
Introduction
In 2023, the FDA issued a warning letter to a pharmaceutical manufacturer after discovering deleted humidity data from their stability chambers—data that could have revealed excursions reaching 94.8% RH over consecutive days. The investigation uncovered that high humidity had compromised capsule integrity and created conditions ripe for microbial contamination, jeopardizing an entire product line.
This isn't an isolated incident. Across industries—from semiconductor fabs losing wafer batches to ESD events to biotech labs facing contamination shutdowns—humidity failures are a critical, often underestimated variable in cleanroom performance. Poor humidity control doesn't just affect product quality; it triggers regulatory violations, drives up operational costs, and can halt production entirely.
With ESD damage alone costing the electronics industry over $10 billion annually, and humidity excursions ranking among the top FDA and Health Canada citations for pharmaceutical manufacturers, uncontrolled moisture is one of the costliest risks a cleanroom facility faces. This guide covers the causes, consequences, and practical solutions—from dehumidification systems to environmental monitoring—that help facilities maintain compliant, stable humidity year-round.
TLDR: Key Takeaways
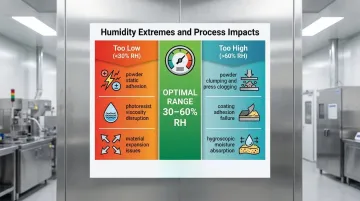
- Cleanroom humidity typically ranges from 30–60% RH, with exact targets driven by process sensitivity and contamination risks
- Three control methods dominate: steam humidification for precision, desiccant dehumidification for ultra-low moisture, and hybrid systems for flexibility
- Poor control triggers electrostatic discharge (ESD) below 30% RH and microbial growth above 60% RH
- Industry-specific regulations (FDA 21 CFR Part 211, SEMI standards) dictate actual humidity targets; ISO 14644-4 provides the underlying design framework
- Hybrid systems can reduce cooling costs by 25–45% and cut reactivation energy by 35–50% compared to conventional single-method approaches
Why Humidity Control Matters in Cleanrooms
Humidity is one of the most consequential environmental variables in a cleanroom. Too low, and static charges accumulate fast enough to destroy sensitive components. Too high, and microbial contamination becomes almost inevitable. Either way, the consequences reach directly into yield, compliance, and cost.
Electrostatic Discharge (ESD) Risks
When relative humidity drops below 30%, surfaces lose the thin moisture layer that naturally dissipates static charges. Materials become insulators, allowing voltages to accumulate rapidly—often exceeding 100V—capable of destroying sensitive components in microseconds.
The semiconductor industry bears the brunt of this risk. Over one-third of field failures in semiconductor manufacturing stem from ESD events, with annual costs exceeding $10 billion globally.
Component yield losses from ESD damage range between 16–22% in manufacturing environments. A single discharge during photolithography can scrap entire wafer lots, reducing yields by 5–20% and triggering costly rework cycles.
Pharmaceutical manufacturing faces similar challenges. Powder handling operations below 30% RH generate static charges that cause materials to cling to equipment or become airborne, compromising batch uniformity and contamination control.
Microbial Contamination Concerns
High humidity creates the opposite problem. Above 60% RH, moisture availability enables bacterial growth, mold proliferation, and spore germination—all catastrophic in sterile manufacturing environments.
A 21-year review of FDA enforcement actions identified environmental molds (Aspergillus penicillioides) as an emerging contamination vector in sterile drug manufacturing, linked repeatedly to moisture control failures. In 2023, Iso-Tex Diagnostics received an FDA warning letter after humidity in their sterile processing cleanroom repeatedly spiked to 94.8% RH over three consecutive days. Investigators cited the firm's use of a portable dehumidifier in the gowning room as evidence of a "poorly designed HVAC system" unable to meet basic environmental requirements.
The microbial risk threshold is straightforward:
- Below 60% RH: Growth and germination are very unlikely
- Above 60% RH: Contamination risk increases sharply, particularly in pharmaceutical compounding and biotech environments
Product Quality and Process Integrity
Beyond contamination and ESD, humidity directly affects material properties and process outcomes:
- Causes powder clumping and press clogging at high humidity; low humidity drives static-related adhesion in tablet compression
- Disrupts coating adhesion and drying rates during tablet coating, producing surface defects
- Affects photoresist viscosity and adhesion during photolithography, with fluctuations causing dimensional variations in circuit patterns
- Causes hygroscopic materials to absorb ambient moisture in powder handling, altering flow characteristics and dosing accuracy
- Drives expansion and contraction in materials as humidity shifts, compromising precision assembly and tolerances
Humidity Control Methods and Technologies
Three primary approaches dominate cleanroom humidity control, each optimized for specific operating ranges and process requirements.
Steam Humidification Systems
Steam-based systems generate moisture through boiling, delivering sterile, mineral-free vapor into the airstream. They excel in applications requiring moderate humidity targets (40–55% RH) with tight control tolerances.
Resistive Electric Steam Humidifiers:
- Use resistive heating elements to boil RO/DI water
- Capacity range: 2.6–147 kg/h per unit (linkable to 1,176 kg/h)
- Control accuracy: ±1% RH
- Minimal maintenance (no mineral buildup with proper water treatment)
- Best for high-precision labs and pharmaceutical facilities
Electrode Boiler Systems:
- Generate steam by passing current through conductive water
- Capacity range: 2–91 kg/h
- Control accuracy: ±5% RH
- Lower capital cost but higher maintenance (cylinder replacement required)
- Not suitable for RO/DI water (requires mineral content for conductivity)
Advantages: Precise control, sterility (boiling kills microorganisms), compatibility with cleanroom-grade water sources, and predictable performance across varying loads.
Desiccant Dehumidification Systems
Desiccant systems absorb moisture from air using hygroscopic materials (typically silica gel or molecular sieves) in a rotating wheel. As one section of the wheel absorbs moisture, another section undergoes regeneration (heating to release captured water vapor).
Performance specifications:
- Capable of dew points as low as -40°F to -70°F (-40°C to -56°C)
- Control accuracy: ±1% RH in high-performance units
- Effective below 30% RH where mechanical dehumidifiers struggle
- Independent of cooling requirements (works at any temperature)
Applications: Essential for semiconductor fabs, lithium battery dry rooms (often requiring <1% RH), and any process involving hygroscopic powders or moisture-sensitive materials.
The main trade-off is higher operating cost from regeneration energy. That said, optimized purge cycles in newer units deliver 30-45% energy savings compared to older designs. Rotors are typically bacteriostatic and silicone-free to prevent contamination.
Hybrid and Integrated Approaches
Neither steam nor desiccant systems alone cover the full range of cleanroom requirements. Combining both provides optimal flexibility — allowing systems to maintain precise targets across wide operating ranges while minimizing energy waste.
Hybrid system benefits:
- Desiccant handles latent load removal (bulk dehumidification)
- Steam provides precise trim control for fine adjustments
- Prevents "fighting systems" (simultaneous humidification and dehumidification)
- Reduces cooling costs by 25-45% and reactivation energy by 35-50% in validated case studies
HVAC integration: Modern cleanroom HVAC systems sequence chillers, heaters, humidifiers, dehumidifiers, and filtration in coordinated loops. Building Management Systems (BMS) use real-time sensor data to make micro-adjustments, maintaining stability within ±1-3% RH.
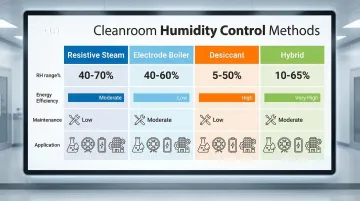
Comparison Table:
| Method | Typical RH Range | Energy Efficiency | Maintenance Needs | Best Applications |
|---|---|---|---|---|
| Resistive Steam | 40-65% RH | Moderate | Low (with RO/DI water) | Pharma, biotech, precision labs |
| Electrode Boiler | 40-65% RH | Moderate | Moderate (cylinder replacement) | General cleanrooms, lower budgets |
| Desiccant | <1% to 50% RH | Lower (high regeneration energy) | Moderate (wheel cleaning) | Semiconductor, battery, hygroscopic materials |
| Hybrid | 1-65% RH | High (optimized load distribution) | Moderate | Multi-process facilities, variable loads |
Industry-Specific Humidity Requirements
Pharmaceutical and Biotech Cleanrooms
Typical range: 35-65% RH, depending on process stage and product sensitivity
Regulatory drivers:
- FDA 21 CFR 211.42(c)(10)(ii): Explicitly requires "temperature and humidity controls" for aseptic processing areas
- FDA 21 CFR 211.46(b): Mandates equipment for adequate humidity control to prevent contamination
- USP <797>: Recommends maintaining relative humidity below 60% to minimize microbial proliferation
Critical processes affected:
- Sterile compounding: Typically targets 45-55% RH to balance microbial control with material stability
- Powder handling: Requires 35-50% RH to prevent static buildup and moisture absorption
- Tablet coating: Needs 40-55% RH for optimal coating adhesion and drying
Semiconductor and Electronics Manufacturing
Typical range: 30-50% RH
Primary concern: ESD protection. The 40-60% RH range is optimal for static dissipation, but most fabs run drier — between 30-50% — to prevent corrosion on metal surfaces and wafers. Ionization systems are typically added to manage the resulting ESD risk.
Critical processes:
- Photolithography: Requires extremely tight control (±1% RH or tighter); photoresist behavior is highly humidity-sensitive
- Wafer handling: Low humidity prevents condensation but increases ESD risk
- Component assembly: Balances ESD protection with moisture-sensitive materials
Standards: SEMI E78 and E129 address electrostatic compatibility, implicitly driving humidity requirements to mitigate charge generation.
Laboratory and Research Facilities
Typical range: 40-60% RH
Drivers:
- ASHRAE guidelines: Recommend 40-60% RH to optimize human respiratory immune function and minimize bacterial/viral survival rates
- Stability testing (ICH Q1A): Requires precise control for long-term studies, typically 25°C/60% RH or 30°C/65% RH
Variability: Research labs often require multiple environmental zones for different testing protocols, demanding flexible HVAC systems capable of maintaining distinct humidity targets in adjacent spaces. This zoning complexity is often one of the first challenges to surface during cleanroom design — and one of the most important to plan for early.
System Design and Integration Considerations
Humidity control needs to be built into the HVAC system from the start. Integration decisions, sensor placement, and control algorithms determine whether a system holds tight tolerances — or constantly overcorrects.
HVAC System Components and Integration
Air handling units (AHUs) integrate multiple subsystems in sequence:
- Filtration: Pre-filters remove particulates before conditioning
- Cooling coils: Reduce temperature and remove moisture (mechanical dehumidification)
- Heating coils: Reheat air to target temperature
- Humidifiers: Add moisture as needed (steam or adiabatic)
- Desiccant wheels: Remove moisture for ultra-low humidity applications
- HEPA filtration: Final filtration before distribution

Sequencing is critical. Poorly designed systems simultaneously humidify and dehumidify, wasting energy and creating instability. Proper sequencing uses "dead bands" where neither system operates, allowing natural drift within acceptable tolerances before corrective action.
Sensor Networks and Control Systems
Distributed humidity sensors provide real-time data to BMS, enabling micro-adjustments to maintain tight tolerances.
Sensor placement best practices:
- Mount duct sensors at least 15 feet downstream from humidifiers to ensure complete moisture absorption
- Avoid "dead legs" (low-airflow zones) that don't represent room conditions
- Use 3D mapping (gridding) during validation to identify stratification and justify permanent sensor locations
Control algorithms: Modern systems use PID (Proportional-Integral-Derivative) control loops with feed-forward capabilities to anticipate load changes before they affect conditions. High-precision applications achieve ±1% to ±3% RH control through continuous micro-adjustments.
Calibration: Sensors drift over time due to chemical exposure or contamination. Baseline recommendation is quarterly calibration, with regulated industries often requiring monthly verification. NIST-traceable calibration certificates are mandatory for FDA/ISO compliance audits.
Energy Efficiency Optimization
Optimized humidity control systems can deliver 20-30% energy savings through:
- Heat recovery: Capturing waste heat from desiccant regeneration to reduce cooling loads
- Variable speed drives (VSD): Modulating fan speeds to match actual load rather than running at full capacity
- Economizer cycles: Using outdoor air when conditions permit, reducing mechanical conditioning requirements
- Demand-based control: Adjusting humidity targets based on occupancy and process schedules
Getting these design elements right from the start is where a turnkey cleanroom partner like ACH Engineering adds real value — handling HVAC integration, sensor layout, and post-installation validation under one project framework, rather than leaving coordination gaps between separate contractors.
Compliance Standards and Best Practices
ISO 14644-4 Framework
ISO 14644-4:2022 addresses cleanroom design, construction, and start-up, with specific emphasis on environmental control including humidity. Unlike prescriptive standards that mandate specific numbers, Clause 6.1 explicitly requires that the customer and designer define humidity requirements based on process risks and product sensitivity.
This places the burden of scientific justification on the facility. Simply stating "we maintain 40-50% RH" is insufficient—you must document why that range is necessary, how it protects product quality, and what happens when excursions occur.
Documentation requirements:
- Continuous monitoring logs: Secure, backed-up data showing RH trends over time
- Calibration certificates: NIST-traceable certificates for all sensors, performed by accredited labs
- Validation reports: IQ (Installation Qualification), OQ (Operational Qualification), and PQ (Performance Qualification) documents proving the system meets design specifications under load
- Excursion investigations: Documented root cause analysis for any deviation beyond alert/action limits
Industry-Specific Regulatory Requirements
Pharmaceutical (FDA / Health Canada):
- 21 CFR Part 211.42(c)(10)(ii): Requires temperature and humidity controls for aseptic processing
- 21 CFR Part 211.46(b): Mandates equipment for adequate humidity control to prevent contamination
- Health Canada GUI-0036 / GUI-0119: Canadian GMP guidelines establishing equivalent environmental control expectations for drug manufacturers
Semiconductor:
- SEMI E78/E129: Address electrostatic compatibility, implicitly driving humidity requirements
HVAC Design:
- ASHRAE Standard 170: Specifies humidity ranges for healthcare facilities (e.g., Operating Rooms: 20-60% RH), often serving as a baseline for biological cleanrooms
Key shift in enforcement: Recent FDA warning letters emphasize the adequacy of investigations when excursions occur, not just whether you stayed within range. Failure to investigate humidity deviations—even if product quality is retrospectively deemed unaffected—now triggers primary citations.
Working with an engineering partner like ACH Engineering during design and validation phases ensures your humidity control documentation meets cGMP, FDA, USP 797, ISO, and NAPRA requirements—from initial specification through final commissioning.
Troubleshooting Common Humidity Issues
Three issues account for the majority of cleanroom humidity service calls: humidity drift, uneven distribution, and high energy consumption. Each has predictable causes — and systematic fixes.
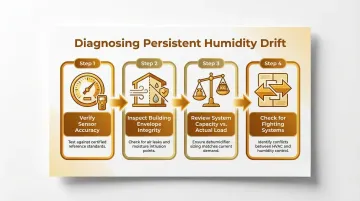
Persistent Humidity Drift
Common causes:
- Sensor calibration errors (capacitive sensors drift due to chemical exposure)
- Air leaks in the building envelope (infiltration/exfiltration)
- Inadequate system capacity for actual load
- Seasonal outdoor air changes overwhelming conditioning capacity
Troubleshooting sequence:
- Verify sensor accuracy with a calibrated reference instrument
- Inspect envelope integrity (doors, pass-throughs, ceiling plenums)
- Review system capacity vs. actual load (has occupancy or equipment changed?)
- Check for simultaneous humidification and dehumidification (fighting systems)
If levels are consistent overall but vary across zones, uneven distribution is the likely culprit — driven by airflow rather than capacity.
Uneven Humidity Distribution
What to check:
- Poor airflow patterns (dead zones, short-circuiting)
- Inadequate mixing (insufficient air changes per hour)
- Stratification (temperature/humidity layering)
How to fix it:
- Conduct airflow modeling — computational fluid dynamics (CFD) analysis — to identify problem zones
- Adjust diffuser locations or add circulation fans
- Increase air changes per hour (ACH) to improve mixing
- Relocate sensors to representative locations (not dead zones)
When humidity control is working but energy bills are climbing, the problem usually lies in control logic or equipment sizing — not the hardware itself.
High Energy Consumption
What to check:
- Simultaneous humidification and dehumidification (poor control sequences)
- Oversized equipment cycling on/off frequently
- Inefficient control algorithms (aggressive setpoints, no dead bands)
How to fix it:
- Optimize control sequences (implement dead bands, adjust PID tuning)
- Right-size equipment based on actual load profiles
- Implement heat recovery from dehumidification processes
- Use variable speed drives to modulate output rather than cycling
Frequently Asked Questions
How to control humidity in a cleanroom?
Cleanrooms use dedicated HVAC systems with integrated humidifiers (steam or cleanroom-grade systems) and dehumidifiers (mechanical chillers or desiccant wheels) controlled by distributed sensor networks. These systems maintain precise humidity levels, typically 30–60% RH depending on the application, using real-time feedback loops and automated adjustments.
Is 70% humidity in a room too high?
Yes, 70% RH is too high for most cleanroom applications. It raises microbial growth risk, condensation potential, and material degradation. Most cleanrooms target 30–60% RH, with pharmaceutical sterile manufacturing often aiming for 45–55% RH to balance microbial control with material stability.
What is the typical humidity range for pharmaceutical cleanrooms?
Pharmaceutical cleanrooms typically maintain 35–65% RH depending on the process. USP <797> specifically recommends staying below 60% RH to control contamination risks, making regulatory compliance the primary driver for humidity targets in these environments.
How often should cleanroom humidity sensors be calibrated?
Quarterly calibration is the baseline, with regulated industries often requiring monthly verification based on sensor drift history and process criticality. All calibrations should be NIST-traceable and documented — continuous monitoring with automated alerts is strongly recommended for audit readiness.
What causes static electricity in cleanrooms?
Low humidity (below 30–35% RH) prevents static charges from dissipating naturally. The thin moisture layer that normally forms on surfaces and conducts charge away evaporates, turning materials into insulators. This allows electrostatic discharge (ESD) to build rapidly, damaging sensitive components and products.
Can I use a standard humidifier in my cleanroom?
No. Standard humidifiers introduce contaminants, minerals (white dust), and microorganisms — including biofilm-derived endotoxins — into the air. Cleanroom-grade units use purified water (RO/DI), stainless steel construction, and steam generation to meet ISO 14644-4 contamination control requirements.


