
Introduction: The Critical Role of Cleanrooms in Modern Healthcare
Hospital-acquired infections (HAIs) affect approximately 1 in 31 hospital patients on any given day, costing healthcare systems across North America billions annually. The COVID-19 pandemic accelerated adoption of cleanroom protocols across wards previously considered low-risk. Yet many healthcare administrators still associate cleanrooms exclusively with operating rooms, overlooking the full range of clinical environments that require controlled conditions.
Modern hospital cleanrooms extend far beyond surgical suites. They encompass USP 797/800 compounding pharmacies, pathology laboratories, burn units, and isolation areas — each with its own contamination risks and regulatory requirements.
Each application demands precise environmental control — from HEPA filtration and pressure differentials to temperature regulation and antimicrobial surfaces. This guide breaks down what effective healthcare cleanroom design looks like across these environments, and what facility administrators need to plan for at every stage.
TLDR: Key Takeaways for Hospital Cleanroom Design
- Hospital cleanrooms typically operate at ISO Class 7–8, with sterile compounding areas requiring ISO Class 5 (USP 797)
- USP 800 mandates negative pressure containment for any space handling hazardous drugs
- Core design elements include HEPA filtration (99.97% efficiency), proper airflow patterns, pressure control (positive for sterile areas, negative for hazardous zones), and 30+ air changes per hour
- Modular construction cuts delivery time to 8–16 weeks (vs. 6–12 months traditional) with 20–30% cost savings and minimal disruption to active facilities
- Material selection focuses on seamless, non-porous surfaces like epoxy-coated walls and heat-welded vinyl flooring that withstand rigorous disinfection protocols
Understanding Cleanroom Classifications for Healthcare Facilities
ISO 14644-1 Classification Standards
ISO 14644-1:2015 defines nine cleanliness classes based on maximum allowable particle concentrations per cubic meter. Healthcare facilities primarily operate in ISO Class 5, 7, and 8 ranges, with stricter classifications reserved for the most critical applications.
Hospital Cleanroom Classifications:
| ISO Class | Max Particles/m³ (≥0.5 µm) | Healthcare Application |
|---|---|---|
| ISO Class 5 | 3,520 | Laminar flow hoods, critical surgical fields |
| ISO Class 7 | 352,000 | Sterile compounding buffer rooms, hazardous drug areas |
| ISO Class 8 | 3,520,000 | Ante-rooms for positive pressure buffer rooms |

Most hospital applications require ISO Class 7 or 8 — these classifications meet HVAC feasibility constraints while satisfying regulatory minimums. ISO Class 5 is reserved for primary engineering controls (PECs) like laminar airflow hoods where direct drug preparation occurs, and for surgical fields during critical procedures.
Studies show that laminar airflow isolation systems significantly reduce sepsis and cross-contamination in burn units, where immunocompromised patients face heightened infection risk — a direct outcome of correct classification.
US Federal Standard 209E and Legacy Classifications
ISO 14644-1 has largely replaced it, but Federal Standard 209E — withdrawn in 2001 — still shows up in older facility specs and renovation documents. When upgrading existing healthcare spaces, teams need to translate legacy classifications into current ISO equivalents:
- Class 100 ≈ ISO Class 5
- Class 10,000 ≈ ISO Class 7
- Class 100,000 ≈ ISO Class 8
Healthcare-Specific Regulatory Requirements
USP 797 - Sterile Compounding: USP 797 establishes minimum standards for preventing contamination in sterile preparations. Key requirements include:
- Primary Engineering Controls must maintain ISO Class 5 air quality under dynamic conditions
- Buffer rooms require ISO Class 7 with minimum 30 air changes per hour (ACPH), with at least 15 ACPH supplied through ceiling-mounted HEPA filters
- Ante-rooms need ISO Class 8 for positive pressure buffer rooms, ISO Class 7 for negative pressure areas
- Minimum positive pressure differential of 0.02-inch water column (5 Pa) between classified areas
USP 800 - Hazardous Drug Handling: USP 800 focuses on worker protection when handling chemotherapy and other hazardous drugs:
- Containment Secondary Engineering Controls (C-SEC) must be physically separated, externally vented, and maintained at negative pressure between 0.01-0.03 inches water column
- Sterile hazardous drug compounding requires minimum 30 ACPH in buffer rooms
- Non-sterile hazardous drug compounding needs minimum 12 ACPH
- All exhaust must be HEPA-filtered and discharged outside
CDC Biosafety Level 2 (BSL-2) Guidelines: Hospital pathology labs handling patient blood work and moderate-risk infectious agents like Salmonella, Hepatitis B, and HIV must follow BSL-2 requirements, including Class II Biological Safety Cabinets for aerosol-generating procedures, directional inward airflow, and self-closing doors.
Critical Design Components for Hospital Cleanrooms
HVAC Systems and Air Filtration
HEPA and ULPA Filtration: HEPA (High-Efficiency Particulate Air) filters form the foundation of cleanroom air quality, removing 99.97% of particles at 0.3 microns (the most penetrating particle size). ULPA (Ultra-Low Penetration Air) filters provide even greater efficiency at 99.999% for applications requiring extreme sterility, though standard hospital cleanrooms typically rely on HEPA filtration.
Air Changes Per Hour (ACH): ACH rates determine how quickly room air is replaced with filtered air. ASHRAE Standard 170 requires minimum 20 total ACH for operating rooms, with at least 4 outdoor ACH. USP 797 buffer rooms need ≥30 ACPH. Higher ACH rates dilute contaminants faster but increase energy costs.
Airflow Patterns:
- Unidirectional (Laminar) Flow: Air moves in parallel streamlines at uniform velocity, sweeping particles away from critical zones. ASHRAE 170 specifies unidirectional downward airflow in ORs, covering surgical areas with diffuser arrays extending 12 inches beyond the table.
- Turbulent Flow: Used in less critical zones like ante-rooms, where mixing dilutes contaminants rather than directing them away.
Recent systematic reviews indicate that laminar airflow ventilation does not consistently reduce surgical site infections compared to conventional turbulent ventilation for general surgeries, though HEPA filtration remains essential.
Pressure Control and Airflow Management
Pressure differentials prevent contamination by controlling airflow direction between zones.
Positive Pressure Systems: Used in sterile environments (USP 797 compounding, operating rooms), positive pressure pushes air outward, preventing unfiltered air from entering. The minimum differential is 0.02-inch water column (5 Pa) between classified areas.
Negative Pressure Systems: Hazardous areas (USP 800 compounding, pathology labs) use negative pressure to pull air inward and exhaust it outside through HEPA filters, containing potentially dangerous contaminants. USP 800 requires negative pressure between 0.01-0.03 inches water column.
Pressure Cascade Design: Progressive pressure differentials between adjacent zones prevent cross-contamination. For example: Buffer Room (+0.05") → Ante-room (+0.03") → Corridor (0.00").
Temperature and Humidity Control Systems
Precise environmental control prevents microbial growth and ensures material stability.
Temperature Requirements:
- Compounding pharmacies must maintain 20°C (68°F) or cooler to ensure worker comfort in full cleanroom garb and drug stability
- Operating rooms typically operate at 68-75°F (20-24°C)
Humidity Control: Relative humidity should stay below 60% to prevent microbial proliferation. ASHRAE 170 specifies 20-60% RH for operating rooms. Continuous monitoring via external readouts supports daily compliance logging.
Personnel and Material Flow Design
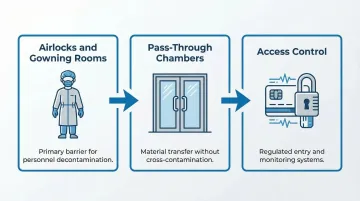
Three physical design elements work together to control contamination at the human and material level:
- Airlocks and Gowning Rooms: Ante-rooms separate "dirty" and "clean" sides, giving personnel space to complete progressive gowning steps before entering sterile areas. Modular cleanroom designs — like those ACH Engineering builds for hospital clients — can integrate these spaces into new construction or retrofit them into existing facilities.
- Pass-Through Chambers: Interlocked pass-throughs move supplies between pressure zones without opening both doors simultaneously, preserving differential control.
- Access Control: Electronic entry systems restrict access to authorized personnel and generate audit trails required by regulatory agencies for compliance documentation.
Healthcare-Specific Cleanroom Applications
Operating Room Cleanroom Design
Operating rooms represent one of the most visible cleanroom applications, though the relationship between ventilation and infection rates is more nuanced than commonly assumed.
HEPA filters direct clean air over the surgical field to minimize surgical site infections (SSIs). The surgical field targets ISO Class 5 conditions during operation, with unidirectional airflow covering the patient and surgical team.
The laminar flow debate complicates OR design decisions. A systematic review in The Lancet Infectious Diseases found that laminar airflow ventilation does not reduce SSIs compared to conventional turbulent ventilation for general surgeries, and additional studies support this finding. The high cost of unidirectional airflow canopies may not yield proportional infection control benefits — but HEPA filtration is still required under all current standards.
Current guidelines recommend:
- ISO Class 5–7 classification for surgical suites
- Minimum 20 total air changes per hour (ACH)
- Controlled temperature and humidity
- Proper unidirectional airflow patterns over the surgical field
USP 797 Sterile Compounding Pharmacies
Sterile compounding pharmacies prepare non-hazardous medications in controlled environments to prevent contamination. USP 797 defines a layered pressure cascade that keeps the cleanest zones protected at all times.
The pressure sequence runs: Ante-room (ISO 8) → Buffer Room (ISO 7) → Primary Engineering Control (ISO 5), with positive pressure increasing toward the cleanest area.
Design requirements include:
- Positive pressure throughout, with ISO Class 7–8 classifications
- Buffer rooms with minimum 30 ACPH, at least 15 ACPH through ceiling-mounted HEPA filters
- ISO Class 5 laminar airflow hoods for direct drug manipulation
- Ante-rooms with dirty-to-clean progression, scrub areas, and proper entry protocols
USP 800 Hazardous Drug Compounding Facilities
Chemotherapy and other hazardous drugs require containment-focused design to protect workers and the environment — the opposite pressure logic from USP 797.
Containment design specifications:
- Negative pressure relative to adjacent spaces (0.01–0.03 inches water column)
- Minimum 30 ACPH for sterile compounding; 12 ACPH for non-sterile
- Direct external exhaust with HEPA filtration
- Physical separation from other pharmacy operations
Required equipment includes Containment Primary Engineering Controls (C-PECs) such as biological safety cabinets, sealed hazardous drug storage, dedicated decontamination zones, and closed-system drug transfer devices.
Pathology and Microbiology Laboratories (BSL-2)
Hospital pathology labs handling patient specimens and infectious agents follow CDC Biosafety Level 2 protocols. These facilities handle moderate-risk agents — including Salmonella, Hepatitis B virus, HIV, and other bloodborne pathogens common in clinical diagnostics.
Key design features include:
- Class II Biological Safety Cabinets for procedures with aerosol or splash potential
- Directional inward airflow to contain aerosols
- Non-porous work surfaces resistant to disinfectants
- Handwashing sinks and self-closing doors
- Negative pressure is recommended but not always required by code
Specialized Healthcare Cleanroom Applications
Beyond the primary pharmacy and lab environments, several other clinical settings require purpose-built cleanroom conditions.
Burn Units
Burn patients with compromised skin barriers face extreme infection vulnerability. Laminar airflow isolation systems significantly reduce sepsis and cross-contamination in burn units, using positive pressure and HEPA filtration to exclude pathogens like Pseudomonas and Serratia.
Isolation Rooms
Patients with infectious diseases require negative pressure isolation with ante-rooms to prevent pathogen escape. Design includes self-closing doors, dedicated ventilation, and continuous pressure monitoring.
Cell and Gene Therapy Preparation
Hospital-based advanced therapy production requires GMP-level environments. Critical processing zones meet ISO Class 5 standards within ISO Class 7 backgrounds, typically housed within Class II Type A2 Biological Safety Cabinets.
Material Selection and Construction Considerations
Wall, Ceiling, and Floor Materials
USP 797 and FGI Guidelines mandate surfaces that are "smooth, impervious, free from cracks and crevices, and non-shedding" to prevent microbial harborage.
Approved Materials:
- Flooring: Seamless, heat-welded vinyl with integral coved bases extending at least 6 inches up walls eliminates cleaning dead zones
- Walls: Epoxy-coated gypsum board or high-pressure laminate panels resist harsh cleaning agents and prevent particle shedding
- Ceilings: Monolithic, scrubbable surfaces with gasketed access openings in restricted areas
Coved corners and sealed penetrations are equally important — they eliminate the gaps where particles accumulate and standard cleaning tools can't reach. ACH Engineering's modular systems include cleanroom coving that delivers compliant, seamless wall-ceiling and wall-floor connections per cGMP requirements.

Surface coatings containing copper and silver can further reduce bacterial burden. Copper alloys destroy pathogens like MRSA and C. difficile, though these treatments supplement rather than replace standard cleaning protocols.
Doors, Windows, and Pass-Throughs
Door Requirements:
- Swing direction matches pressure design (outward for positive pressure, inward for negative)
- Self-closing mechanisms maintain pressure differentials
- Flush designs prevent particle accumulation
- Interlocked pass-throughs prevent simultaneous opening
ACH Engineering's flush cleanroom doors are built to sustain pressure differentials and cleanliness classifications during routine human and material movement — without requiring operational workarounds.
Vision panels allow staff to monitor personnel and processes from outside the cleanroom, maintaining visual oversight without opening doors or compromising sterility.
Furniture, Equipment, and Fixtures
Furnishings and equipment in healthcare cleanrooms must withstand repeated disinfection without degrading or shedding particles:
- Non-shedding materials with smooth, sealed surfaces
- Stainless steel or powder-coated metal construction rated for harsh disinfectants
- Adjustable work surface heights to accommodate different procedures
- Anti-fatigue matting to reduce staff strain during extended procedures
- Equipment layouts that support workflow while maintaining cleanroom protocols
Modular vs. Traditional Cleanroom Construction
Advantages of Modular Cleanroom Systems
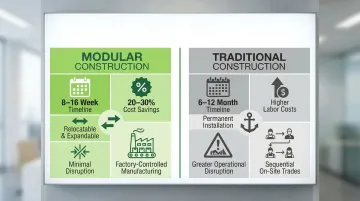
For healthcare facilities that can't wait 6-12 months for a new cleanroom, modular construction changes the timeline entirely.
Time Savings: Modular techniques deliver hospital ward shells in as little as 4 weeks, with complete projects in 7 weeks. Emergency isolation units using modular construction have been assembled in just 5 days. This represents 20-30% reduction in overall construction timelines compared to traditional methods requiring 6-12 months.
ACH Engineering's modular cleanrooms typically install in 8-16 weeks, with design phases taking only a couple of weeks followed by brief delivery periods and rapid on-site assembly.
Beyond speed, modular systems offer meaningful cost and operational advantages:
- Cost savings of 20-30% through factory-controlled manufacturing, reduced on-site labor, and less material waste. Stick-built construction may look cheaper upfront, but skilled labor costs typically close that gap.
- Expandable and relocatable — modular cleanrooms can be reconfigured, moved, or repurposed as patient volumes shift or departments reorganize. That flexibility simply isn't possible with permanent construction.
- Minimal disruption to active areas — because components arrive prefabricated, on-site assembly is fast and contained, protecting adjacent hospital operations throughout the build.
Traditional Stick-Built Construction Considerations
Traditional construction still makes sense in specific circumstances — particularly for complex integrations with existing hospital infrastructure, highly customized architectural requirements, or permanent installations where no future changes are anticipated.
That said, the trade-offs are real:
- Timelines of 6-12 months due to sequential on-site labor
- Higher costs from extended skilled labor requirements
- Greater disruption to active hospital operations throughout the build
- Wet finish drying times that push project schedules further out
Implementation and Project Management
Turnkey Approach: ACH Engineering provides comprehensive turnkey solutions where a single provider handles design, engineering, construction, commissioning, and certification. This eliminates the need to coordinate multiple vendors and ensures clear accountability throughout the project lifecycle.
Phased Construction: Maintaining pharmacy or laboratory operations during renovation requires careful phasing. Modular construction's rapid installation enables facilities to resume work quickly, though specific phased approaches should be discussed with the provider based on operational requirements.
Maintenance, Monitoring, and Compliance
Routine Cleaning and Disinfection Protocols
Healthcare cleanrooms require rigorous daily and periodic cleaning schedules using approved disinfectants.
Cleaning Requirements:
- Daily cleaning of all surfaces following Standard Operating Procedures (SOPs)
- Periodic deep cleaning on weekly or monthly schedules
- Approved EPA-registered disinfectants effective against target pathogens
- Documentation of all cleaning activities for regulatory compliance
Surface sampling and viable air sampling verify cleaning effectiveness. USP 797 — widely referenced across North American healthcare facilities, including Canadian compounding pharmacies — mandates viable sampling every 6 months for Category 1 & 2 CSPs, monthly for Category 3 CSPs.
Environmental and Particle Monitoring Systems
Continuous Monitoring:
- Pressure differentials must be monitored continuously with daily documentation
- Particle counting performed at least every 6 months during certification
- Temperature and humidity logged daily with alarm systems for excursions
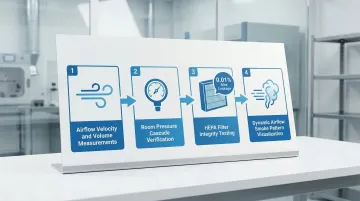
Certification Testing: USP 797 requires certification every 6 months, including:
- Airflow velocity and volume measurements
- Room pressure cascade verification
- HEPA filter integrity testing (maximum 0.01% leakage)
- Dynamic airflow smoke pattern visualization
Research shows microbial growth can run higher in ISO Class 7 support zones than in ISO Class 8 transition zones — a finding that makes continuous monitoring between certification cycles essential, not optional.
Preventive Maintenance and Calibration
Regular Maintenance:
- HVAC systems inspected and serviced quarterly
- Pre-filters replaced monthly to quarterly depending on loading
- HEPA filters assessed during semi-annual certification, replaced when integrity fails or pressure drop indicates end-of-life (typically 6-12 months in hospital environments)
- Control systems calibrated annually
Annual Certification: When selecting a third-party certification vendor, confirm they provide documented airflow testing, HEPA filter integrity verification, and full environmental mapping reports — these records form the audit trail regulators and accreditation bodies will expect to review.
Choosing the Right Cleanroom Partner for Your Healthcare Facility
Selecting a qualified cleanroom provider is critical for project success and long-term performance.
Essential Qualifications:
- Demonstrated experience with healthcare projects and regulatory requirements
- Deep understanding of USP 797/800 and ISO 14644-1 standards
- Membership in professional organizations like ISPE (International Society for Pharmaceutical Engineering)
- Professional engineering certifications (ACH Engineering holds memberships in PEO, PMI, ISPE, and IHSA)
- Project management expertise for complex healthcare environments
Beyond credentials, look for a partner who can deliver end-to-end. Comprehensive solutions covering design, engineering, construction, commissioning, certification, and staff training simplify coordination and ensure single-point accountability — no finger-pointing between separate contractors when issues arise.
ACH Engineering offers this turnkey approach for healthcare facilities across North America, with a structured five-stage process spanning initial estimate, design, construction, commissioning, and final validation. Their modular systems are purpose-built for healthcare applications — operating theaters, Central Sterile Supply Departments (CSSD), and isolation rooms — and their team operates from Ontario to Alberta, serving the broader North American market.
Frequently Asked Questions
What is the typical cost range for installing a hospital cleanroom?
Costs vary based on size, ISO classification, HVAC complexity, and whether it's new construction or renovation. Modular systems typically deliver 20-30% savings over traditional construction through reduced labor and faster installation. Contact ACH Engineering for a project-specific estimate.
How long does it take to design and install a cleanroom in an operating hospital?
Modular cleanroom installations complete in 8-16 weeks: design takes a couple of weeks, followed by brief delivery periods, then just a few weeks for on-site assembly and commissioning. Traditional construction typically requires 6-12 months due to sequential trades and drying times.
Careful phasing minimizes disruption to ongoing healthcare operations. Modular construction's prefabricated components enable facilities to resume critical operations much faster than traditional methods.
What are the ongoing operational costs for maintaining a healthcare cleanroom?
Budget for these recurring expenses:
- HVAC operation (the largest ongoing cost, running continuously to maintain air changes and environmental conditions)
- Filter replacements — pre-filters monthly to quarterly, HEPA filters typically every 6-12 months
- Semi-annual third-party certification per USP 797
- Approved disinfectants, cleaning materials, and ongoing staff training
Can existing hospital spaces be converted into compliant cleanrooms?
Yes, many existing spaces can be retrofitted with proper assessment. Key factors include structural capacity, ceiling height for ductwork, existing HVAC infrastructure, and architectural constraints. Modular systems are well-suited for retrofits, with prefabricated components assembled on-site to minimize disruption. A qualified engineer can identify any limitations compared to purpose-built facilities.
What certifications and testing are required after cleanroom installation?
Commissioning requires airflow measurements, HEPA filter integrity testing (maximum 0.01% leakage), particle counting, pressure differential verification, temperature and humidity mapping, and smoke studies for airflow visualization. Ongoing recertification occurs every 6 months per USP 797.
How do you maintain hospital operations during cleanroom construction or renovation?
Strategies to minimize disruption include:
- Complete work in phases to keep partial operations running
- Relocate services temporarily to alternate spaces during active construction
- Schedule noisy or disruptive tasks during nights or weekends
- Use modular prefabricated components to reduce on-site assembly time
Modular installations typically complete in weeks rather than months, significantly reducing the impact on patient care areas.
Ready to discuss your hospital cleanroom project? ACH Engineering provides comprehensive turnkey solutions for healthcare facilities across North America. Contact their team at sales@achengineering.com or +1 647-406-5721 to discuss your specific requirements and receive a detailed assessment.


