
Introduction
Contamination control isn't optional in industries where a single particle can compromise an entire production batch. Cleanroom storage cabinets serve as critical infrastructure in pharmaceutical manufacturing, semiconductor fabs, and biotechnology facilities—environments where regulatory compliance and product integrity depend on every component meeting stringent cleanliness standards.
The cleanroom storage cabinet market is experiencing rapid expansion, with 2026 representing a critical milestone. Regulatory mandates like EU GMP Annex 1 and FDA 21 CFR Part 211 are driving demand, alongside massive capital investments in semiconductor fabs and biologics manufacturing.
Global semiconductor leaders like Intel are investing $100 billion in new facilities with 600,000 sq ft of cleanroom space, while pharmaceutical companies expand sterile injectable production capacity.
This article examines market size projections through 2026, analyzes the regulatory forces driving growth, and explores emerging technologies like IoT-integrated smart cabinets. We also provide practical guidance for buyers evaluating total cost of ownership.
TLDR: Key Takeaways on Cleanroom Storage Cabinet Market Growth 2026
- Market projected to reach $1.15-$1.35 billion by 2030-2032, growing at 5.8-10.5% CAGR
- Regulatory compliance now mandates contamination-preventing storage (EU GMP Annex 1, FDA CGMP)
- Stainless steel dominates pharmaceutical and semiconductor cleanrooms
- Asia-Pacific accounts for 32% of global growth through 2026
- Material selection directly impacts compliance costs and contamination control effectiveness
Market Overview: Size, Growth & Projections for 2026
The cleanroom storage cabinet market has surpassed the half-billion-dollar threshold and is accelerating toward $1 billion-plus valuations by the early 2030s. Current estimates show:
- 2024 valuation: $794 million to $861 million
- 2030 projection: $1.15 billion (Strategic Market Research)
- 2032 projection: $1.35 billion (Future Market Report)
Growth trajectories vary by research methodology, with Compound Annual Growth Rates ranging from 5.8% to 10.5% through 2029-2032:
- Technavio: $646.2 million increase from 2024-2029 (10.5% CAGR)
- Future Market Report: 5.8% growth through 2032 (conservative estimate)

What explains this variance? Research methodologies differ in key areas:
- Market boundaries: Standalone cabinets only vs. integrated cleanroom furniture systems
- Geographic scope: North America/Europe focus vs. global estimates including Asia-Pacific expansion
- Product definitions: Varying criteria for what qualifies as cleanroom-grade storage
Post-Pandemic Acceleration
COVID-19 reshaped how industries approach contamination control. The pandemic created sustained demand for cleanroom infrastructure as companies recognized the strategic importance of contamination prevention and supply chain resilience.
This shift drove cleanroom storage investments across multiple sectors:
- Pharmaceutical manufacturers: Accelerated vaccine production facility construction with GMP-compliant storage requirements
- Semiconductor companies: Announced unprecedented fab expansions to address chip shortages
- Existing facilities: Upgraded from conventional storage to cleanroom-grade cabinets
- New construction: Incorporated contamination control from design phase rather than retrofitting
Key Drivers Fueling Cleanroom Storage Cabinet Market Expansion
Stringent Regulatory Standards and Compliance Requirements
Regulatory frameworks don't suggest cleanroom storage—they mandate it. The EU GMP Annex 1 (2022 revision) explicitly addresses storage infrastructure in multiple clauses:
- Clause 4.6 requires minimizing "projecting ledges, shelves, cupboards and equipment" to reduce dust accumulation and facilitate cleaning
- Clause 4.5 mandates all exposed surfaces be "smooth, impervious and unbroken" to minimize particle shedding
- Clause 4.10 identifies material transfer as a major contamination source, necessitating controlled storage
FDA CGMP regulations (21 CFR Part 211) establish equally strict storage requirements:
- § 211.42(b) mandates "adequate space for the orderly placement of equipment and materials to prevent mixups and contamination"
- § 211.80(b) requires components be "handled and stored in a manner to prevent contamination"
- § 211.65(a) specifies equipment surfaces must not be "reactive, additive, or absorptive"
Manufacturers must demonstrate compliance through validated storage systems that meet material, design, and cleanability standards. Non-compliance risks warning letters, production shutdowns, and product recalls—making compliant storage cabinets a regulatory necessity, not an operational preference.
Expansion of Pharmaceutical and Biotechnology Manufacturing
The pharmaceutical industry is projected to capture 45.7% of the global cleanroom technology market by 2026, driven by explosive growth in biologics, vaccines, and sterile injectables. All three require ISO 5-7 classified environments where every component—including storage—must support rigorous cleaning and disinfection protocols.
Capital expenditure in pharmaceutical cleanroom infrastructure has accelerated dramatically. Companies are building new facilities for cell and gene therapies, expanding vaccine production capacity, and modernizing legacy facilities to meet updated GMP standards.
Each new facility requires comprehensive storage solutions for active pharmaceutical ingredients (APIs), excipients, sterile garments, and cleanroom consumables.
Semiconductor and Electronics Industry Growth
Advanced semiconductor manufacturing has become extraordinarily sensitive to contamination. Production of 5nm, 3nm, and emerging 2nm chips requires ISO 1 cleanrooms where fewer than 10 particles (0.1 µm) per cubic meter are permitted. At these scales, semiconductor engineers say "every atom now counts"—a single particle can cause defects that destroy entire wafers worth millions of dollars.
The stakes drive massive investments in cleanroom infrastructure, including specialized storage:
- Intel: Investing up to $100 billion USD in Ohio facilities with 600,000 sq ft of cleanroom space
- TSMC: $65 billion USD Arizona investment for three fabs producing 4nm, 3nm, and 2nm chips with 30,000-45,000 wafers per month capacity
- Micron: Building a Singapore plant with 700,000 sq ft of cleanroom space for wafer output starting 2028
Each facility requires specialized storage for wafers, photomasks (reticles), chemicals, and cleanroom consumables—all demanding contamination-free environments.

Emphasis on Product Quality and Contamination Control
Industries are prioritizing contamination prevention because the cost of failure is astronomical. A contaminated production batch can mean:
- Pharmaceutical recalls affecting thousands of patients and costing millions in lost product and remediation
- Semiconductor yield losses where contaminated wafers represent weeks of production time and significant material costs
- Medical device recalls triggering regulatory investigations and liability concerns
Contaminated storage compromises entire production runs. A cabinet that sheds particles contaminates materials stored inside. One that can't be properly cleaned harbors microorganisms that transfer to sterile products. Such risks make proper storage infrastructure a quality control imperative, not just a compliance checkbox.
Market Segmentation: Products, Applications & Geographic Regions
Product Type Segmentation
Stainless Steel Cleanroom Storage Cabinets
Stainless steel dominates pharmaceutical and semiconductor applications due to exceptional durability and cleanability. 304 and 316 stainless steel grades offer corrosion resistance essential for environments using harsh cleaning agents and disinfectants.
Electropolished finishes eliminate grainy textures where pathogens hide, creating smooth surfaces that meet FDA/GMP requirements for non-reactive materials.
These cabinets command premium pricing—wall-mounted 304 stainless steel units can reach $4,401 USD—but offer superior total cost of ownership through longevity and reduced maintenance. They withstand repeated sterilization cycles without degradation and maintain structural integrity for decades.
Primary applications include pharmaceutical sterile manufacturing, biotechnology research, medical device production, and semiconductor wafer storage where material compatibility and cleaning validation are critical.
Polypropylene Cleanroom Storage Cabinets
For applications where stainless steel isn't required, polypropylene provides cost-effective alternatives with pricing ranging from $1,535 to $2,969 USD for advanced configurations. These cabinets offer:
- Broad chemical resistance to acids and alkalis
- Non-metallic construction ideal for metal-free environments
- Minimal particle shedding suitable for ISO 6-8 cleanrooms
- Lightweight properties simplifying installation and reconfiguration
Polypropylene excels in semiconductor wet chemistry areas, chemical storage rooms, and applications requiring corrosive chemical resistance where stainless steel may react. However, they typically aren't suitable for the most stringent pharmaceutical sterile manufacturing or ISO 1-5 semiconductor applications.

Application Segmentation
Cleanroom storage cabinets serve diverse applications across regulated industries:
- Pharmaceutical: Sterile drug manufacturing, compounding pharmacies, API storage
- Biotechnology: Cell culture materials, biological samples, research consumables
- Semiconductor: Wafer storage, photomask cabinets, chemical storage, cleanroom supplies
- Medical Devices: Implant components, diagnostic device manufacturing, sterile packaging materials
- Aerospace: Precision components requiring contamination-free storage
- Research Laboratories: University and corporate R&D facilities with cleanroom requirements
Regional Market Distribution
North America leads in market value, holding approximately 39.4% of cleanroom technology revenue in 2024. This dominance reflects established pharmaceutical manufacturing hubs, significant semiconductor investments driven by the CHIPS Act, and stringent regulatory enforcement by the FDA.
Europe maintains strong market share due to mature pharmaceutical industries and strict EU GMP compliance requirements. The 2022 Annex 1 revision has accelerated facility upgrades and new construction requiring compliant storage infrastructure.
The fastest growth is occurring in Asia-Pacific, projected to contribute 32% of global market expansion through 2029. Key drivers propelling this regional surge include:
- Massive semiconductor fab construction in Taiwan, China, Japan, and South Korea
- Expanding pharmaceutical manufacturing in India serving domestic and export markets
- Government initiatives modernizing manufacturing infrastructure
- Foreign direct investment in cleanroom-dependent industries
Latin America and Middle East & Africa represent emerging opportunities as these regions develop pharmaceutical manufacturing capabilities and attract technology sector investments.
Industries Driving Cleanroom Storage Cabinet Demand
Pharmaceutical and Nutraceutical Manufacturing
Pharmaceutical manufacturing is the largest end-user, requiring specialized storage for APIs, excipients, sterile garments, and cleanroom supplies throughout production facilities.
Sterile compounding facilities—whether hospital pharmacies preparing IV medications or compounding pharmacies creating customized formulations—must store materials in validated cleanroom environments that prevent microbial contamination.
The global pharmaceutical cleanroom market growth directly correlates with storage cabinet demand. As companies build new biologics facilities and expand sterile injectable capacity, they need GMP-compliant storage that supports cleaning validation and regulatory inspections.
ACH Engineering serves pharmaceutical, biotech, and medical device manufacturers across North America with cleanroom solutions that integrate optimized storage infrastructure, ensuring workflow efficiency and regulatory compliance.
Semiconductor and Electronics Production
Semiconductor contamination sensitivity has reached extraordinary levels. At 3nm and 2nm process nodes, manufacturers operate in ISO 1 cleanrooms where contamination control extends to every surface and component.
Storage cabinets must not shed particles, must resist chemicals used in fab processes, and must integrate with automated material handling systems.
Specific storage needs include:
- Wafer storage: Temperature and humidity-controlled environments protecting high-value silicon wafers
- Reticle/photomask cabinets: Specialized storage for optical masks where single particles cause defects
- Chemical storage: Cabinets resistant to corrosive chemicals used in etching and cleaning
- Cleanroom consumables: Particle-free storage for gloves, wipes, and garments
TSMC's addition of 30,000-45,000 wafers per month capacity for ≤2nm nodes in Arizona shows the scale of fab expansion driving storage infrastructure demand.
Biotechnology and Medical Device Industries
Biotechnology research and production require storage for cell culture materials, biological samples, and reagents in contamination-controlled environments. Medical device manufacturing—particularly implantable devices and diagnostic equipment—demands sterile component storage to ensure product safety and efficacy.
The medical device segment alone reached $403.30 million USD in 2019 and continues growing as manufacturers adopt cleanroom standards. Applications include:
- Robotic prosthetics requiring sterile component storage
- Diagnostic instruments demanding contamination-free assembly environments
- Implantable devices needing ISO-certified storage solutions
Storage solutions must match specific cleanliness classifications and material compatibility requirements.
Emerging Applications
Beyond traditional pharmaceutical and semiconductor sectors, cleanroom standards are expanding into new industries:
- Battery manufacturing: Lithium-ion production requires dry rooms (ultra-low humidity environments) with specialized storage for moisture-sensitive materials
- Aerospace: Precision component manufacturing adopting contamination control for satellite components and aircraft systems
- Cosmetics: Premium cosmetics manufacturers implementing cleanroom standards to ensure product purity
- Food & beverage: Aseptic processing facilities requiring cleanroom storage for ingredients and packaging materials
These emerging applications represent significant growth opportunities as more industries recognize contamination control benefits.
Challenges and Market Restraints
High Initial Investment Costs
Cleanroom-compliant storage costs 3-4 times more than conventional industrial furniture. This premium creates budget barriers, particularly for small-to-medium manufacturers and research facilities operating with limited capital budgets. A basic polypropylene cleanroom cabinet starts at $1,535, while stainless steel units easily exceed $4,000—compared to a few hundred dollars for standard storage.
These costs compound across entire facilities. Equipping a pharmaceutical manufacturing suite or semiconductor fab requires dozens of cabinets, with total investments including:
- Storage units: dozens of cabinets per facility
- Installation and integration costs
- Validation and compliance documentation
- Total investment: often reaching six figures
Limited Availability of Specialized Expertise
The cleanroom industry faces workforce shortages affecting project timelines and costs. Deloitte projects 100,000 semiconductor job openings annually through 2030, with similar shortages in pharmaceutical facility design and construction.
This talent gap delays projects as companies struggle to find professionals trained in cleanroom design, installation, and maintenance.
The expertise gap directly affects storage infrastructure decisions. Proper cabinet selection requires understanding ISO classifications, material compatibility, cleaning validation, and regulatory requirements—specialized knowledge that few professionals possess outside dedicated cleanroom firms.
Material and Supply Chain Considerations
Stainless steel price fluctuations create challenges for manufacturers maintaining fixed pricing and profit margins. Supply chain disruptions affecting stainless steel and specialized components can delay projects and increase costs unpredictably.
Environmental concerns are emerging as buyers prioritize sustainability. Traditional cabinet materials face recyclability challenges, and energy-intensive manufacturing processes conflict with corporate sustainability goals.
These considerations are driving demand for eco-friendly alternatives, though options remain limited in the cleanroom storage market.
Emerging Trends and Future Opportunities in Cleanroom Storage
Integration with Smart Monitoring Systems
IoT sensors and RFID tracking are transforming passive storage into intelligent systems providing real-time visibility.
Nexess Solutions' XL RFID Cabinet demonstrates this evolution, offering complete traceability for up to 400 items with automated inventory checks completed in 10 seconds and automatic tracking of calibration dates.
Smart cabinets integrate with building management systems to monitor:
- Temperature and humidity conditions affecting stored materials
- Door access tracking for security and contamination control
- Inventory levels triggering automatic reordering
- Predictive maintenance alerts based on usage patterns
In semiconductor fabs, systems like Fabmatics' CubeStocker use RFID for system-wide carrier identification, enabling automated material handling that reduces human intervention and contamination risk.
Sustainability and Eco-Friendly Designs
Beyond technological innovation, corporate sustainability goals are reshaping cabinet design and material selection. Key developments include:
- Energy-efficient designs reducing operational costs
- Cabinets manufactured from recyclable materials
- Reduced environmental footprint production processes
- Lifecycle assessments demonstrating total environmental impact
ISO 14644-16 standards addressing energy efficiency in cleanroom operations are pushing manufacturers to evaluate storage infrastructure's impact on facility energy consumption. This consideration now factors directly into sustainability metrics and operational cost analyses.
Modular and Customizable Solutions
Flexibility is increasingly important as facilities adapt to changing products, processes, and regulations. Modular cabinet systems offer:
- Adjustable shelving accommodating different storage needs
- Reconfigurable compartments adapting to new workflows
- Expansion capability supporting facility growth
- Quick installation and relocation for facility reconfigurations
The modular cleanroom construction market is growing at 8.5% CAGR, driving parallel demand for modular storage furniture that integrates seamlessly with flexible facility designs.
This trend particularly benefits industries like biotechnology where research focus shifts rapidly and storage needs evolve accordingly. Modular storage solutions align naturally with adaptable cleanroom infrastructures, enabling facilities to reconfigure entire environments—including storage—without costly reconstruction.
Key Considerations for Buyers and Investors
Cleanroom Classification Requirements
Cabinet selection must align with your facility's ISO classification. ISO 14644-1 defines air cleanliness classes from ISO 1 (most stringent) to ISO 9 (least stringent). Material selection, surface finish, and design features must match classification requirements:
- ISO 1-5: Require stainless steel with electropolished finishes, minimal particle generation, seamless construction
- ISO 6-7: May accommodate high-grade polypropylene or stainless steel depending on application
- ISO 8-9: Broader material options acceptable if cleaning protocols are validated
Regulatory inspections verify storage infrastructure matches documented cleanliness classifications. Mismatched cabinets create compliance risks even if the room itself meets air quality specifications.
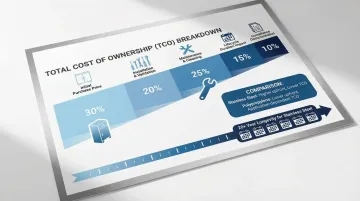
Total Cost of Ownership Analysis
Purchase price represents only one component of total cost. Comprehensive evaluation should include:
- Installation costs: Labor, integration with facility systems, validation testing
- Maintenance requirements: Cleaning labor, replacement parts, repair frequency
- Lifecycle duration: Stainless steel cabinets lasting 20+ years versus shorter-lived alternatives
- Compliance costs: Validation documentation, regulatory inspection preparation
- Operational impact: Downtime for cleaning, accessibility affecting workflow efficiency
Stainless steel's higher upfront cost often delivers lower total cost of ownership through durability, easier cleaning, and reduced replacement frequency.
Polypropylene may offer better economics for less critical applications or budget-constrained projects.
Integration with Complete Cleanroom Design
Beyond cost analysis, storage placement within your facility design significantly impacts operational efficiency. Optimal placement requires considering:
- Workflow optimization: Positioning cabinets to minimize material movement and contamination risk
- Material flow patterns: Ensuring storage locations support efficient production sequences
- Personnel traffic: Reducing congestion and maintaining proper gowning protocols
- Regulatory compliance: Meeting spacing requirements and facilitating inspections
Working with experienced controlled environment providers ensures storage placement integrates seamlessly with overall facility design. ACH Engineering's turnkey approach optimizes cabinet selection and positioning within the broader cleanroom layout, ensuring storage infrastructure supports operational efficiency while meeting regulatory requirements.
This integrated approach delivers better outcomes than selecting cabinets independently from broader facility planning.
Frequently Asked Questions
What is the projected size of the cleanroom storage cabinet market in 2026?
Market estimates for 2026 range from $850-$950 million, with projections reaching $1.15-$1.35 billion by 2030-2032 at a CAGR of 5.8-10.5%.
What are the main differences between stainless steel and polypropylene cleanroom storage cabinets?
Stainless steel provides superior durability and ease of cleaning for stringent pharmaceutical and semiconductor applications. Polypropylene offers cost advantages, chemical resistance, and lightweight properties for less critical applications.
Which industries are the largest consumers of cleanroom storage cabinets?
Pharmaceutical manufacturing, semiconductor production, and biotechnology research are the largest consumers. Medical device manufacturing, aerospace, battery production, and food processing represent fast-growing segments.
What regulatory standards govern cleanroom storage cabinet design and use?
ISO 14644 cleanroom standards and FDA regulations (21 CFR Part 211) establish air cleanliness and material requirements. Industry-specific guidelines influence cabinet design, material selection, and cleaning protocols.
Why is the Asia-Pacific region experiencing the fastest market growth?
Expanding pharmaceutical and semiconductor manufacturing in China, India, and Southeast Asia, combined with government infrastructure investments, are driving rapid adoption. The region is projected to contribute 32% of global growth through 2029.
What emerging technologies are shaping the future of cleanroom storage cabinets?
IoT integration and RFID tracking enable real-time monitoring, inventory management, and automated compliance documentation. Antimicrobial materials, energy-efficient designs, and modular configurations are also transforming the market.


