
Introduction: Why GMP Furniture Standards Matter for Controlled Environments
Furniture represents one of the most common contamination sources in pharmaceutical cleanrooms—yet remains frequently overlooked during facility design. According to EU GMP Annex 1 (revised 2022), all cleanroom surfaces must be "smooth, impervious and unbroken" to minimize particle shedding and microbial accumulation—including every piece of furniture in controlled environments.
Good Manufacturing Practice (GMP) standards extend beyond manufacturing processes to encompass all equipment and furniture within pharmaceutical cleanrooms.
Furniture that doesn't meet GMP requirements can shed particles, harbor microorganisms in crevices, obstruct critical airflow patterns, and degrade under rigorous cleaning protocols. Non-compliant furniture has been cited in FDA 483 observations as a contributing factor to contamination events that led to product recalls and production shutdowns.
You'll learn about material requirements, design standards, classification-specific requirements, and validation procedures necessary to ensure your cleanroom furniture supports contamination control rather than compromising it.
TLDR: Key Takeaways on GMP Furniture Standards
- 316L stainless steel required for Grade A/B areas; approved polymers acceptable for lower grades
- Continuous welds, radiused corners, and sloped surfaces eliminate contamination traps
- Grade A/B demands electropolished 316L with minimal footprint; Grades C/D allow 304 steel or polymers
- Validation requires material certificates, IQ/OQ records, and cleaning documentation
Understanding GMP Standards for Cleanroom Furniture
What GMP Means in the Context of Cleanroom Furniture
Good Manufacturing Practice (GMP) represents regulatory standards ensuring pharmaceutical products are consistently produced to quality standards.
While GMP principles traditionally focused on manufacturing processes, EU GMP Annex 1 (revised 2022) now specifically addresses sterile manufacturing and includes detailed requirements for cleanroom infrastructure—including furniture.
GMP furniture standards align with ISO 14644 cleanroom classifications but add pharmaceutical-specific requirements for materials, design validation, and documentation that go beyond general cleanroom practices. Working with a pharmaceutical cleanroom services provider ensures these requirements are addressed from day one.
The regulatory framework recognizes that furniture is not merely a convenience but a critical component of the contamination control strategy.
Key GMP Principles Applied to Furniture Design
Four fundamental pillars guide GMP furniture specifications:
- Quality by Design - Furniture must be fit for purpose from initial conception, with materials and construction methods selected specifically for cleanroom use
- Documentation - Complete records of material composition, surface finish specifications, and design features must be maintained
- Validation - Proven performance in maintaining sterility through cleaning validation studies and operational qualification
- Traceability - Full history from manufacturing through installation, maintenance, and any modifications
These principles translate into practical requirements. Furniture must actively support contamination control through particle minimization, ease of cleaning, and chemical resistance to disinfectants including sporicides. This is not passive compliance—furniture must be designed to contribute to sterility assurance.
Regulatory Framework and Compliance Requirements
Compliance involves meeting multiple regulatory standards simultaneously:
Primary Regulations:
- EU GMP Annex 1 - Mandates smooth, non-porous surfaces and cleanability by design
- FDA 21 CFR Part 211 - Requires equipment of appropriate design, adequate size, and surfaces that are non-reactive and non-absorptive
- ISO 14644-1 - Defines air cleanliness classification
- ISO 14644-5 - Provides operational guidance for cleanroom furniture and cleaning protocols
Furniture must be specified in the facility's Quality Management System and included in cleaning validation studies. Regulatory inspections will examine furniture specifications, installation qualification (IQ), operational qualification (OQ), and maintenance records as part of routine GMP audits.
Material Requirements for GMP-Compliant Furniture
Approved Materials for GMP Furniture
Material selection is the foundation of GMP furniture compliance. 316L stainless steel is the gold standard for critical areas (Grades A and B) due to its superior corrosion resistance, non-porous surface, and minimal particle shedding.
The molybdenum content (2%) in 316L provides exceptional resistance to pitting and crevice corrosion in chloride-rich environments common with aggressive disinfectants.
Approved alternative materials include:
- High-grade polymers - HDPE, polypropylene, PVDF for specific applications where stainless steel isn't required
- Electropolished 304 stainless steel - Acceptable for Grade C/D areas with less aggressive cleaning regimes
- Powder-coated steel - Only for Grade D support areas, must be pharmaceutical-grade with full curing and chemical resistance validation
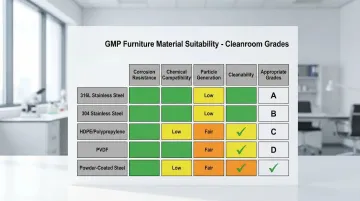
Material Comparison:
| Material | Corrosion Resistance | Chemical Compatibility | Particle Generation | Cleanability | Appropriate Grades |
|---|---|---|---|---|---|
| 316L Stainless Steel | Excellent | Excellent | Minimal | Excellent | A, B, C, D |
| 304 Stainless Steel | Good | Good | Low | Very Good | C, D |
| HDPE/Polypropylene | Very Good | Good | Low | Good | C, D (specific applications) |
| PVDF | Excellent | Excellent | Minimal | Excellent | B, C, D |
| Powder-Coated Steel | Fair | Fair | Moderate | Good | D only |
Material Properties Critical for GMP Compliance
Three critical properties determine material suitability for GMP environments:
Non-Shedding Performance:
- Materials must not release particles, fibers, or ions during operation or cleaning
- Requires inherent material stability and proper surface treatment
- Critical for preventing product contamination
Non-Porous Surface:
- Smooth finish prevents microbial harboring and biofilm formation
- Electropolished stainless steel should achieve Ra ≤ 0.8 μm (32 microinches) for critical areas
- Grade A environments benefit from smoother finishes (Ra ≤ 0.4 μm)
Chemical Inertness:
Materials must resist common disinfectants without degradation:
- 70% isopropanol solutions
- 3-6% hydrogen peroxide
- Quaternary ammonium compounds
- Sporicides including sodium hypochlorite
Furniture must withstand repeated cleaning cycles over a typical 10-15 year lifecycle. Surface integrity cannot degrade, corrode, or lose performance over this period.
Materials to Avoid in GMP Environments
Certain materials are prohibited due to inability to meet GMP requirements:
Prohibited Materials:
- Wood - Porous, impossible to clean or validate, sheds particles and harbors microorganisms
- Unsealed particle board or MDF - Continuous particle shedding, moisture absorption
- Carbon steel - Rust risk that introduces particulate contamination
- Fabrics and textiles - Particle and microbial harboring in fibers
- Porous plastics - Cannot be adequately cleaned or validated
The FDA has issued warning letters citing facilities for "particle board" and "chipping paint" on furniture, identifying these as direct contamination risks and GMP violations.
Surface Treatments and Finishes
Once you've selected appropriate materials, proper surface treatment is essential for GMP compliance.
Electropolishing is the preferred surface treatment for stainless steel in critical areas. This electrochemical process removes surface imperfections, creates a passive chromium oxide layer, and enhances corrosion resistance. The result is a microscopically smooth surface (smooth at the microscopic level) that resists particle adhesion and facilitates cleaning.
Powder coating requirements (when used in Grade D):
- Pharmaceutical-grade formulation
- Fully cured with no outgassing
- Free of cracks, chips, or imperfections
- Validated for chemical resistance to facility disinfectants
Any surface treatment must be documented with certificates of conformity and tested for particle generation under operational conditions.
Design Standards and Construction Features
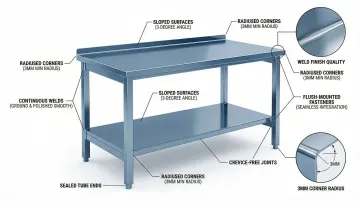
Crevice-Free and Seamless Construction
GMP furniture design must eliminate contamination traps. EU GMP Annex 1 states "there should be no recesses that are difficult to clean effectively."
Achieving this standard requires specific construction techniques:
Required construction techniques:
- Continuous welds - Fully ground and polished to match surface finish
- Sealed joints - No gaps or crevices where particles or moisture can accumulate
- Radiused corners - Minimum 3mm radius on all internal corners to facilitate cleaning
- Sloped surfaces - Minimum 3-degree angle for drainage, eliminating horizontal surfaces where particles settle
- Flush-mounted fasteners - No exposed threads or recessed screw heads
All hollow sections (tube ends) must be capped and sealed to prevent internal contamination that cannot be cleaned.
Mobility and Accessibility Features
Beyond construction standards, furniture must support operational requirements. This includes both mobility for flexible layouts and accessibility for thorough cleaning:
Mobility Requirements:
- Cleanroom-compatible casters with non-shedding wheels (stainless steel or approved polymer)
- Sealed bearings to prevent particle generation
- Locking mechanisms for operational stability
- Alternative: Wall-mounted designs that eliminate floor contact
Mobility directly impacts cleaning effectiveness. Access requirements include:
Cleaning Access:
- Minimum 150mm (6 inches) clearance underneath mobile furniture
- OR fully sealed to floor with coved connections
- No "dead spaces" that cleaning tools cannot reach
Ergonomic Design Within GMP Constraints
Balancing operator comfort with contamination control requires careful design:
- Adjustable height work surfaces - Typically 850-950mm for standing work, with adjustment mechanisms fully sealed
- Ergonomic seating - Any padding or cushioning must be fully encapsulated in non-porous, cleanable material with sealed seams
- Proper working heights - Reduce operator fatigue while maintaining cleanability
Comfort features must never compromise GMP compliance. Engineers must design adjustability and cushioning using cleanroom-compatible materials with fully sealed construction.
Integration with Cleanroom Systems
Furniture placement affects environmental control effectiveness. Proper integration requires careful consideration of airflow patterns and system design.
Airflow Considerations:
- Must not obstruct unidirectional (laminar) flow in Grade A areas
- Perforated or mesh surfaces on shelving in critical zones to maintain airflow
- Minimal footprint in first air zones
- Smoke visualization studies must validate final placement
FDA guidance states that "equipment should not obstruct airflow and, in critical areas, its design should not disturb unidirectional airflow."
Validation Requirements:
- Document furniture placement in facility airflow studies
- Validate designs do not create turbulence near open product
- Confirm no dead zones form in critical areas
Furniture Requirements by Cleanroom Classification
Requirements escalate significantly with cleanroom grade, reflecting increased contamination risk and sterility requirements. The table below outlines how material and design specifications vary across EU GMP Annex 1 classifications.
EU GMP Annex 1 Grade Requirements
| Grade | Particle Limit (≥0.5 μm/m³ at rest) | Material Requirements | Design Requirements | Furniture Presence |
|---|---|---|---|---|
| A | 3,520 | 316L electropolished stainless steel only | Fully welded, no horizontal surfaces, perforated for airflow | Absolute minimum |
| B | 3,520 | 316L preferred; high-grade polymers acceptable for specific applications | Similar to Grade A, must support unidirectional flow | Minimal |
| C | 352,000 | 304 or 316L stainless steel; high-quality polymers acceptable | Cleanable, non-shedding, radiused corners | Moderate |
| D | 3,520,000 | 304 stainless steel, quality polymers, powder-coated steel acceptable | Focus on cleanability and durability | Standard |

Grade A (Critical Zones)
Grade A zones demand zero-tolerance standards. Only 316L electropolished stainless steel is acceptable, with fully welded construction and ground, polished welds.
Key requirements include:
- No horizontal surfaces—all surfaces sloped minimum 3 degrees
- Perforated designs that maintain laminar airflow
- Minimal furniture presence—only essential items
Every piece faces rigorous scrutiny during regulatory inspections. Each item must be justified as essential to the aseptic process.
Grade B (Background Environment for Grade A)
As the background environment for Grade A operations, Grade B maintains strict controls with slightly more flexibility.
Material and design standards:
- 316L stainless steel strongly preferred
- High-grade polymers (PVDF, PTFE) acceptable for specific applications
- Must support unidirectional airflow without disrupting air patterns
- Continuous cleaning and disinfection protocols required
Furniture in these areas must withstand frequent application of sporicides as required by Annex 1 disinfectant rotation protocols.
Grades C and D (Less Critical Areas)
Moving to less critical classifications, Grades C and D allow broader material options while maintaining GMP standards.
Acceptable specifications include:
- 304 stainless steel or high-quality polymers
- Powder-coated steel in Grade D support areas
- More conventional furniture designs
- Cleanable, non-shedding surfaces with proper documentation
Requirements are less strict, but furniture still requires proper specification, validation, and maintenance to support the overall contamination control strategy.
Validation, Documentation, and Maintenance
Required Documentation Package
Material Certificates:
- Material composition verification (316L vs 304)
- Surface finish specifications (Ra values)
- Chemical compatibility data
- EN 10204 3.1 certificates for stainless steel
Design Qualification (DQ):
- Detailed drawings showing GMP-compliant features
- Rationale for material selection
- Cleanability assessment
- Airflow impact analysis
Installation Qualification (IQ):
- Verification of correct materials received
- Dimensional verification
- Surface finish inspection
- Proper installation confirmation
- Photographic documentation
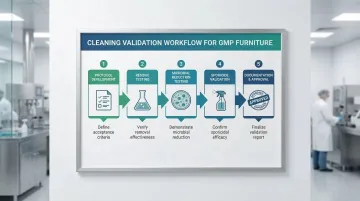
Cleaning Validation Requirements
Beyond documentation, furniture must undergo operational validation as part of your facility's cleaning qualification studies.
EU GMP Annex 1 requires that cleaning processes be validated to remove residues and minimize contamination to acceptable levels. North American facilities must meet equivalent FDA or Health Canada requirements.
Validation must demonstrate:
- Effective removal of process residues
- Effective removal of cleaning agents themselves
- Microbial reduction to acceptable levels
- Sporicide effectiveness on furniture materials
- Repeatability across cleaning cycles
Your disinfectant rotation protocols must be validated specifically on the furniture materials in your facility, including periodic sporicide use to prevent microbial resistance.
Maintenance and Requalification
Ongoing Requirements:
- Documented cleaning schedules and procedures
- Regular inspections for damage, corrosion, or wear
- Revalidation after repairs or modifications
- Traceability records for all maintenance activities
- Inclusion in facility revalidation (every 1-3 years)
Critical: Remove any furniture showing surface damage, rust, or degradation from service immediately. Compromised surfaces represent both contamination risks and GMP violations that can impact regulatory compliance.
Selecting GMP-Compliant Furniture Suppliers
Key Supplier Qualifications
Verify suppliers meet these critical criteria:
Certifications and Experience:
- ISO 9001 certification for quality management systems
- Demonstrated experience in pharmaceutical/cleanroom furniture
- Understanding of GMP requirements and regulatory expectations
- References from similar GMP facilities
Technical Capabilities:
- In-house engineering for custom designs
- Quality control processes for materials and fabrication
- Ability to provide complete documentation packages
- Post-installation support and maintenance services
Documentation to Request from Suppliers
Essential Documentation:
- Material certificates with composition and finish specifications
- Design drawings showing all GMP-compliant features
- Cleaning and maintenance guidelines specific to materials used
- Chemical compatibility data for facility disinfectants
- IQ/OQ templates and support
- References from pharmaceutical/biotech installations
ACH Engineering's Turnkey Approach
ACH Engineering provides comprehensive cleanroom solutions including specification and installation of GMP-compliant furniture as part of turnkey controlled environment projects.
As members of the International Society for Pharmaceutical Engineering (ISPE), the team understands the critical integration between furniture, modular cleanroom components, and overall contamination control strategy.
Integrated Approach Benefits:
- All components work together to meet regulatory requirements—from modular walls and walkable ceilings to laboratory furniture and cleanroom coving
- Eliminates compatibility issues between furniture and cleanroom infrastructure
- Ensures furniture specifications align with cleanroom grade requirements from design phase through validation
Frequently Asked Questions About GMP Furniture Standards
What is the difference between GMP and ISO cleanroom standards for furniture?
ISO 14644 focuses on particle classification and air cleanliness, while GMP adds pharmaceutical-specific requirements for materials, design validation, and documentation. GMP furniture must meet ISO standards plus additional requirements for chemical resistance, cleanability validation, and regulatory traceability.
Can wooden furniture ever be used in GMP-classified cleanrooms?
No. Wood is never acceptable because it's porous, cannot be adequately cleaned or validated, and sheds particles while harboring microorganisms. The FDA has specifically cited wooden furniture as a GMP violation in warning letters.
How often should GMP furniture be revalidated or recertified?
Revalidate immediately after any repair, modification, or damage, plus during periodic facility revalidation cycles every 1-3 years depending on risk assessment. Furniture showing degradation must be removed and assessed before returning to service.
What documentation is required to prove furniture GMP compliance?
Essential documentation includes material certificates, design qualification documents, installation qualification records, cleaning validation data, and maintenance logs with complete traceability from installation through the furniture's lifecycle.
Are there cost-effective alternatives to stainless steel for Grade C/D areas?
Yes. High-grade polymers like HDPE, polypropylene, and PVDF work well for Grade C/D areas when properly validated. Powder-coated steel is acceptable for Grade D support areas, but all materials must be validated for chemical compatibility with your disinfectants.
How do you clean and disinfect GMP furniture without damaging it?
Follow validated cleaning procedures using approved disinfectants for your facility. Avoid abrasive materials that scratch surfaces. Rinse thoroughly to prevent residue, dry completely to prevent spotting or corrosion, and rotate disinfectants including sporicides per Annex 1.


