
Introduction
A single door opening in a pharmaceutical cleanroom can release millions of particles into a sterile environment, potentially compromising entire batches of medication or critical research. In regulated industries like pharmaceuticals and biotech, even brief lapses in contamination control can result in costly product losses and regulatory violations.
That's where cleanroom airlock doors come in. These specialized systems serve as the first line of defense, acting as transitional barriers between controlled environments and uncontrolled corridors. Using interlocked double doors, they prevent direct air exchange while maintaining critical pressure differentials that keep contaminants at bay.
This article explores how cleanroom airlocks work, the four main configuration types, essential design considerations including pressure management and door materials, performance metrics that validate effectiveness, and selection criteria to match the right system to your facility's specific requirements.
TLDR: Key Takeaways
- Airlocks reduce particle migration by 95% through interlocked doors and pressure control
- Four airlock types serve distinct needs: cascading, bubble, sink, and dual compartment
- Design specs: 10-15 Pa pressure differential, 20-60 ACH, and 15+ second interlocks
- 316L stainless steel with electropolished finishes is the industry standard for pharmaceutical applications due to superior corrosion resistance
- Validation requires IQ/OQ/PQ testing plus 6-12 month re-certification cycles
What is a Cleanroom Airlock?
A cleanroom airlock is a transitional space—also called an anteroom or vestibule—that separates controlled environments from corridors or adjacent spaces.
The fundamental principle is simple: two doors in series that are interlocked to prevent simultaneous opening, thereby maintaining pressure differentials and preventing direct air exchange between zones.
These systems serve four critical functions:
- Contamination control - Creating a buffer zone that traps particles before they reach critical areas
- Pressure cascade maintenance - Preserving the pressure hierarchy between cleanroom classifications
- Personnel/material transition point - Providing a controlled space for movement between zones
- Gowning area - Offering a dedicated space for donning protective garments in some configurations
These functions aren't just theoretical—research confirms their effectiveness. Studies using smoke visualization and particle counting show that airlocks with 20-25 Pa pressure differentials and 16-second door delays reduce particle migration to nearly negligible levels.
Without airlocks, particle migration reaches approximately 50%. With properly configured systems, that drops to less than 5%.
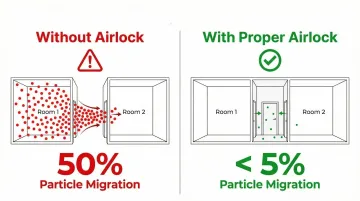
Why airlocks are required across industries:
ISO 14644 standards, FDA aseptic processing guidelines, and EU GMP Annex 1 all require or strongly recommend airlocks for facilities maintaining different cleanliness classifications. The FDA specifically states that pressure differentials of at least 10-15 Pa must be maintained between adjacent rooms of differing classification to prevent contamination migration during normal operations.
Without airlocks, door operation and personnel movement create "dynamic disturbances" that transport particles deep into cleanrooms, compromising sterile manufacturing and research processes.
Types of Cleanroom Airlocks
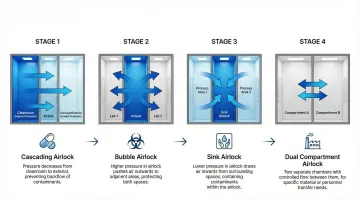
Cascading Airlocks
Cascading airlocks maintain progressively increasing pressure from corridor → airlock → cleanroom, creating a "pressure cascade" where air flows from the cleanest space outward. This configuration protects the cleanroom from corridor contamination.
Typical applications:
- Pharmaceutical manufacturing facilities
- Sterile compounding pharmacies
- Biotech production suites
- Medical device manufacturing
Regulatory guidance recommends maintaining pressure differentials of 10-15 Pa (0.04-0.06 inches w.c.) between each space in the cascade.
This ensures air leakage flows from clean to less clean areas, preventing contaminant ingress.
Bubble Airlocks
Bubble airlocks maintain the highest pressure in the airlock itself, with lower pressure in both the cleanroom and corridor. Air flows outward from the airlock into both adjacent spaces, creating a protective "bubble" that prevents contamination from entering either space.
This design prevents cross-contamination between two spaces that both require protection. Common applications include BSL-3/4 laboratories handling infectious agents, containment facilities with multiple hazard zones, and aseptic processing areas requiring maximum protection.
The bubble configuration shines when both adjacent spaces need isolation from each other, not just unidirectional protection.
Sink Airlocks
Sink airlocks maintain the lowest pressure in the airlock space, with higher pressure in both the cleanroom and corridor. Air flows inward from both sides, containing contaminants within the airlock and preventing outward migration.
Function and applications:
- Contains toxic, infectious, or hazardous materials within the airlock
- Isolation rooms for infectious disease patients
- Potent compound handling in pharmaceutical manufacturing
- Facilities processing carcinogenic or mutagenic substances
Biocontainment zones (BSL-3/4) require negative pressure sink configurations. This ensures no hazardous agents escape to adjacent spaces or corridors.
Dual Compartment Airlocks
Dual compartment systems feature separate entry and exit airlocks with independent pressure control, creating two sequential buffer zones between the cleanroom and corridor.
Advantages:
- Enhanced contamination control through double-barrier protection
- Improved workflow efficiency by separating incoming and outgoing traffic
- Dedicated spaces for gowning (entry) and degowning (exit)
- Reduced pressure disruption from door operations
Research comparing airlock effectiveness ranks dual-compartment designs highest for contamination control.
These systems are preferred in high-volume pharmaceutical manufacturing, semiconductor fabrication facilities, and other applications where continuous personnel flow must be managed without compromising cleanroom integrity.
Key Design Considerations for Airlock Doors
Pressure Differential Management
Maintaining proper pressure differentials is the cornerstone of airlock effectiveness. The industry consensus, based on FDA guidance and EU GMP Annex 1, specifies 10-15 Pa (0.04-0.06 inches w.c.) between adjacent spaces of different classifications.
How pressure differentials are achieved:
- Balanced supply, return, and exhaust airflow through properly sized HVAC systems
- Precise control of air volumes entering and leaving each space
- High-integrity door seals that minimize leakage when closed
The challenge of door operation:
When doors open, pressure differentials effectively disappear. Air leakage through an open doorway vastly exceeds leakage through closed door cracks—by orders of magnitude.
Even fast-response Variable Air Volume (VAV) systems with 1-3 second response times cannot prevent the initial contamination surge during door cycles. Recovery to stable pressure conditions takes 15-20 minutes for particle counts to return to specification, though airflow recovery may occur within 2-3 minutes.
This is why time delays between door openings are critical—the airlock volume and air change rate provide the primary defense during the vulnerable open-door phase.
Door Interlocking Mechanisms
Given these pressure challenges during door operation, interlocks prevent both airlock doors from opening simultaneously, preserving the pressure cascade and contamination barrier.
Mechanism types:
- Mechanical interlocks - Physical barriers (rods, latches) that mechanically prevent the second door from opening until the first fully closes. Simple and robust but less flexible.
- Electrical interlocks - Sensor-based systems using magnetic locks or strikes controlled by programmable logic. Offers flexibility and integration with access control systems.
Best practice: Time-delay interlocks
Research demonstrates that requiring one door to remain closed for 15+ seconds before the second door can open significantly reduces particle migration.
Many pharmaceutical facilities require a delay equivalent to 1-2 air changes (1-3 minutes at 20-60 ACH) to allow purging of contaminants introduced during the first door opening.
Emergency egress requirements:
Building codes (NFPA 101, IBC) mandate that interlocks must fail-safe in emergencies. Systems must include manual release devices (push-to-exit buttons) within 5 feet of doors that interrupt power to locks for at least 30 seconds.
Upon fire alarm activation or power loss, all interlocked doors must unlock automatically to ensure safe egress.
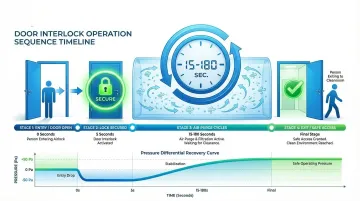
Airflow Patterns and Ventilation Design
Proper airflow within the airlock sweeps contaminants toward return or exhaust grilles, preventing accumulation and migration.
Supply and return placement:
- Supply air generally enters through ceiling-mounted HEPA or ULPA filters
- Return/exhaust grilles are positioned at low wall or floor level
- Airflow should be directed from the clean side toward the dirty side to push contaminants away from the cleanroom
Air change rates:
FDA and ISPE guidelines recommend:
- ISO 8 (Grade C/D): 20-40 ACH minimum
- ISO 7 (Grade B): 40-60 ACH
- ISO 5 (Grade A): Unidirectional flow at 0.36-0.54 m/s
Higher air change rates reduce the required wait time between door openings. At 60 ACH, a complete air change occurs in one minute; at 20 ACH, it takes three minutes. Some pharmaceutical companies require a two-air-change purge before the second door can open.
Door Sealing and Leakage Control
Gaskets, sweeps, and sealing mechanisms minimize air leakage when doors are closed, allowing HVAC systems to maintain pressure differentials without excessive energy consumption.
Sealing components:
- Perimeter gaskets around the entire door frame
- Floor sweeps or drop seals at the door bottom
- Inflatable seals for high-containment or high-traffic applications where standard friction seals might degrade
Material selection:
Gaskets must withstand aggressive cleaning agents. EPDM (ethylene propylene diene monomer) and Viton (fluoroelastomer) are preferred for their chemical resistance compared to standard rubber, which can degrade when exposed to bleach, hydrogen peroxide vapor, or other disinfectants.
Testing standards (ISO 14644-3, IEST-RP-CC006) provide methods for containment leak testing and pressure differential verification. The functional requirement is that door seals must allow the HVAC system to maintain the required 10-15 Pa differential without exceeding fan capacity or creating excessive energy costs.
Size and Spatial Configuration
Airlock dimensions are driven by operational needs, gowning protocols, equipment passage requirements, and accessibility codes.
Minimum dimensions:
- Personnel airlocks: Usually 6 feet x 6 feet minimum, though this is industry practice rather than a codified standard
- Material/equipment airlocks: Sized based on the largest cart or equipment to be transferred
- ADA compliance: Doors must have a clear width of at least 32 inches, with maneuvering clearance (usually 60-inch diameter turning space)
Design considerations:
- Door swing direction and clearances
- Space for gowning benches or storage (if the airlock serves as a gowning area)
- Adequate room for personnel to move without touching walls or doors
- Accessibility for individuals with mobility devices
ACH Engineering tailors airlock spatial design to client-specific workflow requirements and equipment dimensions as part of their turnkey cleanroom solutions, ensuring that operational efficiency is not compromised by inadequate sizing.

Door Materials and Construction
Material selection impacts cleanability, durability, chemical compatibility, and long-term maintenance costs. The right material choice depends on your industry requirements, cleaning protocols, and budget constraints.
Understanding how different materials perform in cleanroom environments helps you balance upfront investment against long-term maintenance and replacement costs.
Material Comparison
| Material | Best For | Key Advantages | Limitations |
|---|---|---|---|
| 316L Stainless Steel | Pharmaceutical, biotech, aseptic areas | Contains 2-3% molybdenum for superior corrosion resistance; electropolished surfaces (Ra < 0.4 µm) resist biofilm; withstands sporicides and H₂O₂ vapor | Higher upfront cost |
| 304 Stainless Steel | ISO 7-8 environments, general cleanrooms | Good corrosion resistance; lower cost than 316L; adequate for standard cleaning protocols | Less chemical resistance than 316L |
| Powder-Coated Steel | Electronics cleanrooms, lower classifications | Moderate chemical resistance; budget-friendly option | Coating can chip and shed particles; not suitable for ISO 5/Grade A/B zones |
| Anodized Aluminum | Electronics, general industrial cleanrooms | Lightweight; good corrosion resistance; easier installation | Less durable than 316L in aggressive chemical environments |
Beyond material choice, construction details determine how well doors maintain contamination control over time.
Construction Features
Key design elements that enhance cleanroom door performance:
- Flush surfaces — Smooth, crevice-free construction for easy cleaning and minimal particle harboring
- Vision panels — Tempered glass or polycarbonate windows allow visual confirmation before opening, reducing unnecessary door cycles
- Integrated hardware — Recessed or flush-mounted handles, hinges, and closers that don't protrude or collect contamination
- Smooth finishes — Electropolished or smooth surfaces (not brushed) minimize surface area for microbial attachment
Performance Metrics and Validation
Validating airlock performance ensures systems meet design specifications and regulatory requirements throughout their operational life. Proper validation protects product quality while demonstrating compliance to auditors and regulatory bodies.
Key Performance Metrics
Critical metrics for airlock performance include:
- Maintain pressure differentials of 10-15 Pa under both static and dynamic conditions
- Achieve door seal leakage rates that allow HVAC systems to maintain pressure efficiently
- Limit particle migration to <5% during door cycles
- Return to specification within 15-20 minutes after door operation (recovery time)
These metrics guide the validation process, which follows a structured three-phase approach.
Validation Requirements
Initial qualification establishes baseline performance through:
- IQ (Installation Qualification) — verify equipment is installed per specifications
- OQ (Operational Qualification) — test that systems operate within design parameters
- PQ (Performance Qualification) — confirm the airlock performs as intended under actual operating conditions
Testing methods (ISO 14644-3) validate these qualifications:
- Airflow visualization (smoke tests) to verify directional flow from clean to dirty
- Recovery testing to measure time required to return to classification after particle challenge
- Pressure differential testing across all door configurations
Re-certification intervals (ISO 14644-2) depend on cleanroom classification:
- ISO Class 5 — maximum 6-month intervals
- ISO Class 6-9 — maximum 12-month intervals
Between these periodic validations, continuous monitoring provides ongoing assurance. Critical applications require real-time monitoring of pressure differentials with alarmed excursions. FDA guidance emphasizes that monitoring systems must document all deviations from specification for investigation and corrective action.

Selecting the Right Airlock System for Your Facility
Selecting the right airlock system determines whether your cleanroom meets regulatory standards while supporting efficient operations. The decision requires balancing technical requirements, regulatory compliance, operational workflow, and budget constraints.
Decision Criteria
ISO cleanroom classification requirements:
- ISO 5/Grade A/B: Requires highest integrity systems with 316L stainless steel, 40-60 ACH, and strict interlocks
- ISO 6-8/Grade C/D: Moderate requirements, 20-40 ACH acceptable
- ISO 9: Basic contamination control sufficient
Industry-specific regulations:
- Pharmaceutical/Biotech: FDA aseptic guidance, EU GMP Annex 1, cGMP requirements
- Semiconductor: SEMI standards for particle control
- Medical devices: ISO 13485 quality management requirements
- BSL labs: CDC/NIH biosafety guidelines for containment
Operational workflow patterns:
- High-traffic facilities benefit from dual compartment designs
- Low-traffic labs may use simpler cascading configurations
- 24/7 operations require robust, low-maintenance systems
Budget constraints:
- Initial capital costs vs. lifecycle costs (energy, maintenance, validation)
- Modular systems offer lower upfront costs and future expandability
- Custom-built systems provide optimal performance but higher initial investment
These criteria directly inform which airlock configuration best fits your specific application.
Matching Airlock Type to Application
| Application | Recommended Type | Key Features |
|---|---|---|
| Pharmaceutical manufacturing | Cascading | 10-15 Pa positive pressure, 40-60 ACH, 316L stainless steel |
| BSL-3/4 laboratories | Bubble or Sink | Negative pressure containment, sealed doors, emergency protocols |
| Semiconductor fabrication | Dual Compartment | High ACH, minimal particle shedding materials, automated controls |
| Medical device manufacturing | Cascading | ISO 7-8 compliance, moderate ACH, cost-effective materials |
| Compounding pharmacies | Cascading | USP 797/800 compliance, proper pressure cascades, gowning space |
Implementing the right airlock system requires evaluating these technical requirements against your facility's specific constraints. ACH Engineering provides cleanroom project management services to help facilities assess requirements and implement optimized airlock solutions as part of modular cleanroom systems, aligning technical specifications with operational and regulatory requirements.
Frequently Asked Questions
What is a cleanroom airlock?
A cleanroom airlock is a transitional space with two interlocked doors that prevents contamination migration between a controlled environment and adjacent spaces. The interlock ensures only one door can open at a time, maintaining pressure differentials that keep contaminants from entering critical areas.
What are the different types of air flow in a clean room design?
Cleanrooms use unidirectional (laminar) flow for ISO Class 5 and cleaner, where air moves in parallel streamlines at 0.36-0.54 m/s, or non-unidirectional (turbulent) flow for ISO Class 6-8 with proper supply/exhaust placement. Airlocks typically use non-unidirectional flow with 20-60 air changes per hour.
How do door interlocking mechanisms work?
Interlocks use mechanical (physical barriers like rods) or electrical (sensor-based magnetic locks) systems to ensure only one door opens at a time. When the first door opens, the second remains locked until the first fully closes, preserving pressure differentials and preventing direct air exchange.
What pressure differential should be maintained in a cleanroom airlock?
Industry standards recommend 10-15 Pa between adjacent spaces of different classifications. ISO 14644-4 allows 5-20 Pa, but FDA and EU GMP guidance specifically recommend the 10-15 Pa range for pharmaceutical and biotech applications.
What door materials are best for cleanroom airlocks?
316L stainless steel with electropolished finish is best for pharmaceutical and biotech applications requiring chemical resistance and frequent cleaning. Powder-coated steel or anodized aluminum work for electronics cleanrooms but pose higher particle shedding risks.
How often should cleanroom airlock systems be validated?
Initial qualification (IQ/OQ/PQ) is required at installation, followed by periodic re-certification based on ISO classification: every 6 months for ISO Class 5, and annually for ISO Class 6-9. Critical applications also require continuous monitoring of pressure differentials with alarmed excursions for immediate investigation.


