
Introduction
A single contamination incident can trigger product recalls, production shutdowns, and regulatory enforcement actions that cost facilities hundreds of thousands of dollars. Root cause analyses consistently identify furniture surfaces as the most significant risk vector for microbial contamination in pharmaceutical and biotech cleanrooms.
Proper cleaning directly impacts your bottom line beyond contamination control. While furniture replacement becomes justified when repair costs exceed 40-60% of replacement value, preventive maintenance through material-specific cleaning protocols extends furniture lifespan and protects capital investments.
This guide provides comprehensive, regulation-compliant cleaning protocols for laboratory furniture across pharmaceutical, biotech, and research facilities. You'll find material-specific procedures, safety requirements, disinfectant selection guidelines, and documentation practices that meet Health Canada GUI-0001, FDA 21 CFR 211.67, and ISO 14644-5 standards.
TLDR
- Match cleaning agents to surface materials—bleach corrodes stainless steel, solvents damage powder coatings
- FDA and Health Canada mandate written cleaning procedures with documented schedules and verification
- Minimum PPE includes chemical-resistant gloves, safety goggles, and lab coats; consult SDS for each agent
- Cleaning removes dirt; disinfection kills microorganisms—both require proper contact times (typically 3-10 minutes)
- Document everything: date, time, personnel, products used, and observations for regulatory inspection readiness
Why Proper Cleaning of Laboratory Furniture Matters
Contaminated laboratory furniture creates three critical risks: compromised research data, regulatory non-compliance, and shortened equipment lifespan. When furniture surfaces harbor residual chemicals or biological contaminants, they directly compromise experimental results and pharmaceutical product quality.
Regulatory bodies explicitly require furniture cleaning protocols:
- FDA 21 CFR 211.67 requires written procedures for cleaning and maintenance, including schedules, methods, and responsibility assignments
- Health Canada GUI-0001 specifies documented sanitation programs specifying cleaning intervals and products
- ISO 14644-5 (2025 revision) mandates an Operations Control Programme (OCP) integrating cleaning, disinfection, and monitoring schedules
These compliance requirements tie directly to operational costs and equipment longevity. Proper maintenance delivers measurable ROI through extended furniture lifespan.
Epoxy resin countertops cost $300-$500 CAD per linear meter initially but last 15-25 years with proper care, requiring only 1-2 replacements over a 30-year horizon. Facilities that invest in pharmaceutical cleanroom equipment designed for cleanability can dramatically reduce long-term maintenance costs. Compare this to melamine laminate at $80-$150 CAD per linear meter but requiring 3-6 replacements due to seam failures and chemical damage.
The higher upfront investment in quality materials combined with rigorous cleaning protocols reduces total lifecycle costs by 40-60%.
Contamination incidents carry catastrophic costs beyond cleaning: product recalls, production shutdowns, and regulatory fines can reach millions of dollars. Cleanroom operations studies consistently identify furniture as the primary contamination risk when cleaning protocols fail.

Safety Preparation and PPE Requirements Before Cleaning
Essential PPE for Laboratory Furniture Cleaning
Protect personnel with appropriate personal protective equipment before beginning any cleaning operation.
Chemical-Resistant Gloves (Selection by Compatibility):
| Glove Material | Compatible With | Important Notes |
|---|---|---|
| Nitrile | Ethanol, isopropyl alcohol, most disinfectants | General-purpose protection |
| Butyl rubber | Acetone, aldehydes, ketones | For aggressive solvents |
| PVC | Mild acids, bases | Never use with acetone (poor resistance) |
Additional Required PPE:
- Safety goggles providing full eye protection against splashes
- Lab coats made from chemical-resistant material covering arms and torso
- Respiratory protection for volatile disinfectants like isopropyl alcohol in poorly ventilated areas (NIOSH REL: 400 ppm TWA)
- Closed-toe, chemical-resistant footwear
Pre-Cleaning Risk Assessment
Once PPE is selected, complete a risk assessment before starting any cleaning procedure.
Identify Surface Hazards:
- Check for residual chemicals or biological materials on furniture surfaces
- Consult Safety Data Sheets (SDS) for all cleaning agents you'll use
- Verify proper ventilation systems are operational
- Confirm emergency eyewash stations and safety showers are within 10 seconds' reach
After identifying hazards, prepare the work area for safe cleaning.
Prepare the Work Area:
- Remove all equipment, materials, and personal items from surfaces
- Secure or disconnect electrical cords and equipment
- Post "Cleaning in Progress" signage to restrict access
- Stage waste containers and verify cleaning supplies are cleanroom-appropriate (low-lint, non-particle-generating)
Documentation: Keep SDS sheets readily accessible for all cleaning agents, as OSHA standards require. Review each product's specific hazards, required PPE, and emergency procedures before use.

Material-Specific Cleaning Protocols for Laboratory Furniture
Epoxy Resin Laboratory Furniture
Epoxy resin surfaces offer excellent chemical resistance but require specific cleaning approaches to maintain their integrity.
Daily Cleaning Protocol:
- Wipe surfaces with mild soap and warm water using soft, lint-free cloths
- Rinse thoroughly with clean water to remove soap residue
- Dry with clean, lint-free towels
Weekly Disinfection:
- Apply 70% isopropyl alcohol to pre-cleaned surfaces
- Allow 3-10 minute contact time per manufacturer specifications
- Wipe dry or allow to air dry
Tough Stain Removal:
- Acetone, mineral spirits, or Crystal Simple Green are safe for epoxy resin
- Always spot-test in a hidden area first
- Apply with soft cloth, wipe immediately, and rinse thoroughly
Critical Warnings - Never Use:
- Abrasive cleaners, steel wool, or scouring pads (will dull the surface)
- Wax-based polishes (leave residue)
- Harsh alkaline solutions above pH 12
- Prolonged heat exposure above 205°F (96°C)
Phenolic Resin Laboratory Furniture
Phenolic resin takes durability further. It provides superior chemical resistance and handles more aggressive cleaning methods than epoxy.
Routine Cleaning:
- Use non-abrasive household cleaners with warm water
- Apply with soft cloths or non-abrasive sponges
- Rinse thoroughly and dry
Deep Sanitization:
- Steam cleaning is safe and effective for phenolic surfaces
- Surfaces resist moisture and do not support bacterial growth
- For disinfection, use 60-70% alcohol solutions, aldehydes, or quaternary ammonium compounds
Stubborn Stain Treatment:
- Small amounts of acetone, white spirit, or turpentine can remove solvent-based marks
- Apply sparingly with soft cloth
- Wipe clean immediately
Materials to Avoid:
- Bleach-based cleaners (can discolor surface)
- Abrasive powders or polishing agents
- Sharp tools or scrapers that can gouge surfaces
- If deep scratches occur, professional refinishing may be required
Stainless Steel Laboratory Furniture
Stainless steel demands careful attention. Improper cleaning can trigger corrosion and permanently damage the surface.
Daily Cleaning Protocol:
- Wipe in the direction of the grain using soft, lint-free cloths
- Use mild detergent and hot water
- Rinse thoroughly with clean water
- Dry immediately to prevent water spots
Stain Removal:
- Light stains: White vinegar or baking soda paste applied with soft cloth
- Polish: Commercial stainless steel cleaners applied in direction of grain
- Rinse thoroughly after any treatment
Critical Warnings - Prevent Irreversible Damage:
Chlorine bleach and chloride-containing cleaners cause irreversible pitting corrosion on stainless steel. Even 5% bleach solutions can cause pitting within hours.
If bleach must be used:
- Dilute immediately to residual chlorine levels below 15-20 ppm
- Limit contact time to absolute minimum
- Rinse immediately and thoroughly with sterile water or 70% isopropyl alcohol
- Grade 316L offers better resistance than 304 but remains vulnerable
Never Use:
- Steel wool or abrasive pads (scratch protective chromium oxide layer)
- Hydrochloric acid or other chloride-containing acids
- Contaminated water (mineral deposits cause staining)
Powder-Coated Steel and Modular Cleanroom Furniture
Powder-coated surfaces combine durability with easy maintenance—when you follow proper protocols.
Proper Cleaning Method:
- Use warm water with mild, pH-neutral detergent (pH 5-8)
- Apply with soft cloths or non-abrasive sponges
- Rinse thoroughly and dry
Immediate Spill Response: Clean spills immediately to prevent coating degradation. Prolonged chemical exposure can compromise the powder coating and expose underlying metal to corrosion.
For pharmaceutical and biotech cleanrooms, modular furniture with seamless powder-coated surfaces eliminates particle entrapment points. ACH engineers these surfaces specifically for GMP compliance, reducing cleaning time while maintaining sterility standards.
Critical Incompatibilities: Solvents and strong alkaline cleaners destroy powder coatings.
Never use:
- Ketones or esters (acetone, MEK)
- Strong alkaline detergents (pH > 9)
- Trichloroethylene or other chlorinated solvents
- Abrasive cleaners or polishes
Laminate and Solid Surface Countertops
Laminate Cleaning:
- Use warm water and non-abrasive cleaners
- Stubborn stains may be treated with hypochlorite bleach for 1.5 minutes maximum, followed by thorough rinsing
- Avoid prolonged water exposure at seams (causes swelling and delamination)
- Protect against heat damage—use trivets for hot items
Solid Surface (Corian) Care:
- Non-porous nature allows use of mild abrasives if needed for tough stains
- Most stains respond to mild soap and water
- Minor scratches and stains can be repaired through professional sanding and refinishing
- Avoid strong acids (>50% acetic acid) and chlorinated solvents (chloroform, methylene chloride)
Cleaning Agent Compatibility by Material
| Material | Safe Agents | Prohibited Agents | pH Range |
|---|---|---|---|
| Epoxy Resin | Soap/water, 70% IPA, acetone, mineral spirits | Abrasives, wax polishes, strong alkalis (pH >12) | 5-8 |
| Phenolic Resin | Non-abrasive cleaners, alcohols, aldehydes, steam | Bleach, abrasive powders, sharp tools | N/A |
| Stainless Steel | Mild detergent, vinegar, baking soda, commercial SS cleaners | Bleach, chloride cleaners, HCl, steel wool | N/A |
| Powder-Coated Steel | pH-neutral detergent, warm water | Acetone, ketones, esters, alkalis (pH >9) | 5-8 |
| Laminate | Non-abrasive cleaners, brief bleach exposure | Acid drain cleaners, >50% formic acid, prolonged water | N/A |
| Solid Surface | Mild soap, mild abrasives (if needed) | Strong acids (>50%), chlorinated solvents | N/A |

General Best Practices for Laboratory Furniture Cleaning
Establish Frequency-Based Cleaning Protocols
Cleaning frequency should be risk-based, aligning with ISO cleanroom classifications and usage intensity.
Daily Protocols:
- Wipe down all work surfaces after each use
- Clean high-touch surfaces in pharmaceutical cleanrooms
- Address spills immediately upon occurrence
- ISO Class 5 primary engineering controls: beginning of shift, before each batch, every 30 minutes during continuous work
Weekly Protocols:
- Deep cleaning and disinfection of all work surfaces
- Thorough cleaning of storage areas and furniture sides
- Rotate disinfectants to prevent microbial resistance
Monthly Protocols:
- Comprehensive inspection for damage, wear, or coating degradation
- Deep cleaning of walls, ceilings, and shelving in ISO Class 7/8 areas
- Verification testing (ATP or surface sampling)
Quarterly Protocols:
- Deep maintenance and refinishing if needed
- Complete documentation review
- SOP effectiveness evaluation

Disinfectant Selection and Contact Time
Effective contamination control requires distinguishing between cleaning and disinfection.
Cleaning vs. Disinfection:
- Cleaning removes visible dirt, debris, and organic matter
- Disinfection kills microorganisms
- Critical principle: Disinfectants only work on pre-cleaned surfaces—organic matter inactivates many biocides
Required Contact Times:
- 70% Isopropyl Alcohol: 1-3 minutes for vegetative bacteria and viruses
- Quaternary Ammonium Compounds: 10 minutes per EPA labels
- Hydrogen Peroxide: 1-10 minutes depending on formulation
- Always follow manufacturer specifications—insufficient contact time makes disinfection ineffective
Disinfectant Rotation Strategy:
Current best practice per USP <1072> and PDA TR70 recommends rotating a standard disinfectant (phenolic or quaternary ammonium compound) with a sporicidal agent (peracetic acid or bleach) on a weekly or monthly basis.
This rotation prevents microbial resistance while addressing spore-forming organisms.
Residue Management:
After disinfection, rinse surfaces with sterile water or 70% IPA after disinfection to remove residues that can inhibit subsequent disinfectants or damage furniture surfaces.
Correct Cleaning Technique and Sequence
Work Pattern:
- Cleanest to dirtiest areas: Prevents cross-contamination
- Top to bottom approach: Gravity carries contaminants downward
- Overlapping strokes: Ensures complete coverage
Two-Bucket Method:
- Bucket 1: Fresh cleaning solution
- Bucket 2: Rinse water
- Never return contaminated cloths to cleaning solution bucket
Material Disposal:
Dispose of cleaning materials according to facility waste protocols. Use appropriate containers for hazardous waste and document disposal of materials contaminated with controlled substances.
Spill Response Protocols
Proper disposal procedures connect directly to spill management. When spills occur, immediate response is critical.
Immediate Response:
- Contain spill to prevent spreading
- Identify chemical or biological agent involved
- Select appropriate cleaning agent based on compatibility
- Don appropriate PPE before cleanup
- Clean from outside edges toward center
- Dispose of contaminated materials properly
Documentation Requirements:
- Record date, time, and location of spill
- Document materials involved and cleaning agents used
- Note any deviations from standard protocols
- Escalate to safety officer for hazardous materials
- Perform decontamination verification before returning area to service
Verification and Validation of Cleaning Effectiveness
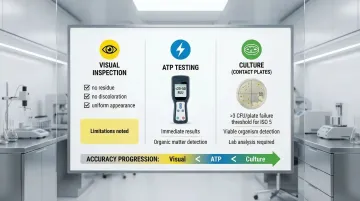
Visual inspection alone cannot validate cleaning effectiveness. Multiple verification methods ensure accuracy:
Visual Inspection Criteria:
- Surfaces free of visible residue, streaks, or particulate matter
- No discoloration or staining
- Uniform appearance across entire surface
ATP Bioluminescence Testing:
- Detects organic matter as a proxy for cleanliness
- Recommended thresholds for high-risk surfaces: <20-50 RLU
- Test immediately after cleaning and after disinfectant contact time
- Stainless steel surfaces typically allow stricter (lower) RLU limits
Surface Sampling and Culture Methods:
- Contact plates or swabs for viable microorganism detection
- USP <797> standards: >3 CFU/plate for ISO 5 surfaces indicates cleaning failure
- Frequency based on risk assessment and regulatory requirements
Cleaning Logs:
Maintain detailed records including date, time, personnel, areas cleaned, products used, lot numbers, observations, and verification results. Retain for 3-5 years minimum for regulatory inspection readiness.
Maintenance Schedules and Documentation for Compliance
Creating Facility-Specific Cleaning Schedules
Develop cleaning schedules based on three critical factors:
Lab Type and Classification:
- Pharmaceutical cleanrooms (ISO Class 5-7): Daily disinfection of all work surfaces, weekly sporicidal rotation
- Biotech research labs (ISO Class 8): Daily cleaning of high-touch surfaces, weekly comprehensive disinfection
- General research laboratories: Weekly deep cleaning, monthly comprehensive maintenance
Usage Intensity:
- High-traffic areas require more frequent cleaning
- Surfaces exposed to hazardous materials need immediate post-use cleaning
- Dedicated-use equipment may follow less frequent schedules
Regulatory Requirements:
- FDA 21 CFR 211.67 mandates appropriate intervals based on risk
- ISO 14644-5 requires documented OCP with justified frequencies
- Health Canada GUI-0001 specifies cleaning intervals in written programs
Documentation Requirements for Regulatory Compliance
Essential Cleaning Log Elements:
- Date and time of cleaning
- Personnel performing cleaning (names and signatures)
- Specific areas and equipment cleaned
- Cleaning agents used (product names and lot numbers)
- Disinfectants used with contact times
- Observations (damage, wear, contamination)
- Verification results (ATP readings, visual inspection pass/fail)
- Supervisor verification signature
Record Retention: General laboratories maintain cleaning logs for 3 years minimum. Pharmaceutical facilities retain records for 5+ years (often product lifecycle plus additional years per FDA requirements).
Standard Operating Procedures (SOPs): Detailed SOPs should cover:
- Responsibility assignments (who cleans what)
- Approved cleaning agent list with dilution ratios
- Step-by-step cleaning methods
- Frequency matrix based on area classification
- Verification procedures
- Documentation requirements
ACH's Maintenance-Friendly Design Features
ACH's cleanroom designs incorporate maintenance-friendly features that reduce cleaning time and effort:
Seamless Construction: ACH's cleanroom coving systems provide seamless wall-ceiling and wall-floor connections that eliminate sharp corners in compliance with cGMP requirements.
These seamless transitions remove contamination collection points and simplify cleaning procedures.
Accessible Surfaces: Walkable ceiling systems allow maintenance personnel to access HVAC systems, filters, and utilities without compromising the cleanroom environment below, reducing maintenance downtime and preventing contamination during service activities.
Material Selection: Modular cleanroom components use GMP-compliant materials selected for long-term cleanability, supporting efficient cleaning validation and contamination control in pharmaceutical and biotech facilities.
These design considerations reduce total ownership costs by cutting cleaning time up to 30% and extending cleanroom component lifespan.
Frequently Asked Questions
What are the guidelines for laboratory housekeeping?
Key principles include establishing regular cleaning schedules based on lab classification, following material-specific protocols, using appropriate PPE, maintaining detailed documentation for compliance, and adhering to facility SOPs aligned with ISPE and ISO cleanroom standards.
How often should laboratory furniture be cleaned and disinfected?
Pharmaceutical cleanrooms (ISO Class 5-7) require daily disinfection, while general research labs need weekly deep cleaning with monthly comprehensive maintenance. High-traffic surfaces may need cleaning after each use based on facility SOPs and risk assessments.
What cleaning agents are safe for different laboratory furniture materials?
Mild soap and water work for most materials, while 70% isopropyl alcohol suits epoxy and phenolic resin. Stainless steel tolerates most cleaners except chlorine bleach. Always test new products in inconspicuous areas first.
What PPE is required when cleaning laboratory furniture with disinfectants?
Minimum PPE includes chemical-resistant gloves (nitrile for most applications), safety goggles, and lab coats. Respiratory protection may be needed for volatile disinfectants in poorly ventilated areas. Always consult Safety Data Sheets (SDS) for specific requirements.
How do you document cleaning procedures for regulatory compliance?
Documentation must include date, time, personnel signatures, areas cleaned, cleaning agents with lot numbers, contact times, deviations observed, and verification results (ATP readings, visual inspection). Maintain logs for 3-5 years minimum.
What are common mistakes to avoid when cleaning lab furniture?
Critical errors include using incompatible cleaning agents (especially chlorine bleach on stainless steel), insufficient disinfectant contact time, inadequate PPE, and failing to verify cleaning effectiveness through ATP or surface sampling. Always consult material compatibility guidelines.


