
Introduction: The Critical Role of Storage in Cleanroom Performance
Pharmaceutical and medical device manufacturers invest heavily in cleanroom infrastructure, yet many still struggle with contamination events. The hidden culprit? Poor storage practices that undermine even the best-designed facilities.
The financial stakes are staggering. A single mold contamination event traced to improper storage resulted in C$24 million in criminal and civil penalties for one pharmaceutical manufacturer.
Beyond regulatory fines, hospitals lose nearly C$650,000 annually in unaccounted supplies due to inventory inaccuracies, with an additional C$2.6 million impact on patient reimbursement.
This guide covers cleanroom storage best practices—from regulatory clearance requirements and material specifications to inventory protocols—helping you avoid costly contamination events and compliance violations.
TLDR:
- Maintain minimum clearances: 8-10" from floor, 18" from ceiling, 2" from walls
- Choose 316 stainless steel over 304 for superior resistance to harsh disinfectants
- Implement FIFO rotation systems with barcode/RFID tracking for inventory control
- Clean storage surfaces daily in high-touch areas, with monthly deep cleaning schedules
- Calculate capacity by turnover rates, not maximum space, to avoid overcrowding
Essential Standards and Requirements for Cleanroom Storage
Spatial Clearance Requirements
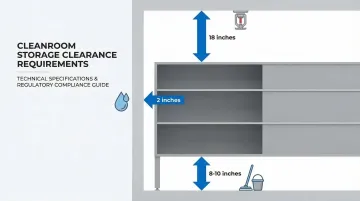
Proper clearances are non-negotiable for maintaining sterility and regulatory compliance. These measurements are mandated by AAMI, AORN, and CDC guidelines:
Minimum clearance standards:
- 8-10 inches from floor - Prevents contamination during cleaning and protects from floor-level contaminants
- 18 inches from ceiling - Ensures fire sprinkler effectiveness per NFPA 13 and maintains airflow
- 2 inches from exterior walls - Prevents condensation contact and facilitates airflow
These requirements become more stringent in higher-grade cleanrooms. In ISO Class 5 environments, storage must not disrupt unidirectional laminar airflow patterns.
Any obstruction creates turbulence that can reintroduce contaminants to sterile areas, compromising the airflow essential for sweeping particles away from critical zones.
Environmental Control Parameters
Temperature, humidity, and pressure differentials directly impact storage integrity:
Temperature control:
- USP <797> recommends maintaining cleanroom suites at 20°C (68°F) or cooler
- AAMI ST79 specifies sterile storage areas at or below 75°F (24°C)
- Temperatures above these thresholds promote microbial growth and compromise packaging materials
Humidity management:
- Maintain relative humidity below 60% per USP <797> to prevent microbial growth
- Keep minimum RH at 30% per ASHRAE guidelines to minimize static electricity that attracts particles
- Monitor continuously—humidity fluctuations can cause packaging materials to degrade
Pressure differentials:
- Maintain minimum 0.02-inch water column positive pressure between classified areas (USP <797>)
- FDA recommends 10-15 Pascals differential between adjacent rooms of different classifications
- Negative pressure trends must be investigated immediately—FDA warning letters frequently cite this deficiency
Air exchange requirements:
- ISO Class 7 rooms require minimum 30 air changes per hour (ACH)
- ISO Class 8 rooms require minimum 20 ACH
- Hazardous drug storage rooms need minimum 12 ACH per USP <800>
- Inadequate air exchanges allow particle accumulation, compromising sterility even when packaging remains intact

Regulatory Compliance Framework
Multiple regulatory bodies govern cleanroom storage design:
Key regulatory standards:
- FDA 21 CFR 211.42(c)(10) - Mandates operations occur within defined areas of adequate size to prevent contamination and mix-ups
- ISO 14644 Part 1 - Governs classification by particle concentration
- ISO 14644 Part 4 - Covers design and construction requirements for cleanroom infrastructure
- AAMI ST79 - Provides guidance on steam sterilization and sterile storage in healthcare facilities
A 2025 FDA warning letter cited Daewoo Pharmaceutical for storing machine tools and spare parts directly within an ISO 5 aseptic processing area, violating 21 CFR 211.42(c)(10).
Documentation requirements:
- Continuously monitor and document pressure differentials, temperature, and humidity
- Maintain cleaning logs with dates, personnel, and disinfectants used
- Track sterilization dates and expiration for all stored items
- Document staff training and competency assessments
- Prepare for inspection readiness with organized, accessible records
Access Control and Traffic Flow
Personnel represent the primary contamination source in cleanrooms. Restricting and controlling access to sterile storage areas is critical.
Access control best practices:
- Implement electronic card systems limiting access to authorized, trained personnel only
- Establish designated pathways that minimize traffic through sterile storage zones
- Create a "line of demarcation" in ante-rooms separating clean and dirty sides per USP <797>
- Position storage near the buffer area to minimize travel distance and contamination risk
- Install monitoring systems tracking door openings to reduce particle counts
Traffic flow optimization:
- Design storage layouts where oldest items (requiring first use) are most accessible
- Position high-turnover items near retrieval points to reduce movement
- Create one-way traffic patterns where possible to prevent cross-contamination
- Minimize the number of personnel entering storage areas simultaneously
ACH Engineering incorporates access control planning from the design phase, using modular solutions that allow optimal storage placement while maintaining flexibility for future layout adjustments as operational needs evolve.
Types of Cleanroom Storage Solutions
Mobile Storage Systems
Mobile cabinets with solid shelves offer flexibility and cleaning accessibility. These systems mount on cleanroom-grade casters, allowing repositioning for deep cleaning or layout optimization.
This flexibility addresses one of cleanroom maintenance's biggest challenges: accessing floor surfaces beneath fixed equipment.
Advantages:
- Enable access to floor surfaces for thorough cleaning
- Adapt to changing workflow patterns without permanent modifications
- Reduce fixed obstructions that disrupt airflow
- Facilitate space reconfiguration as storage needs evolve
Selection considerations:
- Use cleanroom-grade casters with sealed bearings that prevent grease leakage
- Choose non-marking materials (typically polyurethane) that won't leave residue on cleanroom floors
- Ensure wheels lock securely to prevent movement during item retrieval
- Verify load capacity matches your storage requirements to prevent wheel failure
Mobile storage works best for medium-turnover items requiring periodic access rather than constant retrieval. For high-frequency access items, fixed installations near workstations prove more efficient.
Wall-Mounted Storage Units
Wall-mounted cabinets maximize usable floor space in space-limited cleanrooms while keeping floors clear for cleaning.
Benefits:
- Eliminate floor contact, maintaining required clearances automatically
- Free floor space for equipment and personnel movement
- Simplify floor cleaning by removing obstructions
- Integrate seamlessly with modular cleanroom wall panel systems
Installation requirements:
- Anchor securely to wall studs or structural supports—not just to wall panels
- Maintain 2-inch clearance from exterior walls even when wall-mounted
- Ensure mounting hardware doesn't penetrate cleanroom envelope and compromise pressure differentials
- Verify weight capacity of wall structure before installation
Modular cleanroom wall systems can support integrated storage during initial design, eliminating the need for complex retrofitting and ensuring proper structural support from the start.
Open vs. Closed Shelving Systems
The choice between open and closed shelving impacts contamination protection, accessibility, and cost.
| Factor | Open Shelving | Closed Cabinet Systems |
|---|---|---|
| Initial Cost | 30-50% lower | Higher upfront investment |
| Best For | ISO Class 7-8 environments | ISO Class 5-6 environments |
| Access Speed | Quick visual inventory | Requires door opening |
| Cleaning Frequency | More frequent | Reduced maintenance |
| Contamination Protection | Requires barrier covers | Superior sealed protection |
| Ideal Use Case | High-turnover items | Low-turnover, sensitive items |
Choose based on three primary factors: your ISO classification requirements, item turnover rate, and budget constraints. Classes 5-6 typically require closed systems, while Classes 7-8 may use covered open shelving.
Container and Tray Storage Solutions
Beyond general shelving, healthcare and pharmaceutical cleanrooms need specialized storage for wrapped surgical trays, sterilized containers, and medical devices.
Proper design prevents package damage that compromises sterility.
Proper stacking methods:
- Never stack wrapped items more than 2-3 high—pressure compromises sterile barriers
- Use dedicated tray racks with individual slots preventing item-to-item contact
- Position heavier containers on lower shelves to prevent crushing lighter items below
- Maintain spacing between items for air circulation and visual inspection
Container system benefits:
- Rigid containers resist crushing and puncture better than flexible wraps
- Reduce package tearing by 40-60% compared to wrapped trays
- Improve stock rotation visibility with standardized sizes
- Enable more efficient space utilization through uniform stacking
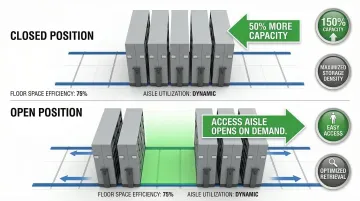
Track-Based High-Density Storage
Track-mounted shelving systems can increase storage capacity by up to 50% in existing cleanroom footprints by eliminating fixed aisles.
System design:
- Shelving units mount on floor tracks, sliding to create access aisles only where needed
- Compact all other units together when not in use
- Mechanical-assist or powered systems reduce physical effort for personnel
ROI considerations:
- Initial investment is 2-3x higher than static shelving
- Payback period typically 3-5 years through avoided cleanroom expansion costs
- Most cost-effective when cleanroom space costs exceed $500-1,000 per square foot
- Justified when storage needs are growing but cleanroom expansion is cost-prohibitive
Critical design requirement: Floor tracks must be cleanable without particle accumulation. Specify sealed track systems with smooth surfaces and ensure cleaning protocols address track channels thoroughly.
Material Selection for Cleanroom Storage
Stainless Steel Storage Options
Stainless steel dominates cleanroom storage due to its non-porous surfaces, chemical resistance, and cleanability. However, selecting the right grade is critical for long-term performance and cost-effectiveness.
304 vs. 316 stainless steel:
- 304 stainless steel: Cost-effective for general applications with good corrosion resistance, but vulnerable to chlorides and harsh disinfectants
- 316 stainless steel: Contains 2-3% molybdenum providing superior resistance to pitting and crevice corrosion from bleach and sporicidal agents
- Cost differential: 316 is 30-40% more expensive than 304, but prevents premature equipment failure in high-disinfectant environments
- Recommendation: Use 316 stainless steel for pharmaceutical and biotech cleanrooms using aggressive disinfection protocols
Solid vs. wire shelving:
- Solid shelves prevent small items from falling through and simplify thorough cleaning
- Wire shelving allows air circulation but creates crevices where particles accumulate
- This design requires more frequent cleaning and inspection
- Solid shelves with perforations offer a compromise—air circulation with easier cleaning
Polymer and Alternative Materials
While stainless steel sets the standard, antimicrobial polymer storage solutions offer cost-effective alternatives with built-in contamination resistance for less demanding applications.
Polymer advantages:
- Built-in antimicrobial properties inhibit bacterial growth on surfaces
- 40-60% lower initial cost compared to stainless steel
- Lighter weight that simplifies installation and reconfiguration
- Complete corrosion resistance to all disinfectants
Durability considerations:
- Lower impact resistance than stainless steel—more prone to denting or cracking
- UV exposure can cause degradation over time in some polymer formulations
- Temperature limitations that require verification against your cleanroom range
- Chemical compatibility varies by polymer type—verify resistance to your specific disinfectants
Lifecycle cost comparison:
| Material | Initial Cost | Lifespan | Maintenance | Best For |
|---|---|---|---|---|
| Stainless Steel | Higher | 15-20 years | Minimal | High-disinfectant environments |
| Polymer | Lower (40-60% less) | 8-12 years | Moderate | ISO Class 8, non-sterile areas |
Calculate total cost of ownership over your expected facility life, not just the purchase price. ACH Engineering evaluates these tradeoffs during the cleanroom design phase to optimize both performance and budget for each cleanroom project.
Surface Finish and Edge Design
Smooth, crevice-free surfaces with rounded edges prevent particle accumulation and wrapper damage.
Essential design features:
- Rounded edges prevent tearing of sterile packaging during handling
- Continuous welds without gaps where particles can lodge
- Smooth surfaces without texture that traps contaminants
- Concealed fasteners that eliminate cleaning challenges
Electropolished finishes:
- Electrochemical process removes surface material creating microscopically smooth finish
- Enhances corrosion resistance by 30 times compared to passivation alone
- Reduces surface area available for microbial attachment
- Meets ASTM B912 standard for pharmaceutical and biotech applications
- Essential for ISO Class 5 environments and recommended for Classes 6-7

When standard finishes are acceptable:
- ISO Class 8 environments with less stringent requirements
- Storage areas for non-sterile components or raw materials
- Budget-constrained projects where electropolishing adds 15-25% to material costs
Best Practices for Organizing and Maintaining Cleanroom Storage
Stock Rotation and Inventory Management
AAMI ST79 explicitly recommends FIFO (First-In, First-Out) rotation to minimize risks of using expired or compromised items.
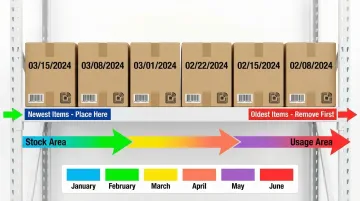
"Left to right" FIFO system:
- Place newest items on the left side of shelves
- Remove oldest items from the right side for use
- Creates intuitive visual flow that staff can follow without extensive training
- Prevents older items from being pushed to the back and forgotten
Labeling best practices:
- Display sterilization date prominently on front of package
- Include expiration date (if time-based sterility applies)
- Use color-coding by month to enable quick visual identification of age
- Position labels consistently in same location on all packages
Barcode and RFID tracking:
- Barcode systems require line-of-sight scanning but offer lower implementation costs
- RFID eliminates line-of-sight scanning, reducing movement and particle generation
- RFID enables real-time inventory visibility without manual scanning
- Supports FDA Unique Device Identification (UDI) requirements for medical devices
- Higher costs deliver ROI within 2-3 years through efficiency gains
Proper Handling Techniques
Improper handling compromises sterile barriers even when storage systems are optimal.
Correct removal methods:
- Lift items from underneath, supporting the full package
- Avoid dragging items across shelves (friction tears wrappers)
- Use both hands for packages over 5 pounds
- Avoid cradling sterile items against body or uniform
Carrying protocols:
- Hold packages away from body to prevent contamination transfer
- Carry at waist height for better control and visibility
- Make multiple trips rather than overloading and risking drops
- Inspect for damage during every touch—handling is the highest-risk moment
Training guidance:
- Demonstrate proper techniques during initial training with actual packages
- Conduct periodic competency assessments with direct observation
- Provide immediate corrective feedback when improper handling is observed
- Document training completion for regulatory compliance
Inspection Protocols
Visual inspection remains standard practice but has significant limitations. One study found operating room staff correctly identified defective wrappers with only 56.1% accuracy overall, and just 67.2% for large defects.
Comprehensive inspection checklist:
- Tears or punctures in packaging material
- Moisture or condensation inside packages
- Worn edges indicating repeated handling or age
- Proper sealing—all edges intact without gaps
- Valid sterilization indicators showing successful processing
- Intact tamper-evident seals on container systems
- Discoloration suggesting exposure to contaminants
Rejection criteria:
- Any package that is wet must be rejected—moisture breaches sterile barriers
- Tears or punctures of any size compromise sterility
- Broken seals indicate potential contamination exposure
- Expired sterilization dates (if time-based sterility applies)
- Packages dropped on floor must be reprocessed
When rejecting items, document the reason, quantity, and lot information for quality tracking and trend analysis.
Gowning Area Storage Integration
Beyond maintaining stored items, consider how storage integrates with facility workflow. Optimal gowning area design positions storage strategically around the line of demarcation.
PPE storage placement:
- Position garment racks on the clean side of the line of demarcation
- Install shelving for booties, shoe covers, and head covers at entry point
- Organize gloves by size in easily accessible dispensers
- Mount face mask/beard cover dispensers at eye level for quick access
Supporting infrastructure:
- Position benches for donning shoe covers before crossing line of demarcation
- Install full-length mirrors for personnel to verify proper gowning
- Place hand-washing stations before gowning area entry
- Position eyewash equipment near storage areas per safety regulations
Cabinet organization:
- Dedicate separate cabinets for each PPE type to prevent cross-contamination
- Label cabinets clearly with contents and size information
- Maintain par levels ensuring supplies never run out during operations
- Store backup supplies outside the cleanroom to minimize traffic
Planning these layouts during initial cleanroom design (rather than retrofitting) ensures compliance with clearance requirements and optimizes personnel flow.
Cleaning and Maintenance Schedules
USP <797> and facility SOPs dictate cleaning frequencies that must be followed consistently.
Recommended cleaning frequencies:
| Area | Minimum Frequency | Notes |
|---|---|---|
| Floors | Daily when compounding occurs | Use appropriate disinfectants for classification level |
| Counters/Work surfaces | Daily when compounding occurs | Clean before and after each shift |
| Storage shelving | Monthly | More frequently in high-touch areas |
| Walls | Monthly | Spot clean visible contamination immediately |
| Ceilings | Monthly | Critical in areas with overhead storage |
Disinfectant selection:
- ISO Class 5 areas require sterile 70% Isopropyl Alcohol (IPA)
- Use EPA-registered disinfectants appropriate for your classification level
- Rotate sporicidal agents weekly or monthly to destroy resistant spores
- Verify compatibility with storage materials—some disinfectants damage polymers
Cleaning without compromising stored items:
- Temporarily relocate items from shelves being deep cleaned
- Clean from top to bottom to prevent recontamination of cleaned surfaces
- Allow disinfectants to air dry completely before replacing items
- Document cleaning activities with date, personnel, and products used
Integration with Cleanroom Design
Incorporating storage planning during the design phase (rather than retrofitting) ensures optimal placement and compliance from the start.
Design phase advantages:
- Structural support for wall-mounted storage built into construction
- Infrastructure positioned for RFID/barcode scanners
- Clearances designed in from start (not discovered as violations later)
- Storage placement optimized for workflow
- 30-50% cost savings vs. retrofitting existing spaces
Turnkey solution benefits:
- Single point of responsibility for storage integration and cleanroom performance
- Coordinated installation minimizing disruption to operations
- Compliance verification before facility acceptance
- Comprehensive documentation supporting regulatory audits
Modular cleanroom approaches allow storage to be expanded or reconfigured as operational needs evolve, with additional features implemented without compromising environmental integrity.
Common Mistakes to Avoid in Cleanroom Storage
Overcrowding and Improper Stacking
FDA observations specifically cite facilities for "clutter within ISO Class 7 environments," including supply bins and unused equipment that impede cleaning and airflow.
Risks of overfilling:
- Disrupted airflow patterns compromise particle removal efficiency
- Storage surfaces become difficult to clean thoroughly
- Increases risk of package damage when retrieving items from back
- Makes visual inspection of package integrity nearly impossible
- Creates trip hazards and safety risks for personnel
Capacity calculation guidance:
- Calculate storage needs based on 2-week maximum inventory for high-turnover items
- Allow 30% buffer capacity for operational flexibility
- Never fill shelves more than 80% of available space
- Consider seasonal variations or production cycles in capacity planning
Beyond managing storage density, proper placement and clearances are equally critical for compliance.
Storing Items on Floors or Too Close to Ceilings
| Violation Type | Requirement | Consequences |
|---|---|---|
| Floor Storage | Minimum 8-10 inches above floor (AAMI/AORN standards) | Exposure to cleaning chemicals, contamination reservoirs, FDA warning letters, potential facility shutdown |
| Ceiling Clearance | Minimum 18 inches below ceiling | Interferes with fire sprinkler effectiveness, disrupts air circulation, blocks HEPA filter airflow |
Visual management tools:
- Apply floor markings showing minimum 8-10 inch clearance zones
- Install shelf labels indicating maximum storage height
- Use colored tape on shelves marking "do not exceed" lines
- Post signage reminding personnel of clearance requirements
Inadequate Staff Training and Supervision
Training program essentials:
- Initial comprehensive training before granting cleanroom access
- Hands-on practice with actual sterile packages and storage systems
- Periodic refresher training at least annually
- Skills assessments with direct observation and documentation
- Immediate corrective training when violations are observed
These fundamentals should be reinforced through ongoing education:
Ongoing education requirements:
- Updates when storage systems or protocols change
- Review of contamination events and root causes
- Reinforcement of proper handling techniques
Effective training requires active supervision to ensure consistent application:
Supervision strategies:
- Conduct random audits of storage area organization and cleanliness
- Monitor access logs for unusual patterns suggesting unauthorized entry
- Review inspection rejection rates to identify training gaps
- Establish accountability for storage area condition by shift or department
Frequently Asked Questions
What are the minimum clearance requirements for storing sterile items in a cleanroom?
AAMI and AORN standards require 8-10 inches from floor, 18 inches from ceiling, and 2 inches from exterior walls. These clearances prevent contamination, ensure proper air circulation, and maintain fire sprinkler effectiveness.
What type of shelving material is best for cleanroom storage?
316 stainless steel is standard for pharmaceutical and biotech cleanrooms due to superior corrosion resistance. Antimicrobial polymer alternatives cost 40-60% less and work well for ISO Class 7-8 environments.
How should sterile inventory be rotated to prevent expiration?
Use the "left to right" FIFO system where newest items are placed on the left and oldest items on the right are removed first. Label packages with sterilization dates and use colour-coding by month for quick identification.
What environmental conditions must be maintained in cleanroom storage areas?
Maintain temperature at or below 75°F (24°C) per AAMI ST79, and relative humidity between 30-60%. Maintain positive pressure differential of at least 0.02-inch water column between classified areas, with 20-30 air changes per hour depending on ISO classification.
Can open shelving be used for sterile storage in cleanrooms?
Open shelving is acceptable if shelves are covered with appropriate barriers protecting from dust and contamination. This approach works best for ISO Class 7-8 environments with less stringent requirements. For ISO Class 5-6 environments, closed cabinet systems with sealed doors provide superior contamination protection and are typically required. Open shelving requires more frequent cleaning but offers lower initial costs and easier visual inventory management.
How can I maximize storage capacity in a limited cleanroom footprint?
Track-based high-density storage systems increase capacity by up to 50% by eliminating fixed aisles. Wall-mounted solutions free floor space while maintaining required clearances. Modular systems can be reconfigured as needs change without permanent modifications.


