ones. The performance case is compelling:
- 40-60% reduction in labour costs
- 2-3x throughput improvements
- Scrap rates dropping from 3-5% to below 0.5%
- 14-20 month typical payback periods
- Up to 80% of contamination events eliminated by removing human operators from critical zones
For medical device manufacturers, these aren't incremental gains — they directly affect product safety, audit outcomes, and long-term competitiveness. Getting the cleanroom infrastructure right from the start is what makes that level of performance achievable.
Frequently Asked Questions
What is cleanroom automation?
Cleanroom automation is the integration of robotic systems, automated material handling, and computerized process control within environmentally controlled manufacturing spaces that meet ISO 14644 standards. It's designed to reduce human intervention and contamination risk while improving consistency in medical device production.
What are the cleanroom requirements for medical devices?
Requirements depend on device risk classification and applicable standards — including ISO 14644 and North American regulations such as FDA 21 CFR 211.42. Non-sterile device manufacturing typically requires ISO Class 7 or 8; sterile production typically requires ISO Class 5–7, with defined particle counts, environmental monitoring, and documentation protocols.
What is a cleanroom in healthcare?
Healthcare cleanrooms are controlled environments used for manufacturing medical devices, compounding pharmaceuticals, and performing sterile procedures. Air filtration, pressure control, and contamination protocols maintain specific cleanliness levels to protect patient safety and product quality.
How long does it take to implement cleanroom automation for medical manufacturing?
Most projects run 12–36 months depending on scope, broken down roughly as:
- Planning and design: 3–6 months
- Equipment procurement and installation: 6–12 months
- Validation and regulatory approval: 6–12 months
Phased implementations can deliver partial operational benefits earlier in the timeline.
What is the typical ROI for cleanroom automation in medical device manufacturing?
ROI typically falls between 14–20 months, driven by production volume and labor costs. Key gains include 30–50% less contamination-related scrap, 200–300% throughput improvement, 40–60% lower long-term labor costs, and reduced recall risk through stronger regulatory compliance.
Can existing cleanrooms be retrofitted with automation systems?
Many existing cleanrooms can be retrofitted with automation if structural capacity, ceiling height, and utility infrastructure are adequate. Modular cleanroom designs offer advantages in accommodating automation changes. Retrofits require thorough validation to confirm environmental controls stay compliant throughout and after installation.


