
Introduction: The Drug Development Speed Challenge
The pharmaceutical industry faces a stark reality: every day of delay costs approximately $800,000 USD in lost prescription sales and $40,000 USD in direct clinical trial expenses. With drug development timelines averaging 10 to 15 years from discovery to market, infrastructure bottlenecks represent existential risks for emerging biotechs and costly delays for established pharma.
Traditional cleanroom construction is one of the worst bottlenecks in this process. Building a dedicated facility typically requires 12 to 18 months for design, construction, and qualification — with large-scale projects stretching to five years. The upfront capital investment runs approximately $2.5 million USD for a single cleanroom environment, a prohibitive barrier for most startups.
Cleanrooms-on-demand are changing this equation — compressing facility access from years to weeks while converting fixed capital costs into flexible operational expenses. This article covers how on-demand cleanroom models work, what pharmaceutical and biotech teams should look for, and how this approach fits into modern drug development timelines.
TLDR
- Cleanrooms-on-demand eliminate 12–18 month construction timelines, providing access in weeks
- Upfront capital costs of USD $2.5M+ convert into flexible, pay-as-you-go operational expenses
- Process control and IP remain yours — only the infrastructure is shared
- Pre-validated facilities reduce compliance burden and accelerate time-to-market
- Modular solutions scale quickly to match changing trial requirements
What Are Cleanrooms-on-Demand?
The Service Model Explained
Cleanrooms-on-demand (COD) moves away from the traditional "build-own-operate" approach to pharmaceutical manufacturing infrastructure. Instead of constructing dedicated facilities, companies rent fully equipped, GMP-compliant cleanroom suites on flexible terms—similar to how cloud computing replaced on-premise servers.
The critical distinction between COD and Contract Development and Manufacturing Organizations (CDMOs) is control. With CDMOs, the vendor performs the manufacturing. With cleanrooms-on-demand, sponsors retain complete control over their processes, personnel, and intellectual property while utilizing the provider's infrastructure.
What's Included in COD Offerings
Standard cleanroom-on-demand packages typically bundle:
- GMP-classified suites rated ISO 5, 7, or 8 depending on application requirements
- Integrated utilities: HVAC systems, compressed air, vacuum, and purified water
- Compliance infrastructure covering environmental monitoring, waste management, and gowning facilities
- Pre-validated systems with IQ and OQ already completed
The provider maintains the facility's compliance status, handles routine maintenance and recertification, and absorbs utility costs. This removes a significant operational burden from facility ownership.
Common Use Cases
Cleanrooms-on-demand serve multiple pharmaceutical development scenarios:
- Producing small batches of investigational drugs for early-phase clinical trials
- Optimizing formulations during active product development
- Validating manufacturing processes during scale-up before committing to a full facility
- Supporting tech transfer when moving processes between development stages or locations
- Adding overflow capacity during peak demand periods or facility renovations
How Cleanrooms-on-Demand Accelerate Drug Development Timelines
Timeline Compression: The Numbers
The timeline difference between facility approaches is stark — and it directly shapes when a drug candidate reaches patients:
| Approach | Typical Timeline | Acceleration | |----------|------------------|--------------|\n| Traditional construction | 12-18 months (up to 5 years for large facilities) | Baseline | | Modular/prefabricated | 9-18 months | 30-50% reduction | | Cleanrooms-on-demand | Immediate to 3 months | 75-90% reduction |
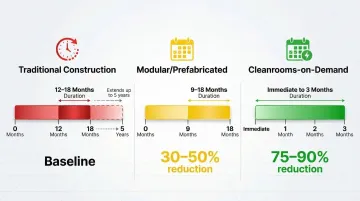
Removing facility construction from the critical path means drug candidates can move from discovery to clinical material production without waiting on concrete and HVAC. That's months returned to the development schedule — not weeks.
Real-World Impact: Case Studies
Pfizer's Biosimilar Acceleration: Using a flexible manufacturing solution for a biosimilar project, Pfizer completed process transfer and documentation in just 5 months, saving an estimated 6 to 12 months compared to traditional facility establishment.
COVID-19 Response: During the pandemic, modular cleanroom providers delivered functional facilities in 30 weeks—effectively halving the standard 52-60 week pre-pandemic timeline and demonstrating what's possible when urgency demands innovation.
Moderna's Strategic Approach: Moderna utilized cleanroom-on-demand facilities as anchor capacity while their dedicated manufacturing sites were under construction, maintaining development momentum without infrastructure delays.
Strategic Flexibility Benefits
Beyond raw speed, cleanrooms-on-demand provide critical operational agility:
- Pivot quickly when trial results require formulation changes or dose adjustments
- Scale capacity up or down as clinical enrollment shifts
- Position manufacturing closer to clinical sites or regulatory agencies as needed
- Avoid large capital commitments before proof-of-concept is established
For emerging biotechs especially, that last point matters most. Committing tens of millions to a dedicated facility before Phase II data exists is a calculated gamble — cleanrooms-on-demand let teams stay nimble until the science justifies the investment.
Cost Benefits: Traditional vs On-Demand Cleanrooms
Capital Expenditure Reality
Traditional cleanroom construction demands substantial upfront investment that many startups cannot secure:
Construction costs in major life sciences hubs:
- Core shell construction: $675 to $1,200 per sq. ft.
- GMP fit-out: Additional $300 to $650 per sq. ft.
- ISO 5 cleanrooms: $1,000+ per sq. ft. due to extreme air change requirements
For a modest 2,000 sq. ft. ISO 7 cleanroom, total capital expenditure easily exceeds $2.5 million before equipment, validation, or operational costs.
The OpEx Advantage
Cleanrooms-on-demand convert these fixed capital costs into variable operational expenses, improving cash flow for early-stage biotechs. Instead of raising millions for infrastructure, companies can allocate capital to R&D, clinical trials, and regulatory activities that directly advance their pipeline.
The "pay for what you use" model aligns costs with production needs. For early-phase trials requiring only small batches, renting cleanroom access for weeks or months costs a fraction of building dedicated capacity that sits idle between campaigns.
Total Cost of Ownership Comparison
Construction is just the starting point — traditional facilities carry substantial ongoing costs that compound over time:
- Energy consumption: Pharmaceutical cleanrooms use 15x more energy than commercial buildings, with HVAC representing over 50% of electricity usage
- Maintenance and recertification: Continuous spending on equipment calibration, filter replacement, and periodic revalidation
- Specialized staffing: Quality assurance personnel, facility engineers, and compliance specialists
- Depreciation: Traditional facilities depreciate over 39 years, while modular systems often qualify for 7-year accelerated depreciation

Cleanroom-on-demand models bundle these expenses into the rental fee, providing cost predictability and eliminating unplanned capital costs when critical equipment fails or regulations change.
Key Considerations When Choosing Cleanrooms-on-Demand
Location and Logistics
Geographic proximity to your operations affects both cost and convenience. Consider:
- Distance to your R&D facilities and key personnel
- Proximity to clinical trial sites for fresh product delivery
- Access to specialized supply chains and vendor networks
- Regional regulatory environments and inspection frequency
Technical Capabilities
Evaluate providers based on:
- Confirm ISO classifications match your product requirements — ISO 7 or ISO 8 for most pharmaceutical applications, ISO 5 for aseptic processing
- Verify batch size capacity today and room to scale if trial volumes increase
- Confirm support for your existing manufacturing equipment or flexibility for custom installations
Provider Credentials
Due diligence on the provider's compliance track record is critical. Look for:
- Memberships such as ISPE (International Society for Pharmaceutical Engineering) showing active engagement with pharmaceutical standards
- Engineering credentials like PEO (Professional Engineers Ontario) confirming qualified technical staff
- Regulatory inspection history and any warning letters or compliance issues
- Client references from similar pharmaceutical or biotech companies
ACH Engineering holds memberships in both ISPE and PEO, which means their team stays current on pharmaceutical engineering standards and regulatory requirements — a useful benchmark when vetting any provider.
Turnkey Solution Value
The best providers offer comprehensive turnkey solutions that include:
- Fast, uncomplicated design services tailored to your process requirements
- Complete installation with minimal disruption to your timeline
- Pre-validated systems reducing your qualification burden
- Ongoing compliance support and facility management
ACH's modular cleanroom solutions follow this model: prefabricated components built offsite compress installation timelines from months to weeks, with full validation support included to keep projects regulatory-ready.
ISO Standards and Compliance in COD Facilities
Understanding ISO 14644-1 Classifications
The ISO 14644-1 standard defines cleanroom classifications based on maximum allowable airborne particle concentrations:
| ISO Class | Max Particles/m³ (≥0.5 µm) | Pharmaceutical Application |
|---|---|---|
| ISO 5 | 3,520 | Aseptic processing, sterile filling |
| ISO 7 | 352,000 | Compounding, support areas |
| ISO 8 | 3,520,000 | Gowning rooms, preparation areas |

Most pharmaceutical manufacturing requires ISO 7 or ISO 8 environments, with ISO 5 reserved for critical aseptic operations. The specific classification depends on product type, dosage form, and intended route of administration.
Regulatory Alignment
FDA guidance aligns ISO 5 with Class 100 critical processing areas, EU GMP Annex 1 links Grade A/B/C/D classifications to ISO particle limits, and Health Canada's GUI-0119 follows the same ISO framework for domestic manufacturers. Reputable COD providers maintain continuous compliance across these frameworks through:
- Real-time environmental monitoring tracking particles, pressure differentials, temperature, and humidity
- Regular recertification with documented particle counts and airflow verification
- Preventive maintenance programs ensuring HVAC and filtration systems perform consistently
- Change control procedures managing any modifications that could affect classification
Pre-Validation Advantages
Meeting these regulatory standards normally requires months of qualification work — which COD facilities have already done for you. The provider has completed Installation Qualification (IQ) and Operational Qualification (OQ) for the facility and utilities before you ever move in.
Your responsibility focuses primarily on Performance Qualification (PQ)—demonstrating that your specific manufacturing process performs consistently within the validated environment. That single shift — from full-stack qualification to process-only PQ — can cut validation timelines by months and meaningfully reduce upfront compliance costs.
The Future of Cleanroom Manufacturing
Industry Trends Toward Flexibility
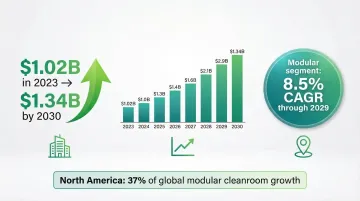
The pharmaceutical industry is shifting decisively toward modular, flexible cleanroom solutions. The U.S. cleanroom technology market in healthcare reached $1.02 billion in 2023 and is projected to grow to $1.34 billion by 2030, driven by demand for adaptable manufacturing capacity.
The modular cleanroom segment is growing even faster, with forecasts showing an 8.5% CAGR through 2029. Pharmaceutical companies are increasingly treating cleanroom capacity as a variable resource rather than a fixed cost — commissioning space as pipelines demand it, then scaling back or redeploying when priorities shift.
Post-COVID Market Dynamics
The COVID-19 pandemic permanently altered facility planning strategies. The urgent need for vaccine manufacturing capacity proved that traditional construction timelines could be compressed dramatically through modular approaches and parallel processing.
That experience normalized rapid deployment as a legitimate strategy — not just an emergency workaround. Today, procurement teams actively plan for cleanrooms-on-demand as part of their standard infrastructure toolkit, not as a fallback option.
North American Market Leadership
North America accounts for 37% of global modular cleanroom growth, driven by several factors:
- High concentration of biotech startups requiring flexible, capital-efficient infrastructure
- Stringent FDA regulations necessitating high-quality controlled environments
- Established provider networks offering turnkey solutions
- Strong venture capital ecosystem supporting innovative infrastructure models
ACH Engineering supports this demand across Canada and the broader North American market, delivering end-to-end modular cleanroom projects — from design and engineering through installation — with the regulatory compliance and project management that pharmaceutical and biotech clients need.
Frequently Asked Questions
What are cleanrooms-on-demand?
Cleanrooms-on-demand are fully equipped, GMP-compliant pharmaceutical manufacturing spaces available for rent on flexible terms. Unlike contract development and manufacturing organizations (CDMOs), you retain full control over your processes and IP while using the provider's pre-validated infrastructure.
How do cleanrooms-on-demand speed up drug development?
By eliminating 12-18 month construction timelines, COD facilities provide immediate or near-immediate access to manufacturing capacity. This removes infrastructure from the critical path, allowing clinical material production to begin as soon as formulations are ready.
How much do cleanrooms cost?
Traditional cleanroom construction runs $2.5M+ in upfront capital, plus ongoing operational costs. Cleanrooms-on-demand shift this to an operational expense — rental rates vary by location, ISO classification, and included services, but the model eliminates large capital commitments for early-stage companies.
What ISO classification do pharmaceutical cleanrooms need?
Most pharmaceutical applications require ISO 7 or ISO 8 environments. Aseptic processing and sterile filling require ISO 5. The specific classification depends on your product type, dosage form, and regulatory requirements for your therapeutic category.
How long does it take to access a cleanroom-on-demand vs building one?
Cleanroom-on-demand access typically takes weeks to 3 months depending on availability and customization needs. Traditional construction requires 12-18 months minimum, with large facilities taking up to 5 years—a gap that directly affects clinical timelines and program momentum.
Why is there no cardboard in cleanrooms?
Cardboard sheds particles and fibers that compromise ISO classifications and contaminate pharmaceutical products. It's also porous, absorbing moisture and harboring bacteria. All materials entering cleanrooms must be transferred from cardboard shipping containers in airlocks or staging areas.
Ready to accelerate your drug development timeline? ACH Engineering designs and builds modular cleanroom solutions for pharmaceutical, biotech, and life sciences applications across North America. From initial design through installation and validation support, our team delivers ISO-compliant facilities faster than traditional construction — without the capital overhead. Contact us to discuss how a modular cleanroom can remove infrastructure from your critical path.


