
Introduction: Understanding ISO Class 8 Cleanroom Requirements
ISO Class 8 cleanrooms sit at a specific intersection: strict enough to protect non-sterile pharmaceutical manufacturing and electronics assembly, yet flexible enough to support packaging operations and support zones adjacent to cleaner classifications. For manufacturers navigating contamination control without the overhead of ISO 5 or 6 requirements, Class 8 is often the practical starting point.
According to ISO 14644-1:2015, ISO Class 8 cleanrooms permit a maximum of 3,520,000 particles ≥0.5 microns per cubic meter of air — the most lenient threshold still considered "clean" under regulated manufacturing standards. That said, achieving and maintaining this classification still demands precise engineering: HEPA filtration, defined air change rates, and structured testing protocols are all required.
This guide covers what you need to know about ISO Class 8 cleanroom requirements — from design specifications and compliance standards to testing protocols and practical implementation across pharmaceutical, biotech, medical device, and electronics industries.
TLDR: Key Takeaways
- ISO Class 8 permits maximum 3,520,000 particles ≥0.5μm/m³ (equivalent to former Class 100,000)
- Requires 99.97% efficient HEPA filtration with non-unidirectional airflow
- Minimum 15–25 air changes per hour to maintain particle count compliance
- Used extensively in pharmaceutical packaging, medical device assembly, electronics manufacturing, and biotech support zones
- Annual re-qualification mandatory per ISO 14644-2, covering particle counts, pressure differentials, and airflow
- Modular construction reduces installation time and costs while maintaining full regulatory compliance
What is ISO Class 8 Cleanroom Classification?
ISO Class 8 cleanrooms are controlled environments where airborne particle concentration is monitored and maintained within specific limits according to ISO 14644-1:2015—the global standard for air cleanliness. These facilities represent the baseline classification for regulated manufacturing environments where particle contamination must be controlled but ultra-clean conditions aren't required.
Particle Count Limits
ISO Class 8 cleanrooms must maintain particle concentrations at or below these maximum levels:
| Particle Size | Maximum Concentration |
|---|---|
| ≥ 0.5 microns | 3,520,000 particles/m³ |
| ≥ 1.0 microns | 832,000 particles/m³ |
| ≥ 5.0 microns | 29,300 particles/m³ |
The standard focuses on particles 0.5 microns or larger because smaller particle concentrations in ISO Class 8 environments are too high to count accurately. Attempting to measure ultrafine particles would create coincidence errors in particle counters and yield unreliable data.
Relationship to Former Standards
ISO Class 8 directly corresponds to the former US Federal Standard 209E Class 100,000 designation. Although Federal Standard 209E was officially withdrawn in 2001, many industry professionals still reference "Class 100,000" when discussing these environments. If your facility documentation or supplier specifications still use 209E language, verify the equivalency mapping before submitting to a regulatory body—terminology mismatches can flag compliance reviews.
Cleanroom Classification Hierarchy
Understanding where ISO Class 8 fits within the broader cleanroom hierarchy helps contextualize its requirements:
- ISO Class 5: 3,520 particles/m³ (≥0.5μm) — used for aseptic processing; roughly 1,000× cleaner than Class 8
- ISO Class 6: 35,200 particles/m³ — common in sterile compounding, 100× cleaner than Class 8
- ISO Class 7: 352,000 particles/m³ — standard for pharmaceutical manufacturing support areas; about 10× cleaner than Class 8
- ISO Class 8: 3,520,000 particles/m³ — baseline for controlled manufacturing, packaging, and non-sterile operations

For facilities operating across multiple cleanliness zones—such as a pharmaceutical plant with both aseptic fill lines and outer packaging areas—ISO Class 8 typically governs the lower-risk zones while stricter classes apply closer to the product.
Core Design Specifications & Requirements for ISO Class 8
Filtration & Particle Control Systems
HEPA filtration is the primary particle control mechanism in ISO Class 8 cleanrooms. Filters must achieve 99.97% efficiency at 0.3 microns (the most penetrating particle size, or MPPS). Terminal HEPA modules or Fan Filter Units (FFUs) deliver filtered air at entry points, with FFUs offering modular flexibility for retrofit applications and spot-treatment of critical zones.
Leak testing procedures follow IEST-RP-CC034 and ISO 14644-3 standards, using aerosol challenges (typically PAO or DOP) to verify filter integrity after installation and periodically throughout the filter's service life.
Airflow Patterns & Air Change Rates
Unlike cleaner classifications that require unidirectional laminar flow, ISO Class 8 cleanrooms typically employ non-unidirectional (turbulent) airflow. This approach relies on dilution rather than displacement to reduce particle concentration. The following specifications define how that dilution is achieved:
- Air changes per hour (ACH): 15-25 ACH minimum, though some applications may require up to 48 ACH depending on process bio-burden
- Ceiling coverage: 5-15% of ceiling area should contain filtration units
- Airflow pattern: Vertical flow from ceiling-mounted HEPA filters to low-wall return air grilles
- Return air placement: Low-wall returns promote top-down airflow that sweeps particles away from work surfaces at 30–36 inches above finished floor, toward floor-level returns
This design prevents re-entrainment of settled particles and maintains cleanliness at critical work heights.
Architectural Finishes & Materials
All surfaces must be non-shedding, easily cleanable, and resistant to chemical sanitization. Material selection varies by application: biotech facilities typically require chemical-resistant coatings, while nanotechnology applications demand low-outgassing materials to prevent molecular contamination.
Wall systems include modular purpose-built panels or epoxy-coated gypsum with smooth, crevice-free surfaces.
Ceiling systems fall into two categories:
- Walkable/panelized systems: Used in pharmaceutical applications requiring frequent maintenance access above the ceiling plane
- Gasketed grid systems: Common in non-sterile applications; tiles seal to grids preventing air bypass
Flooring must meet several overlapping requirements depending on the application:
- Seamless resinous floor systems (epoxy or urethane) or heat-sealed vinyl
- Integral coving where floors meet walls (typically 4-6 inches radius)
- Chemical resistance appropriate to cleaning agents and process materials
- Static control properties (ESD flooring) for electronics applications
- Smooth, non-porous finish preventing microbial growth and particle entrapment
Environmental Controls
Pressure differentials of 10-15 Pascals (0.04-0.06 inches w.g.) must be maintained between ISO Class 8 zones and adjacent unclassified or lower-classification areas. Pressure should cascade from cleanest to dirtiest zones, for example: ISO 7 > ISO 8 > Unclassified. This positive pressure gradient prevents contamination ingress when doors open or air leaks occur.
Key environmental parameters include:
- Temperature: 20-22°C (68-72°F); tighter ranges may apply for specific processes
- Humidity: 30-60% RH — below 30% risks static buildup, above 60% encourages microbial growth and corrosion
- Lighting: 300-500 lux for adequate task visibility
- Noise criteria: NC-50 to NC-55, varying by equipment density
- Static control: Critical for electronics manufacturing applications
- Material outgassing: Low-VOC materials per ISO 14644-8 for sensitive processes

Particle Count Standards & Testing Requirements
ISO 14644-1:2015 Classification Standards
The 2015 revision of ISO 14644-1 introduced two key updates to sampling methodology. The number of required sampling locations is now determined by a lookup table rather than the square root of area calculation used previously. For cleanrooms exceeding 1,000 m², the formula is: NL = 27 × (Area/1000).
The 2015 revision also eliminated the 95% Upper Confidence Limit (UCL) calculation for sample sizes of 2-9 locations. Compliance now requires each individual sampling location to meet the class limit independently—a more stringent requirement that catches localized contamination issues.
Mandatory Testing Protocols
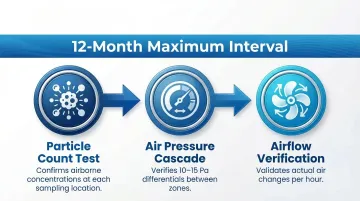
ISO 14644-2:2015 establishes a risk-based monitoring strategy with maximum re-qualification intervals. For ISO Class 8 cleanrooms, three tests are mandatory at 12-month maximum intervals:
- Particle Count Test: Confirms airborne concentrations at each sampling location stay within classification limits
- Air Pressure Cascade: Verifies pressure differentials between zones meet design specifications (typically 10–15 Pa)
- Airflow Verification: Measures air velocity or volume flow to validate actual air changes per hour
Particle counter requirements per ISO 21501-4 include:
- Calibration traceable to national standards
- 50% ± 20% counting efficiency at threshold particle size
- Resolution sufficient to distinguish particle size channels
- Annual calibration verification
Where continuous monitoring systems are in place, re-qualification intervals beyond 12 months may be justified — provided accumulated data demonstrates consistent environmental control.
Industries & Applications for ISO Class 8 Cleanrooms
ISO Class 8 cleanrooms serve multiple industries requiring contamination control without the stringency and cost of ultra-clean environments.
Primary industries and applications:
Pharmaceutical/Nutraceutical Manufacturing
- Secondary packaging operations
- Material airlocks and transfer areas
- Gowning rooms (Grade D equivalent)
- Non-sterile compounding
Medical Device Manufacturing
- Surgical kit assembly
- Packaging of terminally sterilized devices
- Non-implantable device assembly
- Component molding operations
Biotechnology
- Buffer preparation zones
- Equipment staging areas
- Support laboratories
- Media preparation rooms
Electronics & Semiconductor
- Printed circuit board (PCB) assembly
- Lithium-ion battery production
- Solar panel manufacturing
- General electronics assembly
Additional Industries
- Certain packaged food production
- Cosmetics manufacturing
- Aerospace component handling
- Research and development laboratories

ISO Class 8 sits one step above ISO Class 7 in the classification hierarchy — clean enough for controlled product contact, but without the HEPA saturation and pressurization demands of higher-tier environments. For many operations, that distinction directly determines project budget and timeline.
Gowning Requirements & Operational Protocols
Personnel Gowning Standards
Personnel represent the primary contamination source in any cleanroom. ISO Class 8 gowning requirements include:
- Head covers: Bouffant caps covering all hair
- Goggles or safety glasses: When required by process risk assessment
- Beard covers: For personnel with facial hair
- Lab coats or frocks: Made from non-shedding synthetic materials (typically polyester)
- Foot coverings: Disposable booties or dedicated cleanroom shoes
- Gloves: Task-appropriate (vinyl, latex, or nitrile) based on chemical compatibility and allergen concerns
Gowning rooms should be separate airlocks that maintain pressure between the cleanroom and unclassified areas, preventing contamination transfer during the gowning process. They typically hold ISO Class 8 or unclassified status.
Material Controls & Housekeeping
Controlling what enters the cleanroom is just as important as what personnel wear. Prohibited materials beyond airlocks include:
- Cardboard boxes and shipping containers
- Wood pallets or crates
- Standard paper (cleanroom paper only)
- Dry erase boards
- Pencils or other particle-generating writing instruments
These materials shed excessive particles and fibers that overwhelm filtration systems.
Cleaning Protocols (per IEST-RP-CC018)
- Daily cleaning of floors and work surfaces using cleanroom-approved mops and wipes
- Weekly cleaning of walls and ceilings
- HEPA-filtered vacuum systems only (never conventional vacuums)
- Cleanroom-compatible disinfectants validated for efficacy
- Written Standard Operating Procedures (SOPs) for all cleaning activities
- Documentation of cleaning completion and any deviations
All cleaning activities must be documented — SOPs without records provide no audit trail during regulatory inspections.
Achieving & Maintaining ISO Class 8 Compliance
Initial Cleanroom Design & Construction
Successful ISO Class 8 implementation begins with thorough planning. Key considerations include:
- Process requirements analysis: Understanding particle sensitivity, temperature/humidity needs, and material flow patterns
- Space optimization: Efficient layout minimizing unnecessary square footage while accommodating equipment and personnel movement
- Scalability planning: Designing infrastructure to accommodate future expansion without major reconstruction
ACH Engineering handles this process as a turnkey cleanroom engagement—covering design, engineering, and construction under one project team. Their modular build approach typically cuts installation time compared to conventional construction, which matters when a facility needs to reach production quickly.
Validation & Certification Process
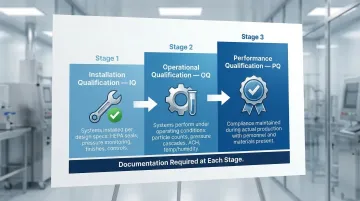
Three-stage validation confirms the cleanroom meets specifications:
Installation Qualification (IQ) verifies that all systems are installed according to design specifications:
- HEPA filters properly sealed and leak-free
- Pressure monitoring systems functional and calibrated
- Architectural finishes meet material specifications
- Lighting, temperature, and humidity controls operational
Operational Qualification (OQ) demonstrates systems perform as intended under operating conditions:
- Particle counts meet ISO Class 8 limits at all sampling locations
- Pressure differentials maintain specified cascades
- Air change rates achieve design targets
- Temperature and humidity remain within acceptable ranges
Performance Qualification (PQ) proves the cleanroom maintains compliance during actual production operations with personnel, equipment, and materials present.
Each stage generates documentation—protocols, test results, deviation records, and summary reports. Regulators expect this paper trail during audits, so maintaining organized records from IQ through PQ is as important as the testing itself.
Ongoing Monitoring & Maintenance
Continuous environmental monitoring systems track:
- Particle counts at critical locations
- Pressure differentials between zones
- Temperature and humidity
- Airflow rates
Preventive maintenance schedules address:
- HEPA filter integrity testing (annually minimum)
- Filter replacement when pressure drop exceeds specifications (typically double initial resistance)
- HVAC system inspection and calibration
- Particle counter calibration (annually)
- Pressure gauge verification
Periodic re-qualification occurs at 12-month maximum intervals per ISO 14644-2, though continuous monitoring programs may justify extended intervals with regulatory approval.
Common Pitfalls & How to Avoid Them
Frequent compliance issues include:
Insufficient sampling locations: Using outdated calculation methods rather than ISO 14644-1:2015 lookup tables results in inadequate spatial coverage, missing localized contamination.
Improper particle counter calibration: Expired calibrations or non-compliant instruments yield unreliable data that regulatory agencies will reject during audits.
Inadequate personnel training: Staff unaware of proper gowning procedures, material transfer protocols, or cleaning requirements inadvertently introduce contamination.
Poor material transfer protocols: Bringing prohibited materials like cardboard into cleanrooms or failing to wipe down items during transfer compromises cleanliness.
Pressure cascade failures: Inadequate monitoring allows pressure reversals where contaminated air flows into clean zones rather than being excluded.
A pre-audit gap assessment is one of the most practical steps a facility can take. ACH Engineering's cleanroom consulting services conduct these reviews to surface compliance issues—such as sampling plan errors or pressure cascade weaknesses—before a regulator does.
Frequently Asked Questions
What are the requirements for ISO Class 8 cleanroom?
ISO Class 8 cleanrooms must maintain maximum 3,520,000 particles ≥0.5μm/m³. Requirements include 99.97% efficient HEPA filtration, 15-25 air changes per hour, non-unidirectional airflow, 10-15 Pa positive pressure versus adjacent areas, appropriate personnel gowning, and annual testing per ISO 14644-2.
How does ISO Class 8 differ from ISO Class 5 and ISO Class 7 cleanrooms?
ISO Class 5 permits 3,520 particles ≥0.5μm/m³ — 1,000× cleaner than Class 8 — and requires 240+ ACH with unidirectional laminar flow. ISO Class 7 permits 352,000 particles with 60 ACH for sterile manufacturing. Class 8 is the least stringent tier, suited for packaging, compounding support, and material transfer zones.
What is ISO 8 used for?
ISO Class 8 serves pharmaceutical packaging and compounding, medical device assembly and packaging, electronics manufacturing, biotechnology support areas, material airlocks, gowning rooms, and other controlled manufacturing operations requiring contamination control but not ultra-clean conditions.
How often must ISO Class 8 cleanrooms be re-certified?
ISO 14644-2 requires re-certification at maximum 12-month intervals, including particle count testing, pressure differential verification, and airflow measurement. Continuous monitoring programs may justify extended intervals with documented evidence of sustained compliance.
Can existing facilities be upgraded to ISO Class 8?
Yes, existing spaces can be retrofitted using modular wall systems and fan filter unit (FFU) installations. Successful upgrades require assessing existing HVAC capacity, structural support for ceiling-mounted equipment, and available space for airlocks.
What are typical costs for ISO Class 8 cleanroom construction?
Costs depend on size, design complexity, and industry requirements. Modular construction typically runs 30-40% less than traditional stick-built cleanrooms and compresses installation from months to weeks, reducing both project costs and operational disruption.


